* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Effects of different doses in continuous veno
Survey
Document related concepts
Transcript
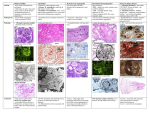
Renal Disease Dr. George Mellotte The Kidneys Two bean-shaped organs, each the size of a fist. Weighing ~0.5% of total body weight 20% of Cardiac output goes to Kidney Adrenal Glands Kidneys Rib Cage On Examination Move with Respiration Ballotable Can get above them Overlying resonance Bladder Rib Cage Function of the Kidney Primary balancing organ needed to keep blood in a stable state Remove waste products and toxins • Urea & Creatinine: • Drugs, toxic substances Maintain fluid and electrolyte balance • Total body water and fluid distribution – 50-70% of body weight is water • Sodium Potassium Maintain normal mineral balance • Calcium Phosphate Regulate acid/base balance Magnesium Function of the Kidney Endocrine Role of the Kidney Regulates blood pressure: Renin-angiotension System and aldosterone Adjusts final concentration of urine Antidiuretic hormone ADH Stimulates the production of red blood cells Erythropoietin Activates Vitamin D (Calcitriol, 1,25(OH)2D3) • Response to Parathyroid hormone Target Organ Damage Heart • Left ventricular hypertrophy • Angina or prior myocardial infarction • Prior coronary revascularization • Heart failure Brain • Stroke or transient ischemic attack Chronic kidney disease Peripheral arterial disease Retinopathy Estimating Renal Function Serum creatinine is widely used BUT Serum creatinine is based on muscle mass ‘Normal’ values are lower for elderly, female or physically inactive patients During early nephron loss, adaptive changes compensate to minimise rise in creatinine What is normal? Newer evidence suggests lower thresholds should be used, especially for women Creatinine : Men Women 50 - 115 umol/l 40 - 95 umol/l Defining Renal Failure Cr Clearance = (140 - age) × weight in Kg × SF serum Cr SF = 1.2 males / 1.05 females Normal GFR = 90-120mls/min Grade Mild Moderate Severe Endstage GFR 60 - 90ml/min 30 - 60ml/min 15 - 30ml/min < 15ml/min Sr Creatinine 100 - 150umol/l 150 - 250µmol/l 250 - 500µmol/l > 500µmol/L Cockcroft & Gault Formula Classification of Chronic kidney disease (CKD) 1 GFR (mL/min/1.73 m2) Kidney damage with normal / GFR 90 2 Kidney damage mild GFR 60–89 3 Kidney damage Moderate GFR 30–59 4 Kidney damage Severe GFR 15–29 5 End Stage Kidney failure < 15 (or dialysis) Stage Description eGFR can be thought of as equivalent to % kidney Function www.kidney.org/professionals/kdoqi GFR, glomerular filtration rate Key Concepts The importance of early identification Kidney Disease Cardiovascular Disease Focus on quality of care before starting Dialysis Slowing the progression of Kidney Disease Slowing the progression of Co - Morbid Disease Interplay of pathophysiology Progressive Kidney Disease Progressive Cardiovascular Disease JNC 7 Report on Hypertension For persons over age 50, SBP is a more important than DBP as CVD risk factor. Starting at 115/75 mmHg, CVD risk doubles with each increment of 20/10 mmHg throughout the BP range. The BP relationship to risk of CVD is continuous, consistent, and independent of other risk factors New Features and Key Messages Thiazide-type diuretics should be initial drug therapy for most, either alone or combined with other drug classes. Certain high-risk conditions are compelling indications for other drug classes. Most patients will require two or more antihypertensive drugs to achieve goal BP. If BP is >20/10 mmHg above goal, initiate therapy with two agents, one usually should be a thiazide-type diuretic. All Patients with nephropathy – particularly diabetic patients Target BP = 125/75 mmHg Re proteinuria - a lower BP is permissible/desirable in order to maximise ACEI & ARB – Target Proteinuria <0.3g/day Benefits of Lowering BP Average Percent Reduction Stroke incidence 35–40% Myocardial infarction 