* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Slide 1
Survey
Document related concepts
Transcript
TUMS Azin Nowrouzi, PhD 1 Functions of H+ • [H+] and therefore pH is important for many biochemical processes: – Transport of oxygen in blood – Enzymatic reactions – Generation of metabolic energy during respiration and photosynthesis – Metabolism and its regulation. – Biomolecules amino acids , proteins, sugars, lipids, nucleic acids, vitamins, and …. are sensitive to pH variations. – Measurement of pH of blood and urine are commonly used in medical diagnoses. 2 Acids and Bases • Acid: a substance that can donate H+. • Base: a substance that can accept H+. • Water (H2O): Both an acid and a base. One H2O molecule, acting as an acid, may donate a proton to another H2O molecule, acting as a base. 3 Self-ionization (autoprotolysis) of water Keq = 1.8 x 10-16 [H2O]=55.5 M because (1000g/L)/(18.015 g/mol) (55.6)Keq = [H+][OH-] (55.6)(1.8 x 10-16) = [H+][OH-] = Kw Kw = 1.0 x 10-14 = [H+][OH-]= [H+]2 [H+] = [OH-] = = 1.0 x 10-7 M pH + pOH = 14 If we put an acid into a solution to decrease the pH the pOH must increase to keep their sum equal to pKw. 4 Definition of buffer • Buffer action = the ability to oppose changes in pH when small amounts of acid or base are added to the solution. • Buffers are mixtures of weak acids and their conjugate bases. 5 Buffers • The mathematical basis of buffer action is given by the Henderson-Hasselbalch equation: 6 • Liquid inside cells (Intracellular fluid) - 40 % • Extracellular fluids - 15 % • Liquid making the plasma of blood - 5 % 7 Fluid spaces in the human body The ICF and ECF are called fluid compartments because they behave as distinct entities. 8 Intracellular fluid 35%-40% BW Total body water Blood plasma (intravascular fluid) 50%-60% BW 4%-5% BW Extracellular fluid 15%-20% BW Interstitial fluid BW = Body Weight Lymph (extravascular) 11%-15%BW Transcellular fluid: Cerebrospinal fluid Intraocular fluid Synovial fluid Pericardial fluid Pleural fluid Peritoneal fluid 9 10 11 Sources of acid and base in the body 12 Types and sources of Acids in the Body • • • • Acid-forming foods are flesh foods, grains, dairy products, a majority of nuts and seeds, beans and peas, simple sugars, fats, and proteins. • Fats as a general class are acidforming because their metabolism produces acetic acid. Simple carbohydrates like white sugar are acid- • forming because they enter the system too quickly and burn too fast. Alkaline-forming foods are vegetables and fruits. Exceptions include asparagus, cranberries, plums, and prunes, which are all slightly acidic. While fruits such as lemons, oranges, grapefruits, tomatoes and pineapples, are acid when you eat them, but, by the process of enzymatic digestion; they are turned into alkaline substances. Volatile (CO2, Carbonic acid) – Can leave the solution and enter the atmosphere. – End product of oxidation of glucose and triglyceride during aerobic metabolism. Fixed (non-carbonic) • Acids that do not leave solution. – Inorganic acids (Sulfuric Acid, Phosphoric Acid) • Catabolism of amino acids, nucleic acids, and phospholipids. – Organic (Lactic acid, Ketone Bodies) • By products of aerobic metabolism, anaerobic metabolism, starvation and diabetes. 13 Production of Dietary Source Quantity Produced (mEq/day) Acid Alkali Acid Alkali Carbohydrate 0 0 0 0 Fat 0 0 0 0 Amino acids Sulfur-containing (Cys, Met) Cationic (Lys, Arg, His) H2SO4 70 HCl 135 100 HCO3- Anionic (Asp, Glu) Organic anions (Acetate, Citrate, Gluconate, Lactate, Malate) Phosphate Net acid production 60 HCO3H2PO4- 30 75 14 • The role of intracellular and extracellular buffers, respiratory, and renal mechanisms in maintaining normal blood pH. 15 Different Lines of Defense against Acid –Base disorders • First Line of defense: Buffer System (buffering) The body has a HUGE buffering capacity, and This system is essentially IMMEDIATE in effect. • Second Line of defense: Respiratory System Alteration; in arterial pCO2 (compensation) minutes to hours • Third Line of defense: Renal System Alteration in HCO3- excretion (correction) several days 16 Acid-Base Balance I. Buffering systems of the body 1. The bicarbonate/carbonic acid system • 2. 