* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Document
Survey
Document related concepts
Transcript
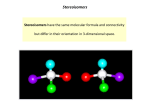
Chapter 6 Stereochemistry Is the study of the static and dynamic aspects of the three-dimensional shapes of molecules. 6.1 Stereogenicity and stereoisomerism 6.1.1 Basic concepts and terminology Constitutional isomers: molecules with same molecular formular but different connectivity between the atoms. e.g.) 1-bromo and 2-bromobutane Stereoisomers: molecules that have the same connectivity but differ in the arrangement of atoms in space. e.g) cis- and trans-2-butene 1. enantiomers: nonsuperimposable mirror images of each other 2. diastereomers: stereoisomers that are not enantiomers - conformational isomers: are interconvertible by rotations about single bonds - configurational isomers: stereochemical isomers including enantiomers and diastereomers. configuration: the relative position or order of arrangement of atoms in space which characterizes a particular stereoisomer. - chiral: any object that is nonsuperimposable with its mirror images - achiral: if an object is not chiral, it is achiral. A molecule is achiral if it is superimposable on its mirror image. A molecule which has a plane of symmetry, a center of symmetry or rotation-reflection symmetry is achiral. An axis of symmetry (C2 axis) -> achiral과 관계 없음 A molecule is achiral if it is superimposable on its mirror image. A molecule which has a plane of symmetry, a center of symmetry or rotation-reflection symmetry is achiral. (나중에 다시 설명) chiral C2 OH Br achiral O Br OH a plane of symmetry (s, S1) Br achiral Br a center of symmetry (i, S2) meso: compounds that contain stereogenic centers but are nevertheless achiral. Classic terminology Optically active: refers to the ability of a collection of molecules to rotate plane polarized light - must have an excess of one enantiomer. Racemic mixture (or racemate): a 50:50 mixture of enantiomers and is not optically active. However, enantiomers that do not have dramatically different refractive indices would not result in measurable rotations. -> in this case, they are optically inactive even though they are chiral. 따라서 optically active란 말은 사용하지 않는 것이 좋음. Chiral center or chiral (asymmetric) carbon: an atom or specifically carbon, respectively, that has four different ligands attached. Chiral carbons exist in molecules that are neither asymmetric nor chiral. Many molecules can exist in enantiomeric forms without having a chiral center. 이 말도 사용하지 않는 것이 좋음. CO2H H OH H OH CO2H chiral center achiral compound More modern terminology Stereocenter (stereogenic center): use this term instead of chiral center, it is stereogenic center if the interchange of two ligands attached to it can produce a new stereoisomer. A non-stereogenic center is one in which exchange of any pair of ligands does not produce a stereoisomer. -> the term ‘stereogenic center’ is broader than the term ‘chiral center’. A CWXYZ center does not guarantee a chiral molecule. However, a CWXYZ group is always a stereogenic center. CO2H H OH H OH CO2H stereogenic center: 두개의 치환기를 바꾸면 stereoisomers 가 생긴다 meso form Typically, a molecule with n stereogenic, tetracoordinate carbons will have 2n stereoisomers - 2n-1 diastereomers that exist as a pair of enantiomers. Epimers: are diastereomers that differ in configuration at only one of the several stereogenic centers. Carbohydrates: a- and b-anomers도 epimers의 한 형태임. 6.1.2 Stereochemical descriptors R, S system (Cahn-Ingold-Prelog system) 1 R1 R2 2 4 R4 R3 3 1 R1 R2 2 3 R4 R3 4 S R rectus (right) sinister (left) higher atomic number: higher priority isotopes (the one with higher mass is assigned the higher priority) Tricoordinate -> stereogenic center phantom atom: the lowest priority S CH3 CH2CH3 H3C H3C high energy barrier CH2CH3 S CH3 S R phantom atom: the lowest priority P CH3 CH2CH=CH2 R high energy barrier CH2CH=CH2 P CH3 S E, Z system lower higher Opposite: E (entgegen) (cf) same: Z (zusammen) If an H atom is on each of the double bond, conventionally, cis and trans can be used. D, L system mainly used for amino acids and carbohydrates Fischer projection Horizontal lines: bonds coming out of the plane of the paper Vertical lines: bonds projecting behind the