20–25% Heart failure 50% In stage 1 HTN and additional CVD risk factors, achieving a sustained 12 mmHg reduction in SBP over 10 years will prevent 1 death for every 11 patients treated. Ambulatory BP Monitoring ABPM is warranted for evaluation of “white-coat” HTN in the absence of target organ injury. Ambulatory BP values are usually lower than clinic readings. Awake, individuals with hypertension have an average BP of >135/85 mmHg and during sleep >120/75 mmHg. BP drops by 10 to 20% during the night; if not, signals possible increased risk for cardiovascular events. CVD Risk Factors Obesity (BMI >30 kg/m2) Physical inactivity Cigarette smoking Hypertension Hyperlipidaemia Diabetes mellitus Microalbuminuria Proteinuria eGFR <60 ml/min Mild Renal failure Creatinine>125 Age (older than 55 for men, 65 for women) Family history of premature CVD (men under age 55syndrome. or women *Components of the metabolic under age 65) Identifiable Causes of Hypertension Chronic kidney disease Renovascular disease Primary aldosteronism Sleep apnea Drug-induced or related causes Chronic steroid therapy and Cushing’s syndrome Pheochromocytoma Coarctation of the aorta Thyroid or parathyroid disease Laboratory Tests Routine Tests • ECG • Urinalysis • Blood glucose, and haemoglobin • Renal profile (potassium, creatinine, calcium) • Lipid profile, Hypokalaemia without diuretics – secondary cause? Optional tests • Microalbuminuria or albumin/creatinine ratio • Renal Ultrasound if renal impairment More extensive testing for identifiable causes is not generally indicated unless BP control is not achieved Lifestyle Modification Modification Approximate SBP reduction (range) Weight reduction 5–20 mmHg/10 kg weight loss DASH diet 8–14 mmHg Salt reduction 2–8 mmHg Physical activity 4–9 mmHg Moderation of alcohol consumption 2–4 mmHg Minority Populations In general, treatment similar for all demographic groups. Socioeconomic factors and lifestyle important barriers to BP control. Prevalence, severity of HTN increased in African Americans. African Americans demonstrate somewhat reduced BP responses to monotherapy with BBs, ACEIs, or ARBs compared to diuretics or CCBs. These differences usually eliminated by adding adequate doses of a diuretic. Risk Factors for Vascular Disease in CKD 1 Traditional Risk Factors Hypertension Left Ventricular Hypertrophy Hyperlipidaemia DOQI Guidelines – Treat similar to Diabetes/Post MI • Total Cholesterol < 4.0 mmol/l • LDL Cholesterol < 2.5 mmol/l • Will require statins – Beware of the patient with Hypocholesterolaemia Marker of nutritional deficiency & of increased mortality risk Risk Factors for Vascular Disease in Renal Failure 2 Renal Specific Risk Factors Proteinuria Anaemia Hyperhomocysteinaemia Hyperphosphataemia Uremia per se? Main Factors in Progression of Renal Failure Genetic / Racial determinants Age of onset of nephropathy Sex Underlying Disease Co-Morbid Disease Blood Pressure control Degree of Urinary Protein Excretion Progression in Renal Failure Destruction of Nephrons Remaining Nephrons Destruction of Nephrons Focal & Segmental Glomerulosclerosis Glomerular Hyperfiltration Renal damage induces hypertension via Plasma volume expansion, Sodium retention, Overactivity of both the sympathetic nervous system and the renin-angiotensin-aldosterone axis, Accumulation of circulating endogenous vasoactive substances. “Early CRF typically results in a 10-20 mm Hg increase in diastolic blood pressure until, and unless, renal impairment is identified and treated” Lancet 2000; 356 147-52 Relationship between achieved BP control and declines in GFR in clinical trials of diabetic & non-diabetic renal disease Blockade of the renin angiotension system is ‘RENOPROTECTIVE’ Evidence based medicine suggests preferred initial therapy is either an ACE Inhibitor or Angiotension II Receptor Blocker 3 Most trials note a 24%- 50% risk reduction of overt nephropathy, independent