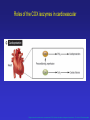
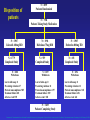
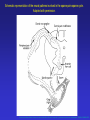
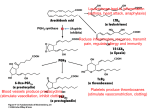
* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Physiological Role for COX-2
Discovery and development of proton pump inhibitors wikipedia , lookup
Psychopharmacology wikipedia , lookup
Metalloprotease inhibitor wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Adherence (medicine) wikipedia , lookup
Discovery and development of ACE inhibitors wikipedia , lookup
Pharmacogenomics wikipedia , lookup
Theralizumab wikipedia , lookup
Discovery and development of cyclooxygenase 2 inhibitors wikipedia , lookup
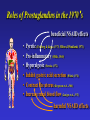
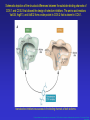
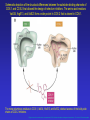
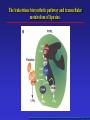
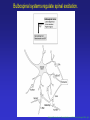
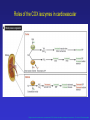
Dúvidas [email protected] Arquivo Anti inflamatório Site www.gilbertodenucci.com Medicamentos • • • • • • Inibidores de Cox não seletivos Inibidores de Cox seletivos para Cox-2 Antidepressivos Anticonvulsivantes Relaxantes musculares Opióides History of inflammation and NSAIDs For 3,500 years inflammation has been treated with preparations originating from plants containing salicylates. • myrtle leaves (Myrtus) • willow bark (Salix) • poplar bark (Populus) • meadowsweet (Spiraea) • wintergreen oil (Gaultheria) Aspirin On 6th March 1899, ‘ Aspirin ’ was registered as a trademark with the Imperial Patent Office in Berlin. WORLDWIDE Production - 36,000 tons / year Use - 70 tablets / person / year acetyl ‘A’ Spiraea ‘SPIRIN’ Cyclo-oxygenase Hypothesis - 1970’s membrane phospholipids (-) Phospholipase A2 glucocorticoids arachidonic acid Classical (-) NSAIDs COX Stomach Kidney Endothelium Platelets PGE2/PGI2 PGE2/PGI2 PGI2 TXA2 gastric cytoprotection renal blood flow haemostasis Physiological Effects Inflammatory Sites PGE2 inflammatory mediators Inflammation Roles of Prostaglandins in the 1970’s • • • • • • Pyretic (Feldberg & Gupta, 1973; Milton & Wendlandt, 1973) Pro-inflammatory (Willis, 1969) Hyperalgesic (Ferreira, 1972) Inhibit gastric acid secretion (Whittle, 1976) Contract the uterus (Bergstrom et al .,1968) Increase renal blood flow (Lonigro et al., 1973) Roles of Prostaglandins in the 1970’s beneficial NSAID effects • • • • • • Pyretic (Feldberg & Gupta, 1973; Milton & Wendlandt, 1973) Pro-inflammatory (Willis, 1969) Hyperalgesic (Ferreira, 1972) Inhibit gastric acid secretion (Whittle, 1976) Contract the uterus (Bergstrom et al .,1968) Increase renal blood flow (Lonigro et al., 1973) harmful NSAID effects Increasing COX-2 selectivity* Comparative upper GI toxicity and COX-2 selectivity naproxen 10 9 8 7 6 5 4 3 2 1 0 nabumetone / 6MNA flurbiprofen tolmetin aspirin indomethacin ketoprofen ibuprofen diclofenac sulindac r2 0.71; p < 0.03 etodolac 1 *WHRI blood/A549 assay 2 3 4 5 6 7 8 9 Increasing upper GI toxicity 10 11 Cyclo-oxygenase Hypothesis - 1990’s membrane phospholipids (-) Phospholipase A2 glucocorticoids (-) arachidonic acid COX-1 constitutive (-) Classical NSAIDS COX-2 (-) inducible Stomach Kidney Endothelium Platelets PGE2/PGI2 PGE2/PGI2 PGI2 TXA2 gastric cytoprotection renal blood flow haemostasis Physiological Effects INDUCTION: mitogens endotoxins cytokines (-) selective COX-2 inhibitors Inflammatory Sites PGE2 inflammatory mediators Inflammation Schematic depiction of the structural differences between the substrate-binding channels of COX-1 and COX-2 that allowed the design of selective inhibitors. The amino acid residues Val434, Arg513, and Val532 form a side pocket in COX-2 that is absent in COX-1. Nonselective inhibitors have access to the binding channels of both isoforms. Biological basis for the cardiovascular consequences of COX-2 inhibition: therapeutic challenges and opportunities - The Journal of Clinical Schematic depiction of the