* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Wholesaler Taskforce
Prescription costs wikipedia , lookup
Drug discovery wikipedia , lookup
Pharmaceutical industry wikipedia , lookup
Pharmaceutical marketing wikipedia , lookup
Toxicodynamics wikipedia , lookup
Environmental impact of pharmaceuticals and personal care products wikipedia , lookup
Environmental persistent pharmaceutical pollutant wikipedia , lookup
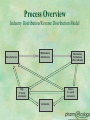
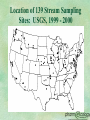
Identifying and Managing Hazardous Pharmaceutical Waste MIRT Meeting May 15, 2002 Charlotte A. Smith, R. Ph., M.S. President 414-258-8359 csmith@ pharmecology.org 262-814-2635 Founded Capital Returns, Inc. in 1991 Pharmaceutical reverse distributor Large quantity hazardous waste generator Applied RCRA definitions item by item Designated over 400,000 items Pioneered seminars to the regulated industry Sold Capital Returns, Inc. in 1997 Affiliated as Chief Regulatory Advisor Development of Knowledge and Systems to Minimize the Destructive Impact of Pharmaceutical Waste on the Environment To Insure Compliance with State and Federal Regulations in a Cost Effective Manner Pharmaceuticals Entering the Waste Stream Wastage of Raw Materials from Manufacturing Process Wastage at the Distributor/ Pharmacy/Healthcare Facility Wastage at the LTCF or other residential facility Expired Pharmaceuticals Wastage at the Consumer Level Metabolites Entering Wastewater How is Pharmaceutical Waste Generated at the Healthcare Facility? IV Preparation General Compounding Spills/Breakage Partially Used Vials/Syringes If Contaminated, Biohazardous Discontinued, Unused Preparations Unused Repacks (Unit Dose) Discontinued Indated Pharmaceuticals Patients’ Personal Medications Outdated Pharmaceuticals Pharmaceutical Industry’s Unique Challenges Substitution of less toxic chemicals usually not possible Lack of application of EPA regulations to finished dosage forms No readily retrievable source of information on specific products Lack of knowledge of environmental regulations and the need to comply RCRA Risk Management & Liability Civil and criminal liability Civil: State/USEPA enforcement Criminal: FBI, Attorney General, Grand Jury Corporate fines: $27,500 per violation/day Personal liability: fines and/or imprisonment No statute of limitations Managers up through CEO When is an Outdated Drug a Waste? At the time and place the decision is made to discard it Two EPA guidance letters to the industry: Merck & Co., 1981 BFI Pharmaceutical, 1991 Enables shipping of potentially creditable outdates to a reverse distributor as product History of Pharmaceutical Reverse Distribution 1987: Prescription Drug Marketing Act made returns processing by wholesalers & manufacturer reps more difficult Late 1980’s, early 1990’s: Several entrepreneurs entered marketplace Hospital oriented Retail oriented Manufacturer oriented Demographics of DEA Registered Reverse Distributors: 38 Facilities in 20 States* Illinois 6 Michigan Florida 6 Missouri Georgia 5 Washington Tennessee 2 Pennsylvania Arizona 2 Wisconsin New Jersey 2 California New York 2 Delaware North Carolina 1 Texas Indiana 1 Ohio Iowa 1 Utah * 2000 DEA FOIA List of Registrants 1 1 1 1 1 1 1 1 1 1 Process Overview Industry Distribution/Reverse Distribution Model Wholesalers, Distributors Manufacturers Mfg. 3rd party processors Pharmacies, Institutions, other indirects Return 3rd party processors Incinerator Reverse Distribution: Current Scenarios Decision to discard is made at the pharmacy/wholesaler By pharmacy/wholesaler personnel By a contracted company Pharmacy/wholesaler becomes the waste generator Decision to discard is made at the reverse distributor Reverse Distributor becomes the waste generator Benefits of Reverse Distribution as an Industry Development of core competency in receipt, handling, shipment of returnable items Results in millions of dollars being returned to healthcare industry Development of core competency in management of waste pharmaceuticals, RCRA and non-hazardous Reduces likelihood of inappropriate disposal by healthcare industry Case Study: Capital Returns, Inc. 20% to 30% of products received are nonreturnable based on manufacturers’ policies and become waste at CRI 10% of non-returnable items are characterized as hazardous waste 2% to 3% of all returns received become hazardous waste The Returns Industry Association Association of pharmaceutical reverse distributors established in May, 1998 Criteria includes a DEA registration to handle controlled substances in schedules II through V RIA provides a unified voice for the industry,and a commitment to high quality standards and protection of the environment Development of Guidelines for Minimum Regulatory Standards Proposed development