* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download A-5 Notes
Compact Muon Solenoid wikipedia , lookup
Grand Unified Theory wikipedia , lookup
ATLAS experiment wikipedia , lookup
Identical particles wikipedia , lookup
Weakly-interacting massive particles wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
Spectral density wikipedia , lookup
Standard Model wikipedia , lookup
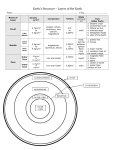
Science 8: Unit A: Mix and Flow of Matter Topic 5: Density What is density? Density is a property of a MATERIAL. It is similar to specific heat capacity. Density is the mass of a substance in a given volume. The standard volume for density is a ml (for gases and liquids) and cm3 (for solids) Note that 1 cm3 = 1 ml The density of a material is a constant physical property. Whether here or on Mars, the density of water is 1 g/ml at 20C and at standard pressure. The Standard for Density The density of water is 1 g/ml at sea level and at room temperature. This is the standard density that all other materials are compared to. Because water’s particles spread out when they turn solid, ice’s density is 0.92 g/ml, lower than water’s. This is why ice floats on water. This is weird because most materials’ densities increase when they become solid. Why do different materials have different densities? According to the particle model of matter, different materials have different particles and these particles can be larger or smaller, heavier and lighter. Also because of the different particle sizes only a certain amount of particles can fit in a given volume. What are some example densities? Air: 0.0012 g/ml Liquid mercury: 13.5 g/ml Gold: 19.3 g/cm3 Copper: 9.0 g/cm3 Lead: 11.3 g/cm3 The Sun: 1.4 g/cm3 (average density) White Dwarf Star: 1’000’000 g/cm3 (theory) The White Dwarf Star: A Case Study A white dwarf star is a dead star. Once our sun burns up all of its hydrogen fuel its gravity will continue to collapse its mass until most of it is squeezed into a volume about the same size as our Earth. Remember that the sun has the mass of 332’946 Earths. This makes the white dwarf very dense, so dense in fact that you would not be able to lift up a pinch full. How do we calculate density? Density = mass ÷ volume. Remember units are g/ml (liquids/gases) or g/cm3 (solids) Short Version of formula: D = m/v Example Example: Tommy wants to know why he’s gained two pounds weight even though he’s been working out four days now. He takes a 18 g sample of fat (taken from a steak) and measures its volume to be 20 cm3. What is the density of body fat? D = m/v = 18g/20 cm3 = 0.9 g/cm3 . He then measures the muscle tissue from the steak and measures the mass of 10 cm3. It turns out to be 10.6 g. So… D = m/v = 10.6g/10 cm3 = 1.06 g/ cm3 So he is replacing less dense fat with denser muscle. Heat and Density Solids tend to be denser than liquids which are denser than gases. When you heat a substance the particles gain thermal energy and spread out. This lowers density. So hot water is less dense than cold water. But remember water is weird: it’s denser as a liquid than a solid.