structural differences between the substrate-binding channels of COX-1 and COX-2 that allowed the design of selective inhibitors. The amino acid residues Val434, Arg513, and Val523 form a side pocket in COX-2 that is absent in COX-1. The more voluminous residues in COX-1, Ile434, His513, and Ile532, obstruct access of the bulky side chains of COX-2 inhibitors. Biological basis for the cardiovascular consequences of COX-2 inhibition: therapeutic challenges and opportunities - The Journal of Clinical Investigation Cumulative Incidence of the Primary End Point of a Confirmed Upper Gastrointestinal Event among All Randomized Patients. COMPARISON OF UPPER GASTROINTESTINAL TOXICITY OF ROFECOXIB AND NAPROXEN IN PATIENTS WITH RHEUMATOID ARTHRITIS - The New England Journal of Medicine – fig 01 Kaplan-Meier analyses of time to thromboembolic events in patients not taking aspirin from the CLASS trial. A, celecoxib versus the combined nonsteroidal antiflammatory agents (NSAIDs) ibuprofen and diclofenac combined (p 0.973 celecoxib vs NSAIDs). Comparison of Thromboembolic Events in Patients Treated With Celecoxib, a Cyclooxygenase-2 Specific Inhibitor, Versus Ibuprofen or Diclofenac - The American Journal of Cardiology Vol. 89 February 15, 2002 Kaplan-Meier analyses of time to thromboembolic events in patients not taking aspirin from the CLASS trial. B, celecoxib versus the individual NSAIDs (p 0.379 and 0.236, celecoxib vs diclofenac and vs ibuprofen, respectively). Comparison of Thromboembolic Events in Patients Treated With Celecoxib, a Cyclooxygenase-2 Specific Inhibitor, Versus Ibuprofen or Diclofenac - The American Journal of Cardiology Vol. 89 February 15, 2002 Kaplan–Meier Estimates of the Cumulative Incidence of Confirmed Serious Thrombotic Events. Cardiovascular Events Associated with Rofecoxib in a Colorectal Adenoma Chemoprevention Trial - n engl j med 352;11 – march 17,2005 Kaplan–Meier Estimates of the Cumulative Incidence of Investigator- Reported Congestive Heart Failure (CHF), Pulmonary Edema (PE), or Cardiac Failure (CF). Cardiovascular Events Associated with Rofecoxib in a Colorectal Adenoma Chemoprevention Trial - n engl j med 352;11 – march 17,2005 Schematic overview of the cyclooxygenase and lipoxygenase pathways Advances in eicosanoid research, novel therapeutic implications - Biochemical and Biophysical Research Communications 396 (2010) 135–139 The 5-lipoxygenase/leukotriene pathway Advances in eicosanoid research, novel therapeutic implications - Biochemical and Biophysical Research Communications 396 (2010) 135–139 The leukotriene biosynthetic pathway and transcellular metabolism of lipoxins. Advances in eicosanoid research, novel therapeutic implications - Biochemical and Biophysical Research Communications 396 (2010) 135–139 Bulbospinal systems regulate spinal excitation. Analgesic Pharmacology: I. Neurophysiology - Vol 12, No 4, July/August 2004 – fig 6 Enrollment, Randomization, and Outcomes 3260 Patients assessed for eligibility 674 Did not undergo randomization to 25 mg of rofecoxib or placebo 403 Were ineligible 160 Withdrew consent 41 Had adverse clinical events 26 Were given 50 mg of rofecoxib 44 Had other reasons 1287 Assigned to 25 mg of rofecoxib 1287 Received treatment and included in safety analysis 410 Discontinued treatment 202 Had adverse clinical events 22 Had adverse laboratory events 112 Withdrew consent 14 Were lost to follow-up 60 Had other reasons 877 Completed 3 yr of treatment 1299 Assigned to placebo 1299 Received treatment and included in safety analysis 319 Discontinued treatment 145 Had adverse clinical events 9 Had adverse laboratory events 88 Withdrew consent 20 Were lost to follow-up 57 Had other reasons 980 Completed 3 yr of treatment Cardiovascular Events Associated with Rofecoxib in a Colorectal Adenoma Chemoprevention Trial - n engl j med 352;11 – march 17,2005 The pharmaceutical industry spends more than $5.5 billion to