3. The phosphate/phosphoric acid buffer system Protein buffering • • • 4. II. III. The role of the erythrocyte in the bicarbonate system The hemoglobin (Hb) system Cellular buffering Bone buffering Ammonia buffering system The lungs The kidneys 17 Important buffers at physiologic [H+] 18 19 1. Bicarbonate/carbonic acid buffer system CO2 as an acid 1. The greatest source of H+. 2. End product of oxidation of glucose and triglyceride during aerobic metabolism. 3. Most important weak acid in body fluids. [H+] x [HCO3-] = 24 x PCO2 20 Importance of bicarbonate buffer system • Most important physiological buffer because: It is an open system: the acid component of the system is volatile (CO2), and can be excreted rapidly by pulmonary respiration. It transports H+ to the kidney and transfer it to other buffers like phosphates to be excreted in urine. It’s in high concentration in plasma (HCO3- = 24 meq/l). It’s the bicarbonate buffer system that is regulated. H2CO3 = PCO2 X 0.03 21 The role of the erythrocyte in the bicarbonate system It is an open system: the acid component of the system is volatile (CO2), and can be excreted rapidly by pulmonary respiration. Carbonic anhydrase (CA): Located in • The cytosol of cells (renal cells and RBC’s). • Along the brush border of the proximal tubule. Membrane-bound enzyme with the active site facing the nephron lumen. • Not found in plasma. 22 Hydrogen ion (H+) secretion and bicarbonate ion (HCO3-) reabsorption in a renal tubular cell It transports H+ to the kidney and transfer it to other buffers like phosphates to be excreted in urine. • Carbon dioxide (CO2) diffuses from the blood or urine filtrate into the tubular cell, where it combines with water in a carbonic anhydrase-catalyzed reaction that yields carbonic acid (H2CO3). • The H2CO3 dissociates to form H+ and HCO3-. • The H+ is secreted into the tubular fluid in exchange for Na+. • The Na+ and HCO3- enter the extracellular fluid. " 23 24 Acid-base homeostasis 25 2. Phosphate/phosphoric acid buffer sytem • Phosphate concentration in the blood is so low that it is quantitatively unimportant. • Phosphates are important buffers intracellularly and in urine where their concentration is higher. • Phosphoric acid is triprotic acid that exists in four forms. Phosphoric acid has a pKa value for each of the three dissociations with three pK values of 2.1, 6.8, and 12.7: H3PO4 ↔ H2PO4- ↔ HPO4-2 ↔ PO4-3 • When a solution of phosphoric acid (H3PO4) is titrated completely, there will exist three buffering regions. • Physiologically, we only need to deal with the pH range between pH 4.5 (acidic urine) and 8.0 (alkaline urine). • For this reason, only two forms of phosphate (H2PO4- and HPO4-2) and one pK value (6.8). • H2PO4- is the acid form, and HPO4-2 is the salt form. 26 The renal phosphate buffer system • The monohydrogen phosphate ion (HPO42-) enters the renal tubular fluid in the glomerulus. A H+ combines with the HPO42- to form H2PO4and is then excreted into the urine in combination with Na+. The HCO3moves into the extracellular fluid along with the Na+ that was exchanged during secretion of the H+. 27 3. Hemoglobin as a buffer system • • • • • Located in RBC. Very important buffer of plasma pH. pK of hemoglobin is dependent on oxygenation. The drop in pH due to CO2 addition to the blood is less than predicted (from 7.40 to 7.37 and not 7.32) because of the action of hemoglobin. This is because in the RBC, hemoglobin acts as a buffer for H+. • • • Hemoglobin protein can reversibly bind either H+ (to the protein) or O2 (to the Fe of the heme group). But that when one of these substances is bound, the other is released (as explained by the Bohr effect). During exercise, hemoglobin helps to control the pH of the blood by binding some of the excess protons that are generated in the muscles. At the same time, molecular oxygen is released for use by the muscles. 28 • Mechanisms of carbon dioxide transport. CA, Carbonic Anhydrase. 