plane of the paper The most oxidized group: top CH2OH (carbohydrates) or R (amino acids): bottom D: dextro, right L: levo, left L D D D L Natural amino acids: L-amino acids Important point No direct relationship between the R/S and D/L and the sign of optical rotation of the molecule. Helical descriptors – M, P system Many chiral molecules lack a conventional center that can be described by R/s or E/Z. -> typically helical, propeller, screw-shaped structures -> a right-handed helix (clockwise): P (plus), a left handed helix (anti-clockwise): M (minus) H H CH3 Cl H3C NO2 NO2 CH3 CH3 H3C H H 6.1.3 Distinguishing enantiomers Chiral column chromatography Enantiomeric excess = (Xa – Xb) x 100, Xa: mole fraction of a, Xb: mole fraction of b High field NMR spectroscopy with chiral shift reagents NMR spectroscopy of derivatives that are diastereomeric Chromatography (HPLC and GC) with chiral stationary phases NMR spectroscopy of derivatives that are diastereomeric H S OH R1 R2 F3C OMe Ph or H R OH R2 R1 eclipsed eclipsed H S R2 COCl (Mosher’s reagent) Methods: (R/S) racemate + (R)-MTPA-Cl OH, NH2, SH 등 R2 O R1 O O (R)-MTPA-Cl methoxy trifluoromethyl phenylacetyl chloride H R R1 OMe O or OMe F3C F3C S: R1 -> upfield R: R2 -> upfield due to anisotropic effect of phenyl ring 50 : 50 (R-R-MTPA : S-R-MTPA) R S ppm R, S peak 결정 sample + (R)-MTPA-Cl Derivatives R 90 S 10 ee 80% a-H NH D L D D L L D OMe D D O F3C S Ph OMe N H O D,L CO2t-Bu OTBS > 98%ee a-H NH D L O F3C S Ph OMe N H O D,L CO2Bn Me > 98%ee Me D D L L D D OMe D Optical activity and chirality Optical activity: the ability of a sample to rotate a plane of polarized light. A rotation to the right: + or dextrorotatory (d) A rotation to the left: - or levorotatory (l) Optical activity establishes that a sample is chiral, but a lack of optical activity does not prove a lack of chirality. Optical activity (a) Specific optical activity [a] [a]D25 -> sodium D line (589 nm emission line of sodium arc lamp) Optical purity (%) = [a] mixture of enantiomer [a] pure enantiomer x 100 6.2 Symmetry and stereochemistry 6.2.1 Basic symmetry operations Proper rotation (Cn) -> a rotation around an axis by (360/n)o that has the net effect of leaving the position of the object unchanged. C2; 180 rotation, C3; 120 rotation Improper symmetry (Sn) -> rotation and reflection; involves a rotation of (360/n)o, combined with a reflection across a mirror plane that is perpendicular to the rotation axis. S1; just a mirror reflection (s) S2; equivalent to a center of inversion (i) 90o 60o 180o 6.2.2 Chirality and symmetry A necessary and sufficient criterion for chirality is an absence of Sn axes; the existence of any Sn axis renders an object achiral. C2 Asymmetric is defined as the complete absence of symmetry. However, many chiral molecules have one or more proper rotation axes-just no improper axes are present. These compounds can be referred to as dissymmetric, essential a synonym for chiral. Thus, while all asymmetric molecules are chiral, not all chiral molecules are asymmetric. 6.3 Topicity relationship Topicity: derived from the same roots as topography and topology, relating to the spatial position of an object. 6.3.1 Homotopic, enantiotopic, and diastereotopic Homotopic: is defined as interconvertable by a Cn axis of the molecule. homotopic hydrogens homotopic hydrogens H HO H H chiral influence cannot distinguish these methyl groups H OH achiral molecules C2 Heterotopic: the same groups or atoms in inequivalent constitutional or stereochemical environment. - Enantiotopic: interconverted by an Sn axis of the molecule (n = 1 in this case). enantiotopic groups, when exposed to a chiral influence (chiral shift reagent를 사용할 시), become distinguishable, as if they were diastereotopic. - diastereotopic: the same connectivity, but there is no symmetry operation that interconverts them in any conformation. 