promote drugs to doctors each year — more than what all U.S. medical schools spend to educate medical students. Major drug companies employ about 90,000 sales representatives — one for every 4.7 doctors in the United States, according to the American Medical Association. Roles of the COX isozymes in cardiovascular Biological basis for the cardiovascular consequences of COX-2 inhibition: therapeutic challenges and opportunities - The Journal of Clinical Investigation Roles of the COX isozymes in cardiovascular Biological basis for the cardiovascular consequences of COX-2 inhibition: therapeutic challenges and opportunities - The Journal of Clinical Investigation Roles of the COX isozymes in cardiovascular Biological basis for the cardiovascular consequences of COX-2 inhibition: therapeutic challenges and opportunities - The Journal of Clinical Investigation Illustration of the expected interaction of baseline cardiovascular and thrombotic risk with components of drug exposure including dose, duration of action, and duration of treatment with a selective inhibitor of COX-2. Biological basis for the cardiovascular consequences of COX-2 inhibition: therapeutic challenges and opportunities - The Journal of Clinical Investigation Biological basis for the cardiovascular consequences of COX-2 inhibition: therapeutic challenges and opportunities - The Journal of Clinical Investigation The spectrum of selectivity for COX inhibition. Discordant dose-response relationships for inhibition of platelet COX-1 (A) and vascular COX-2 (B). Biological basis for the cardiovascular consequences of COX-2 inhibition: therapeutic challenges and opportunities - The Journal of Clinical Investigation Clinical implications of differences in the dose-response relationships for COX-1 and COX-2 of low-dose aspirin (A), a selective inhibitor of COX-2 (B), and a tNSAID (C). The area between the dose-response curves would correspond to benefit (A) and hazard (B and C) and to the size of these effects. Biological basis for the cardiovascular consequences of COX-2 inhibition: therapeutic challenges and opportunities - The Journal of Clinical Investigation Duration of use of tNSAIDs and individual tNSAIDs among current users (use within a month) and risk of myocardial infarction. Biological basis for the cardiovascular consequences of COX-2 inhibition: therapeutic challenges and opportunities - The Journal of Clinical Investigation Disposition of patients N = 3987 Celecoxib 400mg BID N = 1779 Completed Study N = 2208 Withdrawn Lost to follow-up: 0 Preexisting violation: 27 Protocol noncompliance: 585 Treatment failure:691 Adverse event:905 N = 8059 Patients Randomized N = 7968 Patients Taking Study Medication N = 1996 Diclofenac 75mg BID N = 939 Completed Study N = 1057 Withdrawn Lost to follow-up: 0 Preexisting violation: 11 Protocol noncompliance: 197 Treatment failure: 309 Adverse event: 540 N = 1985 Ibubrofen 800mg TID N = 691 Completed Study N = 1294 Withdrawn Lost to follow-up: 0 Preexisting violation: 12 Protocol noncompliance: 365 Treatment failure: 456 Adverse event: 461 N = 3409 Patients Completing Study Comparison of Thromboembolic Events in Patients Treated With Celecoxib, a Cyclooxygenase-2 Specific Inhibitor, Versus Ibuprofen or Diclofenac - The American Journal of Cardiology Vol. 89 February 15, 2002 Comparison of Thromboembolic Events in Patients Treated With Celecoxib, a Cyclooxygenase-2 Specific Inhibitor, Versus Ibuprofen or Diclofenac - The American Journal of Cardiology Vol. 89 February 15, 2002 Comparison of Thromboembolic Events in Patients Treated With Celecoxib, a Cyclooxygenase-2 Specific Inhibitor, Versus Ibuprofen or Diclofenac - The American Journal of Cardiology Vol. 89 February 15, 2002 Comparison of Thromboembolic Events in Patients Treated With Celecoxib, a Cyclooxygenase-2 Specific Inhibitor, Versus Ibuprofen or Diclofenac - The American Journal of Cardiology Vol. 89 February 15, 