* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Document
Survey
Document related concepts
Transcript
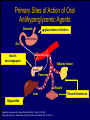
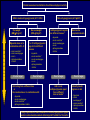
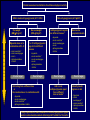
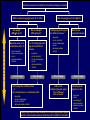
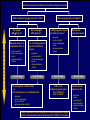
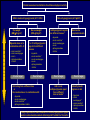
Canadian Diabetes Association 2003 Clinical Practice Guidelines for the Prevention and Management of Diabetes in Canada Getting to Goal in Type 2 Diabetes This material has been reviewed by the Canadian Diabetes Association for its medical and scientific accuracy. Presence of the Canadian Diabetes Association Partners in Progress mark does not constitute an endorsement of the products or services of GlaxoSmithKline Inc. Many patients have inadequate glycemic control 100 100 80 60 US (1988-1994)2 50% 50% 40 20 Percentage of subjects Percentage of subjects CAN (2003)1 0 7% A1C (%) 2 Harris 62% 60 40 38% 20 0 < 7% 1 Harris 80 S, et al, The Diabetes in Canada Evaluation (DICE) Study, ADA 2003, 2162-PO MI, et al. Diabetes Care 1999; 22:403–408. <7% > 7% A1C (%) Proportion of patients with A1C > 7.0 increases with duration of type 2 diabetes 67% 62% 53% 42% 31% <=2 3-5 6-9 10-14 15+ Years T2DM Harris,S et al. CDA 2003; Type 2 Diabetes and Associated Complications in Primary Care in Canada: The Impact of Duration of Disease on Morbidity Load. Proportion of patients with hypertension increases with duration of type 2 diabetes 71% 72% 10-14 15+ 64% 57% 53% <=2 3-5 6-9 Years T2DM Harris,S et al. CDA 2003; Type 2 Diabetes and Associated Complications in Primary Care in Canada: The Impact of Duration of Disease on Morbidity Load. Proportion of patients with dyslipidemia increases with duration of type 2 diabetes 66% 55% <=2 57% 58% 59% 3-5 6-9 10-14 15+ Years T2DM Harris,S et al. CDA 2003; Type 2 Diabetes and Associated Complications in Primary Care in Canada: The Impact of Duration of Disease on Morbidity Load. Proportion of patients with cardiovascular disease increases with duration of type 2 diabetes 48% 29% 21% 24% 15% <=2 3-5 6-9 10-14 15+ Years T2DM Harris,S et al. CDA 2003; Type 2 Diabetes and Associated Complications in Primary Care in Canada: The Impact of Duration of Disease on Morbidity Load. Proportion of patients with microvascular disease increases with duration of type 2 diabetes 62% 42% 44% 6-9 10-14 32% 21% <=2 3-5 15+ Years T2DM Harris,S et al. CDA 2003; Type 2 Diabetes and Associated Complications in Primary Care in Canada: The Impact of Duration of Disease on Morbidity Load. The evolution of management guidelines Studies including UKPDS have highlighted the importance of glycemic control in reducing complications New guidelines include tighter targets for glycemic control Guidelines recognize importance of treating all aspects of the condition Current guidelines therefore include targets for glycemic control lipid levels blood pressure UKPDS: decreased risk of diabetes-related complications associated with a 1% decrease in A1C Percentage decrease in relative risk corresponding to a 1% decrease in HbA1C Observational analysis from UKPDS study data Any diabetesrelated endpoint Diabetesrelated death 21% 21% ** ** All Myocardial cause mortality infarction 14% 14% ** ** Stroke Peripheral vascular disease† Microvascular disease 12% * 19% ** 37% †Lower extremity amputation or fatal peripheral vascular disease *P = 0.035; **P < 0.0001 43% ** ** Adapted from Stratton IM, et al. UKPDS 35. BMJ 2000; 321:405–412. Cataract extraction UKPDS: the benefits of improved glycemic control Improved glycemic control significantly reduces risk of diabetes-related complications UKPDS results indicated that a 1% reduction in A1C would reduce the risk of microvascular complications by 37%, but have less effect (16%) on macrovascular complications Further improvement in sustained glycemic control and reduction in the burden of cardiovascular disease are needed Stratton IM, et al. UKPDS 35. BMJ 2000; 321:405–412. UKPDS demonstrated loss of glycemic control with all agents studied A1C (%) 9 8 Conventional Glyburide Chlorpropamide Metformin Insulin 7 Upper limit of of normal = 6.2% 6 0 0 2 4 6 8 Years from randomization UK Prospective Diabetes Study Group. UKPDS 34. Lancet 1998; 352:854–865. 