이미 stereogenic center를 갖고 있음 the environments of diastereotopic groups are topologically nonequivalent. -> they can be distinguished by physical probes, especially NMR spectroscopy (AB quartet) diastereotopic HR HS CO2- NH3+ phenylalanine meso: achiral Me N Me H enantiotopic H Ph 2H chiral Me N H Me H Ph H 1 H2 AB quartet diastereotopic 6.3.2 Topicity descriptors – Pro-R/Pro-S and Re/Si 1 O Si face Re face pro-S pro-R pro-R pro-S pro-S pro-R R1 2 R2 3 Enzymatic reactions pro-S H H3C pro-S pro-R H liver alcohol dehydrogenase H OH O -pro-R H3C ethanol acetaldehyde H H3C D or T OH T ro D H3C H OH alcohol dehydrogenase D or T H H3C pro-R H H SCoA R H H O pro-R O H3C O H acyl-CoA dehydrogenase - pro-R aand bH SCoA R H O 6.4 Reaction stereochemistry: stereoselectivity and stereospecificity 6.4.1 Simple guidelines for reaction stereochemistry 1. Homotopic groups cannot be differentiated by chiral reagents. 2. Enantiotopic groups can be differentiated by chiral reagents. 3. Diastereotopic groups are differentiated by achiral and chiral reagents. 6.4.2 Stereospecific and stereoselective reactions Stereospecific reaction: one stereoisomer of the reactant gives one stereoisomer of the product, while a different stereoisomer of the reactant gives a different stereoisomer of product. Stereospecific reaction is a special, more restrictive case of a stereoselective reaction. Stereoselective reaction: one in which a single reactant can give two or more stereoisomeric products, and one or more of these products is preferred over the others-even if the preference is very small. Regioselective reaction; when more than one site reacts, this reaction is one where an excess of one of the possible products results. stereospecific stereoselective stereoselective stereospecific inversion Br Me Ph H Ph H Syn addition Ph anti elimination Ph -Ot-Bu Br Me Ph Ph H Ph H -Ot-Bu anti elimination anti elimination Ph Nu: Rm Rs Rl O Rm Rs R Rm Nu: O R Rl preferred Rl R Rm O Rs Regioselective reaction Markovnikov addition Rs Rl OH R Nu major Rm Rs Rl OH Nu R minor 6.5 Symmetry and time scale Time scale is important. three Hs -> equivalent due to fast rotation of C-C bond three Hs -> equivalent but at low temperature (-90 oC), inequivalent due to slow rotation (very clowded system) achiral <- fast inversion chiral <- slow inversion 6.8 Stereochemical issues in chemical biology 6.8.1 The linkages of proteins, nucleic acids, and polysaccharides Proteins planar ~19 kcal/mol rotation barrier ~4 kcal/mol preference Much smaller cis-trans preference 20 natural amino acids (L form) achiral HO2C NH2 HO2C H HO2C HO2C NH2 OH OH NH2 NH2 HO2C HO2C NH2 HO2C NH2 Tyr, Y HO2C NH2 NH2 CO2H N H SH HO2C HO2C H Pro, P Cys, C N H OH Phe, F NH2 Ile, I Leu, L SCH3 Met, M Thr, T Ser, S HO2C HO2C Val, V Ala, A NH2 HO2C NH2 CH3 Gly, G HO2C HO2C NH2 NH2 NH2 H N N Trp, W NH2 Lys, K His, H NH2 HO2C NH NH2 HO2C NH2 HO2C NH2 NH2 CONH2 CO2H CONH2 CO2H H2N Arg, R HO2C NH Asp, D Glu, E Asn, N Gln, Q 3’ Nucleic acids H N H 5’ N N H3C O P O- 3’ O O H H N A=T G≣C O N 5’ N N N N O O P O H N H N H O - N O O O N H N N O Phosphodiester bonds O O O OO P O- HO HO 5' 1' O 4' 3' 5' OH 2' HO (OH) 2'-deoxyribose ribose Base Base O 4' 3' 1' 2' HO (OH) O 5' Base O 4' 3' 1' 2' HO (OH) 2'-deoxyribose ribose 2'-deoxyribose ribose Nucleosides Nucleotides Nucleic acid (RNA or DNA) Bases b-glycosidic linkage Phosphodiester linkages Carbohydrates Functional Glycomics Functional Glycomics Carbohydrate-protein Interactions Toxin • Structural and functional studies of whole carbohydrates • Studies of carbohydrate-protein interactions Bacteria Antibody Hormone • Understanding biological processes Virus • Development of therapeutic agents Protein (Tumor) Cell Biological processes - Inhibitors for carbohydrate biosynthesis - Inhibitors for carbohydrate-binding proteins - Carbohydrate-based vaccines - Finding disease-related markers • Fertilization, development, differentiation, growth, aging Diseases • Tumor metastasis • Inflammation • Bacterial and viral infection Glycoconjugates Carbohydrates exist in the forms of glycoconjugates such as glycolipids and glycoproteins Glycoproteins: glycans attached to proteins Glycolipids: glycans attached to lipids Cell surface carbohydrares anomeric center - Homopolysaccharides - heteropolysaccharides - Complex carbohydrates in which many simple sugars are linked. - Cellulose and starch are the two most widely occurring polysaccharides in plants. Polysaccharides Cellulose (-Glcb1,4Glc-)n 4 - Consists of thousands of D-glucopyranosyl-1,4-b-glucopyranosides. - form a large aggregate structures held together by hydrogen bonds. - is the main component of