of Education & Certification Program Federal and State Regulatory Agencies Governing Reverse Distribution DEA EPA FDA OSHA DOT DOH, Controlled Substance Board State Env Protection State Board of Pharmacy RIA Contact Information David Jenkins Executive Director 703-787-8574 [email protected] www.returnsindustry.com Which Discarded Drugs Become Hazardous Waste? P-listed chemicals Sole active ingredient U-listed chemicals Sole active ingredient Characteristic of hazardous waste Ignitability Toxicity Corrosivity Reactivity P-Listed Chemicals Acutely hazardous LD50 < 50mg/kg Sole active ingredient No concentration threshold Cause entire formulation to be hazardous Weight of container/solvent included >1 kg/month (2.2 lbs) = Large Quantity Generator Examples of P-Listed Pharmaceutical Waste Arsenic Epinephrine Nicotine Nitroglycerin Physostigmine Physostigmine Salicylate Warfarin >0.3% P012 P042 P075 P081 P204 P188 P001 U-Listed Waste May be U-listed due to toxicity, ignitability, corrosivity, reactivity Pharmaceuticals U-listed primarily due to toxicity Sole active ingredient Over 100 kg/month (220 lbs) = Small Quantity Generator Examples of U-listed Pharmaceutical Waste Chloral Hydrate(CIV) U034 Streptozotocin U206 Chlorambucil U035 Lindane U129 Cyclophosphamide U058 Saccharin U202 Daunomycin U059 Selenium Sulfide U205 Melphalan U150 Warfarin<0.3% Mitomycin C U010 U248 Chemotherapy Waste Seven chemotherapy agents are U-listed Waste protocols for “Chemo Waste” Empty vials, syringes, IV’s Treated as infectious medical waste through autoclaving and landfilling or low temperature incineration If not empty, should be placed into Hazardous Waste container “Empty” means all contents removed that can be removed through normal means Characteristic of Ignitability Aqueous Solution containing 24% alcohol or more by volume & flash point<140° F. A Liquid having a flash point <140° F. An ignitable compressed gas (certain aerosols) An oxidizer Hazardous Waste Number: D001 Examples of Ignitable Discarded Pharmaceuticals Rubbing Alcohol Paregoric (CIII) Cleocin T Topical Solution Retin A Gel Listerine Mouthwash Erythromycin Topical Solution Silver Nitrate (oxidizer) Collodion Based Preparations Characteristic of Corrosivity An aqueous solution having a pH < or = 2 or > or = to 12.5 Examples: Primarily compounding chemicals Glacial Acetic Acid Sodium Hydroxide Hazardous waste number: D002 Characteristic of Toxicity Approximately 40 chemicals which meet specific leaching concentrations Examples of potential toxic pharmaceuticals: Arsenic m-Cresol Barium Mercury (thimerosal) Cadmium phenylmercuric acetate) Chloroform Selenium Chromium Silver Lindane Evaluation of Thimerosal Toxicity per Merck Index, Twelfth Edition: Thimerosal (C9H9HgNaO2S) mol. wt. 404.82 C H Hg Na O S 26.70% 2.24% 49.55% 5.68% 7.90% 7.92% 1:1000 Solution: Thimerosal may be used as a preservative in a concentration of 1:1000, which means, by definition, 1 gram in 1000ml of solution. Since thimerosal is 49.55% mercury (see above): 1g thimerosal x 49.55% = 0.4955g mercury. So: 1g thimerosal = 0.4955g Hg 1000ml 1000ml 495.5mg Hg = 495.5mg Hg . 1000ml 1 liter The regulatory limit for mercury is 0.2mg . 1 liter = Characteristic of Reactivity Meet eight separate criteria identifying certain explosive and water reactive wastes Nitroglycerin formulations are considered exempt as of August 14, 2001 under FR: May 16, 2001. States must still adopt. Hazardous Waste Number: D003 Applicable USEPA Interpretative Letters & Hotline Responses Epinephrine Residue in Syringe not P042 Hotline December 1994 Discarded Nitroglycerine Pills are P081 Hotline September 1993; Modified Aug 14, 2001? Disposal of CESQG hazardous waste Hotline August 1999 Anti-Neoplastic Agents January 30, 1986 April 25, 1988 Washington State State-only Dangerous Waste Services National and Regional Seminars to Raise Awareness Confidential On-Site Reviews of Current Practices, Possible Areas of Concern Implementation Assistance in Developing Compliant Systems Web-based PharmEcology Wizard The PharmEcology™ Wizard Web-based database enabling search by product for waste management recommendations Search by NDC, product or generic name, active ingredient Recommendations citing federal regulations and recommended waste streams State regulation alerts if more stringent than federal Risk Management alerts based on professional knowledge (e.g. chemotherapy agents not regulated at the state or federal level) The PharmEcology Wizard Prototype 2-2576-5 Readi-Cat Future Considerations Identify which hazardous wastes as defined present a real threat at levels normally seen in pharmaceuticals Establish a relationship between professional pharmacy organizations and USEPA, state environmental regulatory agencies Work towards exemptions