2002 Incidence of Hypertension, Edema, and Congestive Heart Failure Comparison of Thromboembolic Events in Patients Treated With Celecoxib, a Cyclooxygenase-2 Specific Inhibitor, Versus Ibuprofen or Diclofenac - The American Journal of Cardiology Vol. 89 February 15, 2002 Incidence of Hypertension, Edema, and Congestive Heart Failure Comparison of Thromboembolic Events in Patients Treated With Celecoxib, a Cyclooxygenase-2 Specific Inhibitor, Versus Ibuprofen or Diclofenac - The American Journal of Cardiology Vol. 89 February 15, 2002 Incidence of Hypertension, Edema, and Congestive Heart Failure Comparison of Thromboembolic Events in Patients Treated With Celecoxib, a Cyclooxygenase-2 Specific Inhibitor, Versus Ibuprofen or Diclofenac - The American Journal of Cardiology Vol. 89 February 15, 2002 Incidence of Hypertension, Edema, and Congestive Heart Failure Comparison of Thromboembolic Events in Patients Treated With Celecoxib, a Cyclooxygenase-2 Specific Inhibitor, Versus Ibuprofen or Diclofenac - The American Journal of Cardiology Vol. 89 February 15, 2002 Incidence of Hypertension, Edema, and Congestive Heart Failure Comparison of Thromboembolic Events in Patients Treated With Celecoxib, a Cyclooxygenase-2 Specific Inhibitor, Versus Ibuprofen or Diclofenac - The American Journal of Cardiology Vol. 89 February 15, 2002 Incidence of Hypertension, Edema, and Congestive Heart Failure Comparison of Thromboembolic Events in Patients Treated With Celecoxib, a Cyclooxygenase-2 Specific Inhibitor, Versus Ibuprofen or Diclofenac - The American Journal of Cardiology Vol. 89 February 15, 2002 Biology and therapy of fibromyalgia New therapies in fibromyalgia - Arthritis Research & Therapy Vol 8 No 4 Arnold Summary of findings from pharmacological studies in fibromyalgia 1. Serotonin and norepinephrine reuptake inhibitors improve pain, other symptom domains, function, quality of life, and global wellbeing in patients with fibromyalgia. 2. Selective serotonin and norepinephrine reuptake inhibitors (SNRIs) offer an alternative to cyclic medications (e.g., tricyclics) that are associated with safety and tolerability concerns. 3. The effect of SNRIs on reduction in pain associated with fibromyalgia is independent of their effects on mood. 4. Alpha 2 delta ligands also improve pain, other symptom domains, function, and global well-being in patients with fibromyalgia. 5. Alpha 2 delta ligands improve slow wave sleep. 6. Drugs associated with high risk of abuse and dependence should be avoided. Opiates may contribute to hyperalgesia if used chronically. 7. Although studies are limited, combinations of medications (e.g., combination of an SNRI and alpha 2 delta ligand) may be an option for patients who do not fully respond to a single agent or who have problems with tolerability at higher doses. Biology and therapy of fibromyalgia New therapies in fibromyalgia - Arthritis Research & Therapy Vol 8 No 4 Arnold Schematic representation of the neural patterns involved in the spasm-pain-spasm cycle. Adapted with permission. Commonly Used Muscle Relaxant Therapies for Acute Low Back Pain: A Review of Carisoprodol, Cyclobenzaprine Hydrochloride, and Metaxalone - CLINICAL THERAPEUTICS Commonly Used Muscle Relaxant Therapies for Acute Low Back Pain: A Review of Carisoprodol, Cyclobenzaprine Hydrochloride, and Metaxalone - CLINICAL THERAPEUTICS Severity of somnolence experienced with cyclobenzaprine hydrochloride 5 mg (David G. Borenstein, protocols 006 and 008, McNeil Consumer & Specialty Pharmaceuticals, Fort Washington, Pennsylvania, 2003). Commonly Used Muscle Relaxant Therapies for Acute Low Back Pain: A Review of Carisoprodol, Cyclobenzaprine Hydrochloride, and Metaxalone - CLINICAL THERAPEUTICS Commonly Used Muscle Relaxant Therapies for Acute Low Back Pain: A Review of Carisoprodol, Cyclobenzaprine Hydrochloride, and Metaxalone - CLINICAL THERAPEUTICS Limitações dos ensaios clínicos com