10 Overweight patients Cohort, median values Proportion of patients with A1C < 7.0% on monotherapy at 3, 6 and 9 years Overweight patients Proportion of patients (%) 100 Diet Insulin Sulfonylurea Metformin 80 60 50% 40 20 0 3 6 9 3 6 9 3 6 Years from randomization Turner RC, et al. UKPDS 49. JAMA 1999; 281:2005–2012. 9 3 6 9 Error bars = 95% CI The UKPDS demonstrated progressive decline of -cell function over time -cell function (%) 100 Start of treatment 80 60 40 20 P < 0.0001 0 –10 –9 –8 –7 –6 –5 –4 –3 –2 –1 1 2 3 4 5 6 Time from diagnosis (years) HOMA model, diet-treated n = 376 Adapted from Holman RR. Diabetes Res Clin Pract 1998; 40 (Suppl.):S21–S25. Canadian Diabetes Association 2003 Clinical Practice Guidelines for the Prevention and Management of Diabetes in Canada Glycemic Targets 1998 CDA Treatment Targets Level † Ideal Optimal* Suboptimal (normal nondiabetic) (target goal) (action may be required) (action required) <100 (.04-.06) <115 (<0.07) 116-140 (.07-8.4) >140 (>0.084) Fasting or premeal glucose level (mmol/L) 3.8-6.1 4-7 7.1-10 >10 Glucose level 1-2 h after meal (mmol/L) 4.4-7 5.0-11 11.1-14 >14 Glycated Hb (% of upper limit) e.g., HbA1c assay Inadequate ‡ CMAJ 1998; 159 (8 Suppl):S1-29 Hb = hemoglobin *These levels are likely related to minimal long-term complications but may be impossible to achieve in most paients with type 1 diabetes with current therapies †Attainable in the majority of people with diabetes but may not be adequate to prevent compications ‡These levels are related to a markedly increased risk of long-term complications, requiring a reassessment and readjustment of therapy 2003 CDA Recommended Targets for Glycemic Control A1C** (%) FPG/preprandial PG (mmol/L) 2-hour postprandial PG (mmol/L) Target for most patients 7.0 4.0-7.0 5.0-10.0 Normal range (considered for patients in whom it can be achieved safely) 6.0 4.0-6.0 5.0-8.0 A1C = glycosylated hemoglobin DCCT = Diabetes Control and Complications Trial FPG = fasting plasma glucose PG = plasma glucose Components of Glycemic Control A1C <7%, <6%* Fasting/Preprandial Plasma Glucose 2 h. Postprandial Plasma Glucose 4-7 mmol/L 4-6 mmol/L* 5-10 mmol/L 5-8 mmol/L* *If can be achieved safely Canadian Diabetes Association 2003 Clinical Practice Guidelines for the Prevention and Management of Diabetes in Canada Management of Hyperglycemia in Type 2 Diabetes Lifestyle Intervention The first step in treating type 2 diabetes Nutrition therapy and exercise can improve glycemic control Success of lifestyle intervention related to: patient’s initial fasting plasma glucose level amount of weight loss achieved by patient Only a minority of patients are able to attain treatment targets using lifestyle intervention alone. UKPDS 7: Response of FPG to Diet Therapy in Newly Diagnosed Patients N= 3044, newly diagnosed patients FPG at diagnosis 12.1+/- 3.7 mmol/L Diet counseling Patients with FPG 10-12 mmol/L needed reduction of 28% ideal body weight; to attain FPG <6 mmol/L 16% achieved FPG <6 after 3 months: in the group presenting with FPGs of 6-8 mmol/L 50% met this target in the group presenting with FPGs of 16-22 mmol/L only 10% were successful Metabolism, 1990; 39(3): 905-912 Antihyperglycemic Agents Primary Sites of Action of Oral Antihyperglycemic Agents -glucosidase inhibitors Stomach Carbohydrate Gut Insulin secretagogues Glucose (G) I I Adipose tissue Insulin Pancreas I Liver Biguanides Adapted from Kobayashi M. Diabetes Obes Metab 1999; 1 (Suppl. 1):S32–S40. Nattrass M & Bailey CJ. Baillieres Best Pract Res Clin Endocrinol Metab 1999; 13:309–329. Muscle Thiazolidinediones Key Recommendations • • • • • Antihyperglycemic agents should be initiated if glycemic targets not met after 2-3 months of lifestyle intervention Antihyperglycemic agents should be started concomitantly with lifestyle if A1C levels are greater than 9% The lag period before adding other agent(s) should be kept to a minimum to achieve glycemic targets within 6-12 months Unless contraindicated, metformin should be used first line; other agents should be considered in the order they appear in the treatment algorithm Insulin therapy should be initiated if targets cannot be achieved with lifestyle changes and oral