of BP reduction Recent evidence suggests a synergy between ACE inhibitors and AII Blockers Acute Renal Failure (ARF) A clinical condition – defined as an abrupt rise in Urea & Creatinine (with or without Oligiouria) 3 - 6 % of all hospital admissions develop ARF Incidence higher in complicated cases such as seen in Cardiac surgery Incidence ARFS (Acute Renal Failure Scotland) Study • Rise in Creatinine requiring intervention 237.9 pmp/year • Incidence requiring dialysis 118.7 pmp/year 60% treated in ICU 33% had preexisting renal disease Development of ARF increases the risk of death associated with a particular procedure - 5 fold CASES OF SEVERE ARF (%) Cause Developing Developed Countries Countries ATN Medical Disease Obstetrics Surgery 35 14 8 44 <1 39 Primary renal disease 10 10 Post renal failure 20 4 Other 13 2 Classification of ARF Pre-Renal Renal Pump Failure Hypovolaemia Glomerular 5% Tubular Post Renal Interstitial 10% Ischaemic Nephrotoxic 50% 35% Obstructive Pathophysiology of ARF Not simply a renal hypoperfusion problem Restoration of renal blood flow would correct urine flow Altered renal blood flow post ischaemic insult Changes in renal vasoconstriction/vasodilation Pooling of blood in Renal medulla Changes in inflammatory markers Increased leukocyte adhesion Increased pro-inflammatory markers (TNF, IL6) Increased pro-coagulation activation Tubular Alterations Effects of cytoskeletal Breakdown – loss of polarity Avoiding Renal Failure Prevent Dehydration in High Risk Patients Patients with S. Creatinine >125umol/l (?lower in females) Patients with Diabetes, Peripheral Vascular Disease or Myeloma Controlled trials recommend N. Saline @ 1ml/kg/hr 12 hrs pre & post procedures – particularly important pre Major Surgery or where contrast will be administered CRF patients have a fixed urinary concentrating deficit Role for N-Acetyl Cysteine to prevent contrast Nephropathy Adjust drug dosing where there is renal impairment Renal tubules more sensitive to the effect of nephrotoxins in the presence of renal hypoxia Guidelines for immediate management of patients with oliguria or anuria Assess & correct any respiratory or circulatory impairment Manage any life threatening consequences of renal dysfunction (hyperkalaemia, salt and water overload, extreme acidosis) Exclude obstruction of the urinary tract - Get Ultrasound Establish underlying cause(s) and institute prompt remedial action Get a drug history and alter prescriptions appropriately Get help from senior appropriately trained specialists Initial Investigations Blood Tests SPEP, Urine X-ray FBC, blood film, Coag screen, U/E, LFT's, Bone profile, ABG, Immunology if appropriateANA, DsDNA, C3,C4, CRP, ANCA & Anti GBM ASOT titre Electrolytes & Osmolarity Urine Microscopy Renal Ultrasound Isotope Perfusion scan Diagnostic Imaging Plain films (K.U.B.) Screening tool for renal stones. renal calcification Ultrasound safe, high quality images can be obtained on most patients Good screening tool – esp in ARF Used to evaluate renal size, renal masses and obstruction Intravenous Pyelography (I.V.P.) assesses the collecting system and urinary tract Increasingly being replaced by CT-IVP & MRI Not for patients at risk of contrast nephropathy Renal failure, Multiple Myeloma, Diabetics Volume depleted patients Nuclear Medicine – Renogram Used to asses renal function - DTPA or MAG3 MRI Increasingly being used in general nephrology MR Angiography MR Urography Pre-Renal Vs Established Renal failure Pre-renal RF Inadequate renal perfusion The kidneys concentrating power is normal and the urine produced is highly concentrated Established RF Failure of tubular function The kidney’s concentrating power is severely damaged and the urine produced is dilute Early restoration of effective circulation will avert ATN (Acute Tubular Necrosis) Renal Hypoperfusion V Established ARF Measurement Pre Renal ATN Urinary Na (mmol/l) <20 >40 Fractional excretion of Na (%) <1 >4 Only appropriate if diuretics not given Management of Pre Renal Failure Restore Renal Perfusion Correct Hypovolaemia Target CVP = 10 cm Correct Hypotension Target MAP > 75mmHg Use fluids in first instance Start Inotropic support if response insufficient No Diuresis Frusemide 100 - 250mg IV Pt must be euvolaemic Diuresis Measure hourly urine output & replace losses Lack of response indicates that ATN has developed Guidelines for fluid management in ARF Diuretic therapy Ineffective once ATN is established Inappropriate in inadequately treated prerenal • Will require a higher dose to achieve effect If overloaded • Diuresis should not be at the expense of hypotension • Aim negative balance 0.5 – 1L day unless clinically indicated Replacement for even balance is • Positive 0.5L for insensible losses • This is true irrespective of urine output! Drugs that induce renal damage Damage Class of drug Decrease in renal perfusion Diuretics, ACE inhibitors, Impaired intrarenal haemodynamics Tubular toxicity B-Blockers, vasodilators NSAID’s, radiocontrast Aminoglycosides, Allergic interstitial nephritis amphotericin, cisplatin lactams,(penicillins) NSAID’s ECG changes of Hyperkalaemia Peaked T waves, Flattened P wave Prolonged PR interval sinus arrest Wide QRS complexes & deep S waves Sine Wave V. Fib asystole Management of Hyperkalaemia Hyperkalaemia is a medical emergency and must be corrected immediately. V. Fib likely if K+ > 7.0 mmol/l (in ARF) Rx 1. 2. 3. acidotic 10-20 mls of 10% Calcium Gluconate 50 mls of 50% Dextrose with 12 IU Insulin over 30 mins followed by infusion @ 10ml/hour 50 - 100 mls of 8.4% NaHCO3 if Indications for renal replacement therapy Uncontrollable hyperkalaemia (K+ > 6.5) Salt and water overload unresponsive to diuretics Anuric and/or need to administer fluid/feed Severe uraemia (Creat >500umol/l ARF) Acidaemia Consider if pH <7.2 Multiple indications may trigger earlier intervention Chronic Renal Failure Defined as permanent loss of renal function Prevalence underestimated In USA - while only 0.1% of population require dialysis 5-10 % have renal dsease Most patients have no symptoms until CRF is advanced Advanced CRF often termed End Stage Renal Disease (ESRD) Defined as a GFR <15mls/min Typical symptoms are nausea, anorexia, fatigue, itch and bruising Typical signs are hypertension, ankle swelling, breathlessness and anaemia Chronic Renal Failure Clinical features Symptoms Nausea Fatigue Pruritus Signs Vomiting Obtunded Bruising Hypertension Oedema CCF Anaemia Pericarditis - late Neuropathy - late Laboratory features Urea & Creatinine – – – – – Hyperkalaemia Hypocalcaemia Hyperphosphataemia Metabolic Acidosis Normochromic Anaemia Radiology - Ultrasound – Small kidneys - often scarred Progression of Renal Disease Progression in CRF due in part to secondary factors unrelated to the activity of the initial disease focal segmental glomerulosclerosis Proteinuria Good evidence to support strategies to minimise amount of proteinuria Chiefly - ACE inhibitors - BP control - Diabetic control Normal Renal function Initiating factors Genetic or Enviromental Active Nephropathy Progression of renal disease Disease Modifiers Chronic Renal Failure Treatment Choice Haemodialysis C.A.P.D. /A.P.D. Transplantation Intermittent Complex Hospital Based Simple Independent Patient dependent Not a cure Problems Problems Peritonitis risk Poorly tolerated in cardiac Catheter Malfunction disease Protein losses Vascular access Most suitable Most Suitable Diabetics Active patients Elderly Patients with limited ability Patients living away from a to self care HD unit Nephrotic Syndrome Desirable Scarce resource Problems Graft Failure Infection risk Cancer Risk Only medically fit patients are Transplanted When to start Dialysis Accepted reasons Patient has symptoms of uraemia Kidney function approx 15% of predicted (GFR < 10-15mls/min) • Can