drogas em Fibromialgia 1 - Duração curta 2 - Ponto primário dor (pontos secundários são avaliados de maneira inconsistente) 3 - Não há padronização para avaliar os sintomas 4 - Ponto primário é redução da dor frente ao placebo (não necessariamente melhora do quadro clínico) 5 - Não há consenso sobre conceituação de melhora clínica significativa da dor 6 - Presença de outras patologias (principalmente alterações psiquiátricas) 7 - Maior parte dos pacientes nos estudos são mulheres (extrapolação para homens?) The rationale for testing a dopamine 3 agonist in fibromyalgia is based on evidence that excessive adrenergic arousal may fragment sleep, and enhancement of dopaminergic neurotransmission at the D3 receptors in the mesolimbic hippocampus may reduce expression of arousal and improve sleep. Compared with the placebo group, those patients receiving pramipexole titrated over 12 weeks to 4.5 mg every evening had gradual and significant improvement in pain, fatigue, function, and global status. Central sensitization, a possible pathogenic mechanism of the chronic pain associated with fibromyalgia, is mediated, in part, by the binding of excitatory amino acids (glutamate and aspartate) to the NMDA receptor. NMDA antagonists may inhibit or attenuate central sensitization [97] and potentially reduce pain associated with fibromyalgia. In one clinical study, 48 female patients with fibromyalgia were treated with an open-label combination of tramadol 200 mg/day and increasing doses of dextromethorphan (50 to 200 mg/day), titrated to therapeutic effect or tolerability. Fifty-eight percent(28 of 48) responded to the addition of dextromethorphan and entered a double-blind phase in which the patients were randomized to dextromethorphan and tramadol or tramadol and placebo. A Kaplan-Meier drop-out analysis showed that significantly fewer patients on dextromethorphan and tramadol discontinued treatment compared with patients on tramadol alone [98]. Among these possible medications are the 5-HT3 antagonists (e.g., ondansetron and tropisetron), which have analgesic effects. A randomized, placebo-controlled, double-blind,10 day trial in 418 patients with fibromyalgia evaluated the short-term efficacy of tropisetron at doses of 5 mg/day, 10 mg/day, and 15 mg/day. Significant reduction in pain was noted only in those patients taking 5 mg/day and 10 mg/day, while the effects of tropisetron 15 mg/day were no different from placebo, suggesting a bell-shaped dose response curve Tramadol is a novel analgesic with weak agonist activity at the mu opiate receptor combined with dual serotonin and norepinephrine reuptake inhibition that may exert antinociceptive effects within both the ascending and descending pain pathways. Recently, fibromyalgia trials have focused on new selective serotonin and norepinephrine reuptake inhibitors (SNRIs), which are potent dual reuptake inhibitors but, unlike the tricyclics, do not interact with adrenergic, cholinergic or histaminergic receptors, or sodium channels, and, therefore, lack many side effects of tricyclics. Fibromyalgia patients often develop an increased response to painful stimuli (hyperalgesia) and experience pain from normally non noxious stimuli (allodynia) [6]. Both hyperalgesia and allodynia reflect an enhanced central nervous system processing of painful stimuli that is characteristic of central sensitization [7]. Fibromyalgia is a chronic, musculoskeletal pain condition that predominately affects women. Although fibromyalgia is common and associated with substantial morbidity and disability, there are no US Food and Drug Administrationapproved treatments. Chronic pain is one of the most common reasons prompting visits to healthcare providers; collectively, it possibly disables more people annually than heart disease and cancer combined. Applied topically as a gel or as a cream, NSAIDs have been widely studied as topical agents for sports injury