therapy New Treatment Options for Type 2 Diabetes Stepwise treatment Diet/ exercise Oral Oral monotherapy combination Early aggressive combination therapy as required Oral +/- insulin Insulin Management of Hyperglycemia in Type 2 Diabetes Clinical assessment and initiation of nutrition and physical activity Mild to moderate hyperglycemia (A1C <9.0%) Overweight (BMI 25 kg/m2) Non-overweight (BMI 25 kg/m2) Marked hyperglycemia (A1C 9.0%) Mild to moderate hyperglycemia (A1C <9.0%) Overweight (BMI 25 kg/m2) • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor If not at target L I F E S T Y L E Biguanide alone or in combination with 1 of: Add a drug from a different class or Use insulin alone or in combination with: • biguanide • insulin secretagogue • insulin sensitizer* • alpha-glucosidase inhibitor Timely adjustments to and/or additions of oral antihyperglycemic agents and/or insulin should be made to attain target A1C within 6 to 12 months * When used in combination with insulin, insulin sensitizers may increase the risk of edema or CHF. The combination of an insulin sensitizer and insulin is currently not an approved indication in Canada. Dose-Effect Relationship Maximal Effect Therapeutic effect Half-maximal Side effect Half-maximal Maximal Dose Graph of theoretical dose-effect relationship for many drugs, showing that half-maximal dosages yield far more than 50% of the therapeutic effects and that side effects can increase as the dosage nears maximal levels. Riddle M. Combiningsulfonylureas and other oral agents.Am J of Med. 2000; 108(6A):15S-22S. Dose-Response Curve 1.5 1.0 30 20 0.5 10 0 0 500 1000 1500 2000 2500 Dose Dose-response curve showing GI related effects Riddle M. Combiningsulfonylureas and other oral agents.Am J of Med. 2000; 108(6A):15S-22S. GI Distress Patients (%) Reduction vs. placebo, HbA1c (%) 2.0 Mild to moderate hyperglycemia (A1C <9.0%) Non-overweight (BMI 25 kg/m2) • biguanide • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor L I F E S T Y L E 1 or 2† antihyperglycemic agents from different classes If not at target Add a drug from a different class or Use insulin alone or in combination with: • biguanide • insulin secretagogue • insulin sensitizer* • alpha-glucosidase inhibitor Timely adjustments to and/or additions of oral antihyperglycemic agents and/or insulin should be made to attain target A1C within 6 to 12 months * When used in combination with insulin, insulin sensitizers may increase the risk of edema or CHF. The combination of an insulin sensitizer and insulin is currently not an approved indication in Canada. † May be given as a combined formulation: rosiglitazone and metformin (Avandamet TM) Marked hyperglycemia (A1C 9.0%) • biguanide • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor If not at target L I F E S T Y L E 2 antihyperglycemic agents from different classes † Add an oral antihyperglycemic agent from a different class or insulin* Timely adjustments to and/or additions of oral antihyperglycemic agents and/or insulin should be made to attain target A1C within 6 to 12 months * When used in combination with insulin, insulin sensitizers may increase the risk of edema or CHF. The combination of an insulin sensitizer and insulin is currently not an approved indication in Canada. † May be given as a combined formulation: rosiglitazone and metformin (Avandamet TM) Marked hyperglycemia (A1C 9.0%) L E Basal and/or preprandial insulin L I F E S T Y If not at target Intensify insulin regimen or add • biguanide • insulin secretagogue** • insulin sensitizer* • alpha-glucosidase inhibitor Timely adjustments to and/or additions of oral antihyperglycemic agents and/or insulin should be made to attain target A1C within 6 to 12 months * When used in combination with insulin, insulin sensitizers may increase the risk of edema or CHF. The combination of an insulin sensitizer and insulin is currently not an approved indication in Canada. * * If using preprandial insulin, do not add an insulin secretagogue. Clinical assessment and initiation of nutrition and physical activity Mild to moderate hyperglycemia (A1C <9.0%) Non-overweight (BMI 25 kg/m2) Overweight (BMI 25 kg/m2) • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor 1 or 2† antihyperglycemic