wait until lower if patient remains well Patient develops high serum potassium levels The need to start Dialysis can often be delayed using Erythropoietin (EPO) to minimise symptoms Controversial - does “early start” benefit patient? ?less malnutrition ?less cardiac damage http://www.kidneypatientguide.org.uk/site/pdanim.html Peritoneal dialysis A silastic catheter in the peritoneal cavity Sterile dialysis fluid (supplied as 2 - 5L bags) An area for exchange in the home A pumping device (APD) The dialysis fluid is infused into the peritoneal cavity (which lies around the bowel) and allowed to dwell for 4-6 hours during which time toxic waste products enter the fluid. The fluid is the drained out and replaced – “An exchange”. Each exchange lasts 30 - 40 minutes For CAPD, For APD, done 4 times daily, 7 days a week done 4-6 times nightly using a machine “Home Choice” It is a home based system & requires a committed patient Automated Peritoneal Dialysis (APD) Automated Peritoneal Dialysis (APD) uses a machine to perform the fluid exchanges. Dialysis is done at home, at night while pts sleeps. The APD machine controls the timing of exchanges, drains away the used solution, and fills up the peritoneum with new solution When patient goes to bed, they connect their catheter to the APD machine's tubing and switch on. The APD machine does exchanges for 8 to 10 hours. In the morning, the patient disconnects from the machine. Haemodialysis http://www.kidneypatientguide.org.uk/site/HDanim.html It is a hospital based system – original type of dialysis A vascular access device Machine to pump blood & dialysis fluid An Artificial Kidney A Water treatment unit The machine pumps blood through one side of the membrane in the artificial kidney and dialysis fluid on the other side where toxic waste products are removed and electrolyte imbalances corrected. Each session lasts 4-5 hours and is needed 3 times per week this is independent of travelling time Haemodialysis Dialysis outcomes Survival is dependent on a number of variables Age starting dialysis Duration of Dialysis Cardiovascular Disease Diabetes Country of dialysis Expected death rate is 8-40 times that of controls (10 -15% pa) Transplantation Recipient Evaluation Cardiovascular risk, Viral screen, Urological assessment Only 30% patients on dialysis are fit for transplan list Average waiting time is gone up to 24 months All transplants done in Beaumont Patients require long term Immunosuppression Average 1 year graft survival 92% Average Graft survival 12-20 yrs Main cause of graft loss currently • Death of patient! - mainly due to Cardiac disease USRDS - Projected half-life of all transplants 1988 to 1995 The projected half-life for transplants living donors 12.7 years 1988 21.6 years 1995 Cadaveric donors 7.9 years 1988 13.8 years 1995, Censoring for patients who died with functioning grafts, Living Donors 16.9 years 1988 35.9 years 1995 Cadaveric Donors 11.0 years 1988 19.5 years 1995 NEJM Mar 2000; 342:605-612 Graft Loss Rejection Kidney fails due to “wearing out” Cancer related problems Return to Dialysis Patient dies from another disease with a functioning graft ZRAP052/0301 Date of Preparation: March 2001 Dipstick Urinalysis – Haematuria Dipstick urinalysis detects Haem protein (either red blood cells or haemoglobin) Highly sensitive but many false positive tests Confirm with urine microscopy. Transient haematuria is relatively common in young subjects and is not indicative of disease. • Yearly urinalyses in 1000 men between the ages of 18 & 33 haematuria – 39% at least once – 16% two or more occasions BMJ 1984 288:20 Negative tests reliably excludes abnormal haematuria Coexistent dipstick proteinuria is usually significant and should be investigated further Dipstick Urinalysis – Protein Standard dipstick detects albumin >300mg/l highly specific, but not very sensitive Measures urinary protein concentration The