pain, osteoarthritis, postoperative pain, and eye pain; these agents include diclofenac (patch/gel), ibuprofen (cream), ketoprofen (gel), piroxicam (gel), eltenac(gel), and aspirin. Mean concentration vs. time profiles of diclofenac in plasma (closed circles), subcutaneous adipose (open triangles) and skeletal muscle tissue (closed squares) after the final dose of a 3-day regimen of either MIKA Diclofenac Spray Gel 4% applied to the skin of the thigh (left panel) or with VOLTAREN® 50 mg enteric coated tablets given orally (right panel) in 12 healthy males. Results are presented as mean ± SE. Plasma (●), subcutaneous tissue (▲), skeletal muscle tissue (■) Main pharmacokinetic parameters for diclofenac obtained in plasma (total drug) and subcutaneous and skeletal muscle tissue (free drug) of the thigh in 12 healthy males after the final dose of a 3-day multiple dose regimen of either topical application of MIKA Diclofenac Spray Gel 4% or oral administration of VOLTAREN® 50 mg enteric coated tablets Favourable dermal penetration of diclofenac after administration to the skin using a novel spray gel formulation - Br J Clin Pharmacol 60 :5 573–577 573 Inhibitor ratios of NSAIDs in human blood/A549 cells IC50s COX-1 COX-2 COX-2 selectivity 1 <<0.01 >>100x L745,337 1 0.1 10x nimesulide, NS398, meloxicam, celecoxib 1 1 1x etodolac, diclofenac, sulindac 1 10 0.1x ibuprofen, ketoprofen, diflunisal, indomethacin, aspirin, tolmetin 1 10 0.1x flurbiprofen, naproxen, nabumetone/6MNA? Human whole blood assay • Predictive for GI damage? Rank 11 10 9 8 7 6 5 4 3 2 1 0 indomethacin flurbiprofen tolmetin naproxen aspirin ketoprofen ibuprofen sulindac diclofenac etodolac nabumetone Comparative upper GI toxicity - ARAMIS database (Singh et al., 1998) Meloxicam: Expected concentration in whole blood (15 mg, once daily) 1.8 1.8 COX-1 1.6 1.4 1.4 1.2 1.2 1.0 1.0 0.8 0.8 0.6 0.6 0.4 0.4 0.2 COX-2 0.2 168 0.0 192 0.0 0 24 48 72 96 120 Time (hours) 144 Estimated IC50 for COX Drug Concentration [µg/mL] 1.6 Effect of meloxicam on platelet COX-1 and monocyte COX-2 % inhibition ± SE 100 90 80 70 J1 J 1 COX-1 J 1 COX-2 60 J 1 50 40 1 30 20 15 mg, ss: Cmin -Cmax: 0.46 - 1.10 µg/mL 10 0 -10 0.01 J 1 1 0.1 1 10 Concentration in whole blood (µg/mL) 20 Importance of Drug Plasma Levels Meloxicam Naproxen 7.5 mg / day Cmin - Cmax : 0.23 - 0.55 µgml-1 2 x 500 mg / day Cmin - Cmax : 23 - 50 µgml-1 % Inhibition 100 80 COX-1 COX-2 60 40 20 0 0.01 0.1 1 10 100 concentration in whole blood µgml-1 0.01 0.1 1 10 100 concentration in whole blood µgml-1 Nimesulide: Expected concentration in whole blood (100 mg, twice daily) 4.0 3.0 COX-1 3.0 2.0 2.0 1.0 1.0 COX-2 0.0 0 24 48 72 96 120 Time (hours) 144 168 0.0 192 Estimated IC50 for COX Drug Concentration [µg/mL] 4.0 Nimesulide: Expected concentration in whole blood (200 mg, twice daily) 4.0 4.0 3.0 COX-1 3.0 2.5 2.0 2.0 1.5 1.0 1.0 COX-2 0.5 0.0 0 24 48 72 96 120 Time (hours) 144 168 0.0 192 Estimated IC50 for COX Drug Concentration [µg/mL] 3.5 Meloxicam • • • • Selective COX-2 inhibitor in vitro. Preferential inhibitor of PG release at inflammatory sites. Potent anti-inflammatory drug in vivo. Superior therapeutic margin. “A potent anti-inflammatory agent with reduced risk of side-effects” Structures of COX-2-selective compounds O O Meloxicam Nimesulide CH3 N O OH CH3 NH DuP 697 S O S NO2 Br S S O H N N CH3 F O CH3 S O F O Etodolac Flosulide NS-398 F H3C H N COOH CH3 O O O O H H N N CH3 NO2 S O CH3 S O O O Structures of COX-2-selective compounds F SC 58125 O O MK 966 S CI 1004 F CH3 N O S N CF3 O (CH3) C N Na H N NH2 C (CH3) CH3 F S O O Celecoxib (SC58635) H2NSO2 N RS 57067000 O N N CF3 CH2 Cl CH3 CH3 N NH CH3 O Distribution of COX-2 Present under ‘basal’ conditions: CNS (cortex, hippocampus and amygdala), stomach (small amount in superficial mucosa), kidney. After treatment with hormones or other regulatory factors: Ovaries, kidney (macula densa). After treatment with