agents from different classes 2 antihyperglycemic agents from different classes † Basal and/or preprandial insulin • biguanide • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor • biguanide • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor If not at target If not at target If not at target If not at target L I F E S T Y L E Biguanide alone or in combination with 1 of: Marked hyperglycemia (A1C 9.0%) Add a drug from a different class or Use insulin alone or in combination with: • biguanide • insulin secretagogue • insulin sensitizer* • alpha-glucosidase inhibitor Add an oral antihyperglycemic agent from a different class or insulin* Timely adjustments to and/or additions of oral antihyperglycemic agents and/or insulin should be made to attain target A1C within 6 to 12 months Intensify insulin regimen or add • biguanide • insulin secretagogue** • insulin sensitizer* • alpha-glucosidase inhibitor Key Changes • A1C <7% (< 6% if can be achieved safely) • Aim to achieve targets within 6-12 months • Start with combination therapy or insulin for patients with A1C > 9% • Consider insulin at any stage of treatment • Vascular protection to further reduce cardiovascular risk Canadian Diabetes Association 2003 Clinical Practice Guidelines for the Prevention and Management of Diabetes in Canada Getting to Goal in Type 2 Diabetes This material has been reviewed by the Canadian Diabetes Association for its medical and scientific accuracy. Presence of the Canadian Diabetes Association Partners in Progress mark does not constitute an endorsement of the products or services of GlaxoSmithKline Inc. Supplementary Slides: Key Recommendations Recommendation 1 In people with type 2 diabetes, if glycemic targets are not achieved using lifestyle management within 2 to 3 months, antihyperglycemic agents should be initiated [Grade A, Level 1A ]. In the presence of marked hyperglycemia (A1C > 9.0%), antihyperglycemic agents should be initiated concomitant with lifestyle counselling [Grade D, Consensus]. Recommendation 2 If glycemic targets are not attained when a single antihyperglycemic agent is used initially, an antihyperglycemic agent or agents from other classes should be added. The lag period before adding other agent(s) should be kept to a minimum, taking into account the pharmacokinetics of the different agents. Timely adjustments to and/or additions of antihyperglycemic agents should be made in order to attain target A1C within 6 to 12 months [Grade D, Consensus]. Recommendation 3 This choice of antihyperglycemic agent(s) should take into account the individual and the following factors: Unless contraindicated, a biguanide (metformin) should be the primary drug used in overweight patients [Grade A, Level 1A]; and Other classes of antihyperglycemic agents may be used either alone or in combination to attain glycemic targets, with consideration given to the information in Table 1 and Figure 1 [Grade D, Consensus for the order of antihyperglycemic agents listed in Figure 1]. Recommendation 4 In people with type 2 diabetes, if individual treatment goals have not been reached with a regimen of nutrition therapy, physical activity and sulfonylurea [Grade A, Level 1A], sulfonylurea plus metformin [Grade A, Level 1A] or other oral antihyperglycemic agents [Grade D, Consensus], insulin therapy should be initiated to improve glycemic control. Recommendation 5 Combining insulin and the following oral antihyperglycemic agents (listed in alphabetical order) has been shown to be effective in people with type 2 diabetes: alpha-glucosidase inhibitors (acarbose) [Grade A, Level 1A] biguanide (metformin) [Grade A, Level 1A] Insulin secretagogues (sulfonylureas) [Grade A, Level 1A] Insulin sensitizers (thiazolidinediones) [Grade A, Level 1A] (The combination of an insulin sensitizer plus insulin is currently not an approved indication in Canada.) Recommendation 6 Insulin may be used as initial therapy in type 2 diabetes [Grade A, Level 1A], especially in cases of marked hyperglycemia (A1C > 9.0%) [Grade D, Consensus]. Recommendation 7 To safely achieve optimal postprandial glycemic control, mealtime insulin lispro or insulin aspart is preferred over regular insulin [Grade B, Level 2]. Recommendation 8 When