* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Slide 1
Survey
Document related concepts
Transcript
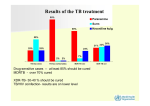
Group - 3 DOTS-Plus Meeting 12 Apr 2005 Task • Information system – Recording – Reporting • Linkages with field and follow-up for discharged patients • Drugs – Drug procurement – Drug packaging – Drug distribution system Patient flow (linkages with field and records) …1 • Fill sputum culture form for Cat II failure • Send sputum/patient to IRL with culture form and treatment card • RNTCP Lab committee to provide guidelines for packaging and transportation of sputum for culture • MDR diagnosis at IRL communicated to DTO by email, telephone and post/courier • MDR Lab register at IRL • DTO/MOTC conducts address verification • Patient sent to State MDR Hospital for admission Patient flow (linkages with field and records) …2 • Hospital fills up patient details record and treatment book and patient I-Card – Treatment book consists of Rx card, adverse reaction monitoring chart, bacteriology chart • Local DOT provider and family treatment supporter identified and trained • Discharge after one month with 1 week drug supply to patient and copy of treatment book, referral form and drugs for remaining IP (8 months) sent to DTO (courier/messenger) • DTO to send back portion of referral form to State hospital as feedback on receipt of patient and drugs Patient flow (linkages with field and records) ….3 • Sputum samples for culture as and when required to be sent to IRL by courier (patient could also go if possible) • To reduce number of cultures consider doing culture only when sputum AFB becomes negative. Reduce no. of cultures to the optimum scientifically acceptable. ??? • RNTCP to fund specimen shipment / travel of patient to IRL/State hospital • DTO to send copies of treatment book to State hospital at the end of every quarter • State hospital to maintain MDR TB register • State hospital to supply CP drugs to DTO 6-monthly (first supply before IP is over) Recording system • Laboratory request forms (will act as referral form to IRL) – Culture/Sensitivity form • • • • MDR Lab Register at IRL Patient details record (case sheet) at hospital Patient I-Card Patient treatment book – Consists of Rx card, adverse reaction monitoring chart & bacteriology chart • Referral form to DTO for ambulatory Rx • MDR TB Register at State hospital Reporting • Case finding report (quarterly, annually) • Conversion report (quarterly and annually; after 13 to 15 months later, just like RT report of DOTS) – E.g. patients initiated on treatment in Q1 2005 will be reported in the Q1 2006 report • Treatment outcome report (quarterly and annually; after 31 to 33 months later, i.e. after 10 quarters) – E,g. patients initiated on treatment in Q1 2005 will be reported in Q3 2007 • MDR-TB register to be computerized in the form of a line-list at State hospital/IRL level and networked • Separate quarterly drugs and lab supplies report from State Drug procurement • Procurement only at national level • Annual procurement with 6-monthly tranches • Two mechanisms available – ICB – GLC – GOI to take decision Drug procurement (…2) ICB Advantages • Lower cost • Readily acceptable to WB • Quality assured • Known system for country Disadvantages • Long lead time GLC Advantages • Shorter lead time • Quality assured • International standards Disadvantages • Higher costs • Port clearance for drugs sourced from outside country • Taxation • Mechanism of transfer of funds to GLC May be good to have 2 procurement mechanisms: ICB for WB funded States and GLC for GFATM funded states Drug packaging • Recommended packaging of drugs into multi-drug single day blisters • State hospital to further package drugs into 8-month IP and 6-month CP packs • Additional loose drugs need to be considered Drug distribution system • Manufacturer to send drugs directly to State hospitals • RNTCP to provide funds and guidelines for drug stores at hospital • State hospital releases drugs to DTO • Quarterly reporting of drug stocks to CTD