LPS, cytokines etc.: Monocytes, macrophages, endothelial cells, synoviocytes, chondrocytes, osteoblasts, amnion, CNS, pulmonary epithelial cells, smooth muscle cells. Regulation of COX-2 Expression The COX-2 promoter contains binding sites for NFkB, glucocorticoids, IL-6 and other cytokines. COX-2 is upregulated by LPS, IL-1, TNF, EGF, PDGF, TPA and serum. Induction has been seen in macrophages, endothelial cells, fibroblasts, amnion cells, glomerular mesangial cells and osteoclasts. COX-2 down-regulated by glucocorticoids, cycloheximide and anti-sense agents. Inflammatory Role for COX-1 ? In rat peritoneal and alveolar macrophages COX-1 contributes to PG synthesis in basal and stimulated states. (Wilborn et al., 1995) Positive feedback between PGE2 and COX-2: PGs generated from endogenous COX-1 may be involved in the initial induction of COX-2. (Mertz et al., 1994) Overall increase of COX-1 levels in rheumatoid synovial tissue is likely due to markedly increased cellularity. (Crofford, 1996) Physiological Role for COX-2 ? Levels of COX-2 mRNA (similar to COX-1mRNA) High - prostate, lung Medium - stomach, small intestine, uterus and mammary gland Low - kidney, liver, pancreas, testis, thymus, brain (O’Neill & Ford-Hutchinson, 1994) STOMACH Under basal conditions COX-2 is present in the surface mucous cells of the fundic and pyloric regions of the stomach. (Iseki et al., 1995) Relationship between dose and GI toxicity Relative risk of toxicity (+ 95% confidence limits) 12 Ibuprofen Naproxen Indomethacin 6 0 low/medium high Dose (Garcia Rodriguez, 1997) Relative risk of toxicity (+ 95% confidence limits) Comparative upper GI toxicity of NSAIDs at high doses 36.4 20 10 0 (Garcia Rodriguez, 1997) Ibuprofen >1500 mg Naproxen Diclofenac Indomethacin >750 mg >100 mg >75 mg Celecoxib in RA patients • RA patients withdrawn from NSAID therapy and those with disease flare up used for study • subjects given placebo or celecoxib (40, 200 or 400 mg b.i.d.) for 4 weeks • relative to placebo, improvements in: • patient global assessment • duration morning stiffness • number of tender or painful joints • no increase in adverse events relative to placebo • at 400 mg b.i.d. no effect on platelet function (Lipsky & Isakson, 1997) Celecoxib in short term pain • subjects had >2 molars removed • given placebo, aspirin (650 mg) or celecoxib (100 or 400 mg) • pain assessed for up to 8 h; endpoint being requirement for rescue medication (RM) • placebo takers: 60% required RM within 1h, 90% within 3 h. • aspirin takers: 60% required RM within 4 h. • celecoxib takers: 50% required RM within 4 h. (Lane, 1997) Celecoxib in medium term pain subjects with painful knee osteoarthritis studied for 2 weeks given placebo, or celecoxib (40, 100 or 200 mg, b.i.d.) placebo takers 12% decrease in joint pain after 1 week 14% dropout rate over 2 weeks celecoxib takers 20-30% decrease in joint pain after 1 week 1% dropout at 100 mg dose; 4% dropout at 200 mg dose (Lane, 1997) DFU in primate pyresis I.p. temperature monitored in conscious monkeys given LPS (6 µg/kg, i.v.) followed 120 min later by vehicle, diclofenac (3 mg/kg, p.o.) or DFU (1 or 3 mg/kg, p.o.). Body temperature increased by max. of 2.07±0.07 °C after 140 min and remained elevated for up to 240 min. At 240 min body temperatures were 1.77±0.13 °C DFU: 1 mg/kg, 1.15±0.07 °C; 3 mg/kg, 0.10±0.10 °C diclofenac: 0.32±0.20 °C vehicle: conclude that pyresis is entirely dependent upon COX-2 activity (Chan et al., 1997) DFU in primate pyresis I.p. temperature monitored in conscious monkeys given LPS (6 µg/kg, i.v.) followed 120 min later by vehicle, diclofenac (3 mg/kg, p.o.) or DFU (1 or 3 mg/kg, p.o.). Body temperature increased by max. of 2.07±0.07 °C after 140 min and remained elevated for up to 240 min. (Chan et al., 1997) Eleveation in body temperature (°C) DFU in primate pyresis 2 1 0 vehicle (Chan et al., 1997) DFU (1 mg/kg) DFU (3 mg/kg) Diclofenac (3 mg/kg)