29 H+ attachment to hemoglobin T state R state 30 Cellular (protein) buffer system • Proteins can act as buffers. • Because each protein molecule is both a weak acid and a weak base. • The weakly acidic carboxylic acids counteract rising pHs while the weakly basic amino groups can counteract falling pHs. • Plasma proteins and hemoglobin (blood's oxygen-carrying pigment, a protein) enhance the blood's buffering capacity. • Human albumin contains 585 amino acid residues, which includes 61 glutamic acid, 24 arginine, 26 aspartic acid, and 16 histidine amino acids. Dependent on the pH, albumin can act as a conjugate base (accepts protons) or a conjugate acid (donates protons). (1) (2) (3) (4) (5) If pH = 3.0, its net charge is +60 (it has 60 protons to donate). If pH = 4.9, it is a zwitterion, electrically neutral. If pH = 7.5, its net charge is -10 (it can accept 10 protons) If pH = 8.6, its net charge is -20 If pH = 11.0, its net charge is -60 • Proteins also help with buffering within cells. 31 The ammonia buffer systems 32 4. Bone buffer systems • When protons are added to the body – The immediately available buffers are those of the blood and extracellular fluid – But, after some time, intracellular buffers and bone play their role. • The uptake of protons by bone can occur in exchange for surface Na+ and K+ and by dissolution of bone mineral (bone carbonate). – Bone contains 80% of total CO2 (CO32-, HCO3- and CO2) in the body. – Two-thirds in the form of CO32- complexed with Ca2+, Na+, and other cations in bone crystals. – One-third in the form of HCO3- in the hydroxyapatite crystals. • Chronic metabolic acidosis is dangerous because of progressive bone lysis and in children, growth retardation. 33 34 Acid-basic disorder evaluation Constant [HCO3- ] [CO2] pH= pK +log 24 = 6.1+log 1.2 [HCO3- ] [CO2] Changes lead to acidosis or Alkalosis Metabolic Respiratory =6.1+1.3= 7.4 > 1.3 Alkalosis kidneys Lungs or Normal < 1.3 Acidosis [HCO3- ] [CO2] 35 Respiratory Acidosis: [CO2] ↑ • A process that drives blood pH below 7.35. • Occurs when CO2 is not eliminated from the body at normal rate. • Usual cause is decreased respiration or hypoventilation. • When respiration is restricted, the concentration of dissolved carbon dioxide in the blood increases, making the blood too acidic. Such a condition can be produced by asthma, pneumonia, emphysema, or inhaling smoke. • An elevated plasma PCO2 is called hypercapnia. • Respiratory system is unable to eliminate all of the CO2 generated by peripheral tissues. 36 Respiratory Alkalosis: [CO2] ↓ • A process that drives blood pH above 7.45 • Results from excessive breathing or hyperventilation. • Hyperventilation causes too much dissolved CO2 to be removed from the blood, which decreases the carbonic acid concentration, which raises the blood pH. • Often, the body of a hyperventilating person will react by fainting, which slows the breathing. • Problems with this are relatively uncommon. • When plasma PCO2 is below normal levels, the condition is called hypocapnia. • Temporary hypocapnia can be produced by hyperventilation. • Condition seldom persists long enough to cause a clinical emergency. 37 Metabolic Acidosis: [HCO3- ]↓ • Decrease in blood pH that results when excessive amounts of acidic substances (metabolic acids such as lactic acid or ketone bodies) are released into the blood. • This can happen through – prolonged physical exertion, – diabetes – restricted food intake (starvation). • Occurs when bicarbonate ion concentrations decrease. The acids are buffered by HCO3- lowering its levels. • The normal body response to this condition is to increase breathing to reduce the amount of dissolved carbon dioxide in the blood. This is why we breathe more heavily after climbing several flights of stairs. • Can be caused by the inability to excrete hydrogen ions from the kidneys and any condition causing severe kidney damage. 38 Metabolic Alkalosis: [HCO3- ]↑ • Occurs when bicarbonate ion concentrations become elevated. • This can result from the ingestion of alkaline materials, and through overuse of diuretics. • Bicarbonate ions (from alkaline foods) interact with hydrogen ions in solution to form carbonic acid and the reduction of H+ ions causes symptoms of alkalosis. • Again, the body usually responds to this condition by slowing breathing, possibly through fainting. • Cases of severe metabolic alkalosis are relatively rare. 39 The four primary acid-base disturbances 40 Normal and compensated states of pH and acid-base balance represented as a balance scale A. B. C. D. E. • When the ratio of bicarbonate (HCO3-) to carbonic acid (H2CO3, arterial PCO2 .03) = 20:1, the pH = 7.4. Metabolic acidosis with an HCO3-;H2CO3 ratio of 10;1 and a pH of 7.1. Respiratory compensation lowers the H2CO3 to 0.6 mEq/L and returns the HCO3-:H2CO3 ratio to 20;1 and the pH to 7.4. Respiratory alkalosis with an HCO3-;H2CO3 ratio of 40;1 and a pH of 7.7. Renal compensation eliminates HCO3-, reducing serum levels to 12 mEq/L, returning the HCO3-;H2CO3 ratio to 20;1 and the pH to 7.4. Normally, these compensatory mechanisms are capable of buffering large changes in pH, but may not return the pH completely to normal, as illustrated here. 41 42 Determining acid/base disorder Practice using the following gases: pH=7.3 PCO2= 60 HCO3-= 24 "______?