insulin given at night is added to oral antihyperglycemic agents, insulin glargine may be preferred over NPH to reduce overnight hypoglycemia [Grade B, Level 2] and weight gain [Grade B, Level 2 ]. Recommendation 9 All individuals with type 2 diabetes currently using or starting therapy with insulin or insulin secretagogues should be counselled about the recognition and prevention of drug-induced hypoglycemia [Grade D, Consensus]. Supplementary Slides: Alternate Animation for Treatment Algorithm Clinical assessment and initiation of nutrition and physical activity Mild to moderate hyperglycemia (A1C <9.0%) Non-overweight (BMI 25 kg/m2) Overweight (BMI 25 kg/m2) • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor 1 or 2† antihyperglycemic agents from different classes 2 antihyperglycemic agents from different classes † Basal and/or preprandial insulin • biguanide • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor • biguanide • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor If not at target If not at target If not at target If not at target L I F E S T Y L E Biguanide alone or in combination with 1 of: Marked hyperglycemia (A1C 9.0%) Add a drug from a different class or Use insulin alone or in combination with: • biguanide • insulin secretagogue • insulin sensitizer* • alpha-glucosidase inhibitor Add an oral antihyperglycemic agent from a different class or insulin* Timely adjustments to and/or additions of oral antihyperglycemic agents and/or insulin should be made to attain target A1C within 6 to 12 months Intensify insulin regimen or add • biguanide • insulin secretagogue** • insulin sensitizer* • alpha-glucosidase inhibitor Clinical assessment and initiation of nutrition and physical activity Mild to moderate hyperglycemia (A1C <9.0%) Non-overweight (BMI 25 kg/m2) Overweight (BMI 25 kg/m2) • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor 1 or 2† antihyperglycemic agents from different classes 2 antihyperglycemic agents from different classes † Basal and/or preprandial insulin • biguanide • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor • biguanide • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor If not at target If not at target If not at target If not at target L I F E S T Y L E Biguanide alone or in combination with 1 of: Marked hyperglycemia (A1C 9.0%) Add a drug from a different class or Use insulin alone or in combination with: • biguanide • insulin secretagogue • insulin sensitizer* • alpha-glucosidase inhibitor Add an oral antihyperglycemic agent from a different class or insulin* Timely adjustments to and/or additions of oral antihyperglycemic agents and/or insulin should be made to attain target A1C within 6 to 12 months Intensify insulin regimen or add • biguanide • insulin secretagogue** • insulin sensitizer* • alpha-glucosidase inhibitor Mild to moderate hyperglycemia (A1C <9.0%) Overweight (BMI 25 kg/m2) • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor If not at target L I F E S T Y L E Biguanide alone or in combination with 1 of: Add a drug from a different class or Use insulin alone or in combination with: • biguanide • insulin secretagogue • insulin sensitizer* • alpha-glucosidase inhibitor Timely adjustments to and/or additions of oral antihyperglycemic agents and/or insulin should be made to attain target A1C within 6 to 12 months * When used in combination with insulin, insulin sensitizers may increase the risk of edema or CHF. The combination of an insulin sensitizer and insulin is currently not an approved indication in Canada. Clinical assessment and initiation of nutrition and physical activity Mild to moderate hyperglycemia (A1C <9.0%) Non-overweight (BMI 25 kg/m2) Overweight (BMI 25 kg/m2) • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor 1 or 2† antihyperglycemic agents from different classes 2 antihyperglycemic agents from different classes † Basal and/or preprandial insulin • biguanide • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor • biguanide • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor If not at target If not at target If not at target If not at target L I F E S T Y L E Biguanide alone or in combination with 1 of: Marked hyperglycemia (A1C 9.0%) Add a drug from a different