wood and plant fiber. - is not digested in human body but is digested in herbivore (초식동물). Starch (녹말 綠末 또는 전분 澱粉) - is digested into glucose. - can be separated into two fractions 1) amylose, insoluble in cold water, 20% by weight of starch, 1,4-a-glycoside polymer 2) amylopectin, soluble in cold water, 80% by weight of starch contains 1,6-a-glycoside branches approximately every 25 glucose units in addition to 1,4-a-links. amylose (-Glca1,4Glc-)n Amylopectin In human, glycosidases highly selectively hydrolyze 1,4-a-linkage in starch but not 1,4-b linkage in cellulose. Monosaccharides in mammalian glycoconjugates OH OH O HO HO OH OH O HO HO OH O OH HO OH NHAc OH OH D-N-acetyl glucosamine (GlcNAc) D-Glucose (Glc) D-Galactose (Gal) OH OH OH HO O HO HO HO OH HO2C O O HO HO OH OH OH NHAc D-N-acetyl galactosamine (GalNAc) OH OH CO2H O AcHN D-Mannose (Man) O HO HO OH D-Glucuronic acid (GlcA) H3C OH N-Acetylneuraminic acid (NeuAc) D-Xylose (Xyl) HO HO HO OH HO O O HO O OH OH OH OH OH OH OH OH O L-Fucose (Fuc) OH O OH O O HO O OH O O OH Glycosidic Bonds Blood type HO OH HO O HO OH HO OH O AcHN O O HO O H3C O OH OH OH Blood group A OH O O OR NHAc HO HO HO OH OH O HO O O OH O HO O HO O H3C OH OH O OR NHAc H3C O HO O O OH OH OH OH OH Blood group B Blood group O O OR NHAc Pathogen Infection by Carbohydrate-protein Interactions pathogens DNA or RNA √ Human influenza viruses (haemagglutinin protein) preferentially adhere to NeuNAca2,6Gal residues on epithelial cells (상피세포) of the lungs and upper respiratory tract. √ Avian influenza viruses (AI, 조류독감 바이러스) are specific for NeuNAca2,3Gal residues on intestinal epithelial cells. √ Some of Helicobacter pyroli expresses Leb-binding adhesin (BabA) and sialyl Lex-binding adhesin (SabA) and thus adhere to the human gastric mucosa expressing these glycans. √ Cholera toxin adheres to ganglioside GM1 in host cells. Tamiflu: a drug for influenza OH OH O HO HO OH OH OH O HO HO O OH NHAc OH D-N-acetyl glucosamine (GlcNAc) D-Glucose (Glc) OH OH OH HO Transition state forOaction HO OH of influenza neuraminidase HO HO O OH NHAc D-Mannose (Man) CO2H O AcHN O OH OH HO HO OH OH D-Galactose D-Glucose (Gal) (Glc) D-N-acetyl glucosa OH HOTamiflu (독감 치료제) 2COH O HO O OH HO HO OH OH OH HO HO HO NHAc D-N-acetyl galactosamine (GalNAc) OH OH HO HO HO OH OH HO OH O-sugar HO OH OH N-Acetylneuraminic acid (NeuAc) N-acetyl neuraminic acid O neuraminidase OH OH essential for influenza virus D-Xylose (Xyl) D-Glucuronic acid (GlcA) (GalNAc) D-N-acetyl galactosamine H3C OH OH O AcHN OH OH OH D-Mannos CO2H OH OH O OH HO HO OH L-Fucose (Fuc) acid (NeuAc) N-Acetylneuraminic D-Xylos Stereochemical Terminology Absolute configuration. A designation of the position or order of arrangement of the ligands of a stereogenic unit in reference to an agreed upon stereochemical standard. Achiral Not chiral. A necessary and sufficient criterion for achirality in a rigid molecule is the presence of any improper symmetry element (Sn including σ and ί). A chirotopic. The opposite of chirotopic. See “ chirotopic” below. Anomers. Diastereomers of glycosides or related cyclic forms of sugars that are specifically epimers at the anomeric carbon (C1 of an aldose, or C2, C3, etc., of a ketose). Anti. Modern usage is to describe relative configuration of two stereogenic centers along a chain. The chain is drawn in zigazg form, and if two substituent s are on opposite sides of the plane of the paper, they are designated anti. See also “syn”, “antiperiplanar”, and “ anticlinal”. Anticlinal. A term describing a conformation about a single bond. In A-B-C-D, A and D are anticlinal if the torsion angle between them is between 90 and 150 or -90 and -150. See Figure 2.7. Antiperiplanar. A term describing a conformation about a single bond. In A-B-C-D, A and D are antiperiplanar if the torsion angle between them is between +150° to -150° . See Figure 2.7. Apical, axial, basal, and equatorial. Terms associated with the bonds and positions of ligands in trigonal bipyramidal structures. Asymmetric. Lacking all symmetry elements (pointing group C1). All asymmetric molecules are chiral. Asymmetric carbon atom. Traditional term used to describe a carbon with four different ligands attached. Not recommended in modern usage. Atactic. A term describing