________ , ______?________ , _______?______" Step one: Determine whether pH is acidic or basic Step two: Evaluate PCO2 Step three: Evaluate HCO3- or base excess Step 4: Put all the information together and look for compensation. 43 Step One: Determine whether pH is acidic or basic • pH must always be determined first when assessing acid/base balance. – If pH is > 7.40, blood is more basic. If pH is < 7.40, blood is acidic. – If pH is between 7.35 and 7.45, the acid base disturbance is compensated. • Determine which the pH is: an acid or base; then mark an ‘A’ for acid, ‘B’ for base or ‘N’ for normal beside the pH. You have now determined which imbalance, (acidosis or alkalosis) is present. • From the above example: • ph = 7.3 " A“, PCO2=60 ________, HCO3=24 _________ "______?________, ______?_______ , ___Acidosis___" 44 Step Two: Evaluate PCO2 • PCO2 is the respiratory component of acid/base balance. **Remember that PCO2 is an acid. • A PCO2 > 40 favors resp. acidosis • A PCO2 < 40 favors resp. alkalosis • Normal range for PCO2 is 35 - 45. • Mark and ‘A’ , ‘B’ or ‘N’ beside the PCO2 value. • In the example, PCO2 = 60 so we put an "A" beside the PCO2. "______?______, Respiratory Acidosis" 45 Step three: Evaluate HCO3- or base excess (BE) • HCO3- < 22 favors metabolic acidosis HCO3- > 26 favors metabolic alkalosis • A positive base excess (BE) favors metabolic alkalosis; a negative base excess favors acidosis. • Mark a ‘A’ , ‘B’ or ‘N’ beside the value. In our example HCO3 = 24 so we put an "N" beside the value. 46 Step 4: Put all the information together and look for compensation • Circle all the letters. Match the ones that are the same. • In our example, the pH is acidic, the PCO2 is acidic and the HCO3 is normal. Therefore the disorder is a respiratory acidosis because the PCO2 and the pH match. • So, we can fill in 2 blanks: "___________ respiratory acidosis." 47 Determine the compensation 1. Uncompensated-The gases are uncompensated if the pH is outside the normal range ie <7.35 or > 7.45 – In the above example the gases are uncompensated because the pH is 7.3 2. Partial compensation: pH is within the normal range of 7.35 - 7.45 but is not 7.40 3. Complete compensation: pH is 7.4 “Uncompensated Respiratory Acidosis” 48 Hypoxemia • Normal PO2 is 80 to 100 mmHg (for a healthy person, less than 60 years old, breathing room air). • Hypoxemia- Definition: deficient oxygenation of the blood. **Measured by PO2 • PO2 < 80 : mild hypoxemia PO2 < 60 : moderate hypoxemia PO2 < 40 : severe hypoxemia • age: For every year above 60, a patients’ normal PO2 drops by 1 mm Hg 49 Examples: Arterial blood gas interpretation PO2=70 pH= 7.33 A B or N PCO2= 48.3 A B or N HCO3=25.8 A B or N PO2=88 pH= 7.45 A B or N PCO2= 43 A B or N HCO3=30.1 A B or N 50 51 52 53 54 55 56 57 Example: Evaluation of acid-base status • • • • • • • • In a patient with chronic respiratory acidosis: Arterial PCO2 = 60mmHg Plasma [HCO3-] = 32 mmoles/L Arterial pH = 7.35 BE = [HCO3-]observed – [HCO3-]predicted Observed [HCO3-] is calculated from pH and pCO2 Predicted [HCO3-] = 24 + (2.733 + 0.52Hb)(7.4-pH) Predicted [HCO3-] = 24 + 10(7.4-pH) if Hb is normal and equal to 15g/100ml of blood. 58 59 60 Davenport diagram 61 62 Modified Henderson Equation This is the equation used to derive [HCO3-] from pH and PCO2. [H+] x [HCO3-] = 24 x PCO2 63 The acid-base nomogram 64 Protonation and deprotonation of histidine residues in Hb and other proteins. 65






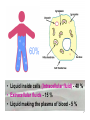





























































![ACID-BASE BALANCE Acid-base balance means regulation of [H + ]](http://s1.studyres.com/store/data/000604092_1-2059869358395bda26ef8b10d08c9fb9-150x150.png)