categories are only a rough guide Patients with persistent proteinuria should undergo a a 24-hour urine measurement of protein excretion. Proteinuria > 1g/24 hrs – consider renal biopsy may biopsy at lower levels Microalbuminuria Protein excretion above normal but below the threshold of “Standard Dipstick” Albuminuria normally <20mg/24 hrs (15 µg/min); Microalbuminuria = 30-300mg/24 hrs (20-200 µg/min) Albumin-to-creatinine ratio microalbuminuria = 2.25 - 3.4 mg alb/mmol creatinine Risk factor in Diabetic Nephropathy High incidence of false positives Microalbuminuria Early marker of Diabetic Nephropathy Usually develops within 10 years of onset of DM Duration of disease before onset of Microalbuminuria correlates with risk of progression to nephropathy Microalbuminuria < 10 years - Most progress Microalbuminuria > 10 years 30 -50 % progress Outcome much better than original studies – ?effect of active Rx Dipstick Urinalysis – other Pyuria - detects White cells in urine Leukocyte esterase – (75 – 95% sensitivity) Nitrites - indicates bacterial infection Enterobacteriaceae convert urinary nitrate to nitrite False negative at low colony counts UTIs Touted as simple and inexpensive screen for UTI May detect pyuria not associated with infection Nitrite alone insufficient for diagnosis Abnormalities on dipstick urinalysis seen with UTI should be shown to resolve with clinical cure Major Clinical Syndromes of Glomerular Disease Nephrotic Syndrome Nephritic syndrome Rapidly Progressive Glomerulonephritis Chronic Glomerulonephritis Persistent urinary abnormalities with no symptoms Proteinuria Indicative of significant renal disease Glomerular Proteinura » predominantly Albumin » >3.5g/day - classifies as “nephrotic range” » selectivity index useful » amount correlates with long term prognosis Tubular Proteinuria » usually < 2 g/day » due to a failure to reabsorb small molecular weight proteins e.g.. B2 Microglobulin Light Chain Disease Glomerular Proteinuria Primary GN Minimal Change* IgA Nephritis FSGS* Membranous* Hereditary Alport’s*, Infectious SBE, HIV*, Hepatitis, Immunological/Systemic Disease Vasculitis , SLE, PAN, Wegner’s, Goodpastures Diabetes*, Pregnancy-associated, Drugs Pencillamine, Gold, NSAIDs, Heroin*. Neoplasm's Solid organ CA*, Lymphoma, Leukaemia Others Amyloid*, Renal Tx rejection *Typically nephrotic range Nephrotic Syndrome Hypoalbuminaemia, (<30g/L) Proteinuria (>3.5g/day) Generalised oedema (JVP = N) hypercholesterolaemia +/- hypertriglyceridaemia Associated with - Increased risk of infection - Increased clotting tendency Pt > 10 years of age should have a renal Biopsy Acute Poststreptococcal Glomerulonephritis Principally a disease of children (M>F) Characteristic 10 day latent period between sore throat and renal disease Nephrotic Urine - ‘Smoky Brown’ haematuria - oliguria Associated with oedema and hypertension Dx rising ASO titre, throat culture - streptococcal A, renal biopsy Rapidly Progressive Glomerulonephritis Progression to ESRF within weeks or months of onset Focal necrotizing GN with crescent formation on renal biopsy Can form part of a vasculitic process Anti GBM disease ANCA positive vasculitis Churg strauss SLE any CT Can complicate any primary GN Goodpasture’s disease Wegners Granulomatosis microscopic polyarteritis disease Outlook for recovery poor unless treated early Steroids & Cyclophosphamide Pulse Methyl Prednisolone - plasmapheresis IgA Nephritis Commonest form of GN worldwide - 30% typically young males (M:F = 3:1) 66% - macroscopic haematuria following (1224 hours) onset of sore throat or URTI may produce ARF - often recurrent 33% -- persistent proteinuria and haematuria No serological marker 50% raised circulating IgA levels Long term risk of CRF = 25 - 50% Membranous Glomerulonephritis Clinical Features 80-90% > 30 years of age - m:f = 2:1 80% have nephrotic range proteinuria (>3.5g/day) commonest cause in adults Microscopic