the relative configuration along a polymer backbone. In an atactic polymer, the stereochemistry is random-no particular pattern or bias is seen. Atropisomers. Stereoisomers ( can be either enantiomers or diastereomers) that can be interconverted by rotation about single bonds and for which the barrier to rotation is large enough that the stereoisomers can be separated and do not interconvert readily at room temperature. Chiral. Existing in two forms that are related as non-congruent mirror images. A necessary and sufficient criterion for chirality in a rigid molecule is the absence of any improper symmetry elements. Chiral center. Older term for a tetracoordinate carbon or similar atom with four different substituents. More modern, and preferable, terminology is “stereogenic center” (or “stereocenter”) Chirotopic. The term used to denote that an atom, point, group, face, or line resides in a chiral environment. Cis. Describing the stereochemical relationship between two ligands that are on the same side of a double bond or a ring system. For alkenes only, Z is preferred. Configuration. The relative position or order of the arrangement of atoms in space that characterizes a particular stereoisomer. Conformers or conformational isomers. Stereoisomers that are interconverted by rapid rotation about a single bond. Constitutionally heterotopic. The same groups or atoms with different connectivities. D and L. An older system for identifying enantiomers, relating all stereocenters to the sense of chirality of D- or L-glyceraldehyde. See discussion in the text. Generally not used anymore, except for biological structures such as amino acids and sugars. Diastereomers. Stereoisomers that are not enantiomers. Diastereomeric excess (de). In a reaction that produces two diastereomeric products in amounts A and B, de = 100% (|A – B|) / (A + B). Diastereotopic. The relationship between two regions of a molecule that have the same connectivity but are bit related by any kind of symmetry operation. Dissymmetric. Lacking improper symmetry operations. A synonym for “chiral”, but not the same as “asymmetric” . Eclipsed. A term describing a conformation about a single bond. In A-B-C-D, A and D are eclipsed if the torsion angle between them is approximately 0°. Enantiomers. Molecules that are related as non-congruent mirror images. Enantiomeric excess (ee). In a reaction that produces two enantiomeric products in amounts A and A´ , ee = 100% (|A – A´|) / (A + A´). Enantiotopic. The relationship between two regions of a molecule that are realated only by an improper symmetry operation, typically a mirror plane. Endo. In a bicyclic system, a substituent that is on a bridge is endo if it points toward the larger of the two remaining bridges. See also “exo” . Epimerization. The interconversion of epimers. Epimers. Diastereomers that have the opposite configuration at only one of two or more stereogenic centers. Erythro and threo. Descriptors used to distinguish between diastereomers of an acyclic structure having two stereogenic centers. When placed in a Fischer projection using the convention proper for carbohydrates, erythro has the higher priority groups on the same side of the Fischer projection, and threo has them on opposite sides. Exo. In a bicyclic system, a substituent that is on a bridge is exo if it points toward the smaller of the two remaining bridges. See also “endo” . E, Z. stereodescriptors for alkenes (see discussion in the text). Gauche. A term describing a conformation about a single bond, In A-B-C-D, A and D are gauche if the torsion angle between them is approximately 60°(or -60°). See section 2.3.1. Geminal. Attached to the same atoms. The two chlorines of 1,1-dichloro-2,2-difluoroethane are geminal. See also “vicinal”. Helicity. The sense of chirality of a helical or screw shaped entity ; right (P) or left (M). Heterochiral. Having an oppsite sense of chirality. For example, D-alanine and L-leucine are heterochiral. See also “homochiral”. Heterotopic. The same groups or atoms in inequivalent constitutional or stereochemical environments. Homochiral. Having the same sense of chirality. For example, the 20 natural amino acids are homochiral – they have the same arrangement of amino, carboxylate, and side chain