class or Use insulin alone or in combination with: • biguanide • insulin secretagogue • insulin sensitizer* • alpha-glucosidase inhibitor Add an oral antihyperglycemic agent from a different class or insulin* Timely adjustments to and/or additions of oral antihyperglycemic agents and/or insulin should be made to attain target A1C within 6 to 12 months Intensify insulin regimen or add • biguanide • insulin secretagogue** • insulin sensitizer* • alpha-glucosidase inhibitor Clinical assessment and initiation of nutrition and physical activity Mild to moderate hyperglycemia (A1C <9.0%) Non-overweight (BMI 25 kg/m2) Overweight (BMI 25 kg/m2) • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor 1 or 2† antihyperglycemic agents from different classes 2 antihyperglycemic agents from different classes † Basal and/or preprandial insulin • biguanide • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor • biguanide • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor If not at target If not at target If not at target If not at target L I F E S T Y L E Biguanide alone or in combination with 1 of: Marked hyperglycemia (A1C 9.0%) Add a drug from a different class or Use insulin alone or in combination with: • biguanide • insulin secretagogue • insulin sensitizer* • alpha-glucosidase inhibitor Add an oral antihyperglycemic agent from a different class or insulin* Timely adjustments to and/or additions of oral antihyperglycemic agents and/or insulin should be made to attain target A1C within 6 to 12 months Intensify insulin regimen or add • biguanide • insulin secretagogue** • insulin sensitizer* • alpha-glucosidase inhibitor Mild to moderate hyperglycemia (A1C <9.0%) Non-overweight (BMI 25 kg/m2) • biguanide • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor L I F E S T Y L E 1 or 2† antihyperglycemic agents from different classes If not at target Add a drug from a different class or Use insulin alone or in combination with: • biguanide • insulin secretagogue • insulin sensitizer* • alpha-glucosidase inhibitor Timely adjustments to and/or additions of oral antihyperglycemic agents and/or insulin should be made to attain target A1C within 6 to 12 months * When used in combination with insulin, insulin sensitizers may increase the risk of edema or CHF. The combination of an insulin sensitizer and insulin is currently not an approved indication in Canada. † May be given as a combined formulation: rosiglitazone and metformin (Avandamet TM) Clinical assessment and initiation of nutrition and physical activity Mild to moderate hyperglycemia (A1C <9.0%) Non-overweight (BMI 25 kg/m2) Overweight (BMI 25 kg/m2) • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor 1 or 2† antihyperglycemic agents from different classes 2 antihyperglycemic agents from different classes † Basal and/or preprandial insulin • biguanide • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor • biguanide • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor If not at target If not at target If not at target If not at target L I F E S T Y L E Biguanide alone or in combination with 1 of: Marked hyperglycemia (A1C 9.0%) Add a drug from a different class or Use insulin alone or in combination with: • biguanide • insulin secretagogue • insulin sensitizer* • alpha-glucosidase inhibitor Add an oral antihyperglycemic agent from a different class or insulin* Timely adjustments to and/or additions of oral antihyperglycemic agents and/or insulin should be made to attain target A1C within 6 to 12 months Intensify insulin regimen or add • biguanide • insulin secretagogue** • insulin sensitizer* • alpha-glucosidase inhibitor Clinical assessment and initiation of nutrition and physical activity Mild to moderate hyperglycemia (A1C <9.0%) Non-overweight (BMI 25 kg/m2) Overweight (BMI 25 kg/m2) • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor 1 or 2† antihyperglycemic agents from different classes 2 antihyperglycemic agents from different classes † Basal and/or preprandial insulin • biguanide • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor • biguanide • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor If not at target If not at target If not at target If not at target L I F E S T Y L E Biguanide alone or in