from and expansion of RCRA where appropriate Work towards consistent standards of enforcement Resources www. ourstolenfuture.org www. lindane.org/world/environment/water www. h2e-online.org “Safely Managing Hazardous Materials and Hazardous Waste,” ASHP Clinical Midyear, 2001, Handouts on CDRom RCRA On-Line www.epa.gov/rcraonline RCRA Hot Line 1-800-424-9346 Improper Discard of Toxic Drugs Hurts Environment, Leads to Fines, AJHP, Vol 58, #17 September 1, 2001 pp 1576-1578. Resources www. pharmecology.org Go to link to article “Bad Medicine” January 2001 Your Risks in Handling Outdated and Unusable Drugs: A Guide to JCAHO and Regulatory Standards. Capital Returns, Inc., 1998 Call 1-800-950-5479 A Guide on Hazardous Waste Management for Florida’s Pharmacies, www. floridacenter.org. Guidelines for Reverse Distributors: Minimum Federal Regulatory Standards, www.returnsindustry.com Appendix Endocrine Disruptors: Emerging Contaminants Cause for Concern? Endocrine Disruptors: chemicals that interfere with the normal function of the endocrine system Mimic hormone, trigger identical response, block a hormone Do not follow the normal dose/response curve Active at much lower doses, especially in the fetus and newborn Estradiols, testosterone, progesterone Lindane USGS Water Quality Study* First nationwide reconnaissance of occurrence of pharmaceuticals, hormones, other organic wastewater contaminants 139 streams in 30 states, analyzed for 95 different OWCs 82 of the 95 detected in at least one sample One or more OWCs found in 80% of stream samples 13% of sites had more than 20 OWCs *http://toxics.usgs.gov/pubs/OFR-02-94/index.html Location of 139 Stream Sampling Sites: USGS, 1999 - 2000 Drug Residues In Ambient Water: Initial Surveillance in New Mexico* NM Environment Department Scientific Laboratory Division Drug detection limits into low ppt (10 ng/L) Test included Darvon, Dilantin, Prozac other antidepressants, prednisone, estrogens, caffeine, tamoxifen 8 sites positive, 16 negative Next study will include antibiotics, lipidregulators, and cardiovascular pharmaceuticals * http://www.nmenv.state.nm.us/gwb/drugs.html New Mexico Drug Residue Sampling Red = Positive California has tested for lindane, reported contamination levels, banned use on humans for lice and scabies* *www.lindane.org/world/environment/ water/national.htm Our Stolen Future: How Endocrine Disruptors May Be Threatening Our Lives and Survival Seven years synthesizing research on endocrine-disrupting chemicals Extensive database Estrogen mimics, blockage of testosterone, disruption of thyroid hormones www.ourstolenfuture .org Generations at Risk: Reproductive Health and the Environment Ted Schettler, MD. Gina Solomon, MD, Maria Valenti, Annette Huddle Reproductive Physiology and Toxicology Reproductive and Developmental Effects of Selected Substances and Human Exposure A Guide to Investigating Environmental Threats Low sperm counts Infertility Genital deformities Hormonally triggered human cancers Neurological disorders in children Hyperactivity Attention deficit Developmental & reproductive problems in wildlife Early Warnings Copenhagen, Denmark: 1992 Review of 61studies by Niels Skakkeback, University of Copenhagen Average human male sperm counts dropped by almost 50% between 1939 and 1990 Incidence of testicular cancer jumped Undescended testicles and shortened urinary tracts rising among young boys Early Warnings DES (Diethylstilbestrol) Used in the 1950’s - early 1970’s to prevent miscarriages - potent estrogenic Resulted in daughters of mothers given DES developing clear-cell cancer of the vagina, other reproductive effects Sons not studied well, but indications of reproductive abnormalities, testicular cancer Early Warnings Steady 1% a year increase in breast cancer rates since WWII Exposure during prenatal period critical 51 synthetic chemicals now identified as hormone disruptors; at least half are persistant and resist natural decomposition Mother’s breast milk is contaminated Identification of Endocrine Related Pharmaceuticals Estrogens Progesterones Testosterones Corticosteroids Glucosteroids Thyroid Identification of Pharmaceuticals as Endocrine Disruptors* Chloroform: Reproductive Ketoconazole: Reproductive Nizoral® Lindane: Estrogen/Androgen Malathion: Thyroid Ovide® Permethrin: Androgen mimic Elimite®, Acticin®, Nix® *http://www.ourstolenfuture.org/Basics/chemlist.htm * Generations at Risk: Reproductive Health and the Environment Identification of Pharmaceuticals as Endocrine Disruptors* Nonylphenol: Estrogen Resorcinol: Thyroid Arsenic: Glucocortocoid Trisenox®, Atrivex® Mercury: Reproductive/Thyroid Thimerosal, Mersol®, Aeroaid® *http://www.ourstolenfuture.org/Basics/chemli st.htm Generations at Risk: Reproductive Health and the Environment