groups. Has also been used as a synonym for “enantiomerically pure”, but this is not recommended, because homochiral already as a well-defined term before this alternative usage became fashionable. Homotopic. The relationship between two regions of a molecule that are related by a proper symmetry operation. Isotactic. A term describing the relative configuration along a polymer backbone. In an isotactic polymer, all stereogenic centers of the polymer backbone have the same sense of chirality. Meso. A term describing a achiral member of a collection of diastereomers that also includes at least one chiral member. Opitcally active. Rotating plane polarized light. Formerly used as a synonym for “chiral”, but this is not reconmmended. Prochiral. A group is prochiral if it contains enantiotopic or diastereotopic ligands or faces, such that replacement of one ligand or addition to one face produces a stereocenter. See section 6.3.2. R, S. The designations for absolute stereochemistry (see earlier discussion in the text). Racemic mixture or racemate. Comprised of a 50:50 mixture of enantiomers. Relative configuration. This refers to the configuration of any stereogenic center with respect to another stereogenic center. If one center in a molecule is known as R, then other centers can be compared to it using the descriptors R* or S*, indicating the same or opposite stereochemistry, respectively. Resolution. The separation of a racemic mixture into its individual component enantiomers. Scalemic. A synonym for “non-racemic” or “enantiomerically enriched”. It has not found general acceptance, but is used occasionally. S-cis and s-trans. Descriptors for the conformation about a single bond, such as the C2-C3 bond in 1,3-buadiene, or the C-N bond of an amide. If the substituents are synperiplanar, they are termed s-cis (“s” for “single”); if they are antiperiplanar, they are termed s-trans. Stereocenter. See “stereogenic center”. Stereogenic center. An atom at which interchange of any two ligands produces a new stereoiosmer. A synonym for “stereocenter”. Stereogenic unit. An atom or grouping of atoms at which interchange of any two ligands produces a new stereoisomer. Stereoisomers. Molecules that have the same connectivity, but a different arrangement of atoms in space. Stereoselective. A term describing the stereochemical consequences of certain types of reactions. A stereoselective reaction is one for which reactant A can give two or more stereoisomeric products, B and B’, and one or more product is preferred. There can be degrees of stereoselectivity. All stereospecific reactions are stereoselective, but the converse is not true. Stereospecific. A term describing the stereochemical consequences of certain types of reactions. A stereospecific reaction is one for which reactant A gives product B, and stereoisomeric reactant A’ gives stereoisomeric product B’. There can be degrees of stereospecificity. Stereosprcific does not means 100% stereoselective. Syn. Modern usage is to describe the relative configuration of two stereogenic centers along a chain. The chain is drawn in zigzag form, and if two substituents are on the same side of the plane of the paper, they are syn. See also “anti”, “synperiplanar”, and “synclinal”. Synclinal. A term describing a configuration about a single bond. In A-B-C-D, A and D are synclinal if the torsion angle between 30° and 90° (or -30° and -90°). See Figure 2.7. Syndiotactic. A term describing the relative configuration along a polymer backbone. In a syndiotactic polymer, the relative configuration of backbone stereogenic centers alternate along the chain. Synperiplanar. A term describing a conformation about a single bond. In A-B-C-D, A and D aresynperiplanar if the torsion angle between them is between + 30° and –30°. See Figure 2.7. Tacticity. A generic term describing the stereochemistry along a polymer backbone. See “atactic”, “isotactic”, and “syndiotactic”. Trans. A term describing the stereochemical relationship between two ligands that are on opposite sides of a double or a ring system. For alkenes only. E is preferred. Vicinal. Attatched to adjacent atoms. In 1,1-dichloro-2,2-difluoroethane, the relationship of either chlorine to either fluorine is vicinal. See also “geminal”.