combination with 1 of: Marked hyperglycemia (A1C 9.0%) Add a drug from a different class or Use insulin alone or in combination with: • biguanide • insulin secretagogue • insulin sensitizer* • alpha-glucosidase inhibitor Add an oral antihyperglycemic agent from a different class or insulin* Timely adjustments to and/or additions of oral antihyperglycemic agents and/or insulin should be made to attain target A1C within 6 to 12 months Intensify insulin regimen or add • biguanide • insulin secretagogue** • insulin sensitizer* • alpha-glucosidase inhibitor Marked hyperglycemia (A1C 9.0%) • biguanide • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor If not at target L I F E S T Y L E 2 antihyperglycemic agents from different classes † Add an oral antihyperglycemic agent from a different class or insulin* Timely adjustments to and/or additions of oral antihyperglycemic agents and/or insulin should be made to attain target A1C within 6 to 12 months * When used in combination with insulin, insulin sensitizers may increase the risk of edema or CHF. The combination of an insulin sensitizer and insulin is currently not an approved indication in Canada. † May be given as a combined formulation: rosiglitazone and metformin (Avandamet TM) Clinical assessment and initiation of nutrition and physical activity Mild to moderate hyperglycemia (A1C <9.0%) Non-overweight (BMI 25 kg/m2) Overweight (BMI 25 kg/m2) • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor 1 or 2† antihyperglycemic agents from different classes 2 antihyperglycemic agents from different classes † Basal and/or preprandial insulin • biguanide • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor • biguanide • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor If not at target If not at target If not at target If not at target L I F E S T Y L E Biguanide alone or in combination with 1 of: Marked hyperglycemia (A1C 9.0%) Add a drug from a different class or Use insulin alone or in combination with: • biguanide • insulin secretagogue • insulin sensitizer* • alpha-glucosidase inhibitor Add an oral antihyperglycemic agent from a different class or insulin* Timely adjustments to and/or additions of oral antihyperglycemic agents and/or insulin should be made to attain target A1C within 6 to 12 months Intensify insulin regimen or add • biguanide • insulin secretagogue** • insulin sensitizer* • alpha-glucosidase inhibitor Clinical assessment and initiation of nutrition and physical activity Mild to moderate hyperglycemia (A1C <9.0%) Non-overweight (BMI 25 kg/m2) Overweight (BMI 25 kg/m2) • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor 1 or 2† antihyperglycemic agents from different classes 2 antihyperglycemic agents from different classes † Basal and/or preprandial insulin • biguanide • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor • biguanide • insulin sensitizer* • insulin secretagogue • insulin • alpha-glucosidase inhibitor If not at target If not at target If not at target If not at target L I F E S T Y L E Biguanide alone or in combination with 1 of: Marked hyperglycemia (A1C 9.0%) Add a drug from a different class or Use insulin alone or in combination with: • biguanide • insulin secretagogue • insulin sensitizer* • alpha-glucosidase inhibitor Add an oral antihyperglycemic agent from a different class or insulin* Timely adjustments to and/or additions of oral antihyperglycemic agents and/or insulin should be made to attain target A1C within 6 to 12 months Intensify insulin regimen or add • biguanide • insulin secretagogue** • insulin sensitizer* • alpha-glucosidase inhibitor Marked hyperglycemia (A1C 9.0%) L E Basal and/or preprandial insulin Intensify insulin regimen or add L I F E S T Y If not at target • biguanide • insulin secretagogue** • insulin sensitizer* • alpha-glucosidase inhibitor Timely adjustments to and/or additions of oral antihyperglycemic agents and/or insulin should be made to attain target A1C within 6 to 12 months * When used in combination with insulin, insulin sensitizers may increase the risk of edema or CHF. The combination of an insulin sensitizer and insulin is currently not an approved indication in Canada. * * If using preprandial insulin, do not add an insulin secretagogue.