haematuria found in 50% of adults 25% hypertensive at diagnosis 4 - 10% have underlying neoplasm Renal vein thrombosis (10 -20%) • Increased risk albumin < 20g/L Minimal Change GN 76% of Nephrotic syndrome in children accounts for only 25% adults 93% steroid sensitive in children - no need to biopsy Most relapse off steroids at least once. recurrent relapsers - cyclophosphamide excellent long term prognosis renal biopsy Normal on Light microscopy Effacement of foot processes on EM classical association - Hodgkins lymphoma, NSAIDS Focal Segmental Glomerulosclerosis Presents with nephrotic syndrome in 75% secondary FSGS consequent on glomerular scarring IgA Nephritis Post vasculitis Sickle cell disease Alport’s disease HIV infection IV drug abuse (Heroin) reflux Histology - focal & segmental sclerosis Can recur in renal Tx - 23% ~ graft loss 10% MesangioCapillary GN -MCGN (Membranoproliferative GN) Presentation - Nephrotic (50%) - Nephritic (25%) Histologically Type 1 - Subendothelial deposits Type 2 - Dense deposit disease Associated with low complenent levels C3 nephritic factor Partial lipodystrophy No treatment shown to be effective 50 % ESRF at 10 years Can recur in renal Tx - 15 -35% ~ graft loss 10% Autosomal Dominant Polycystic Kidney Disease 2 Types PKD 1 PKD 2 Prevalence (Europe) 1 : 500 - 1 : 1000 patients Sex 85% 15% 8 - 10% of dialysis Males = Females Clinical onset Typically 20’s - 50’s Pathophysiology Disease begins in utero Multiple cysts, lined by tubular-type cells Cysts contain uriniferous fluid, blood or pyogenic secretions Cysts can arise anywhere along the nephron only 1 - 5% of nephrons are involved Intervening areas show nephrosclerosis and chronic interstitial nephropathy Renal failure “70% by age 70” Progresses to ESRF in about 10yrs once serum creatinine rises above normal Rate of progression of CRF usually similar in families - Progression is faster with PKD1: Median age of ESRF = 56 years PKD2: Median age of ESRF = 68 years high BP - gross haematuria proteinuria - pregnancy male sex - larger kidneys Age at presentation Subaracnoid Haemorrhage Risks & Prevalence overestimated Berry aneurysms 4% young adults rising to 10% in elderly 10% 65% risk of rupture Tend to cluster in families Prevalence in asymptomatic patients is felt to be lower Role of screening controversial Risk of hypertensive stroke or intracerebral haemorrage is still 10x higher than risk of subarachnoid GENETICS 2 genes involved PKD 1 Short arm of chromosome 16 Encodes polycystin 1 - ? adhesion PKD 2 Long arm of chromosome 4 Encodes polycystin 2 ? cation channel GENETICS Complete penetrance & variable expression Onset of the disease may be earlier if inherited from the mother Rate of progression of CRF varies from family to family & within families Positive Family history in > 60% Remainder ? spontaneous mutation DIAGNOSIS Ultrasound Very sensitive and specific Especially in Patient > 30 years of age Detects cysts as small as 1 - 1.5 cm Increased false negatives in young patients Characteristically multiple cysts in both kidneys which are large CT (with contrast ) More sensitive than USS Detects cysts of 0.5cm Definitive radiological test Genetics Only 1 - 5% of nephrons develop cysts even though they all carry a copy of the abnormal gene 2 hit hypothesis At a cellular level ADPKD seems to be acting as a recessive trait A ‘ second hit ‘ or mutation in the normal copy of the gene seems to have to occur sporadically before the nephron will produce cysts Renal Ostodystrophy Develops early in course of CRF First signs at GFR~ 40mls/min Treatment Goals PTH = Phosphate = Calcium = Treatment 150 - 300ng/l (2-3x normal) in ESRD 0.8 – 1.4 mmol/l 2.1 – 2.4 mmol/l Restrict phosphate in diet Phosphate Binders – Ca & non Ca based Activated Vitamin D (calcitriol / alfacalcidolol/parcalcitriol) New agents being developed - calcimimetics Increasing concern regarding vascular calcification