* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Composition of Earth Outline: • Earth`s Stats and internal structure
Survey
Document related concepts
Transcript
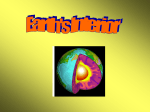
Composition of Earth Outline: • Earth’s Stats and internal structure – Chemical v. Physical Properties • Mohorovičić discontinuity=“Moho” • Composition of Minerals The Earth’s Stats heterogeneous in composition 3rd planet from sun 93,000,000 miles from sun Diameter ~8,000 miles Circumference ~25,000 miles Mass 5.9x1021 tons (5,900,000,000,000,000,000,000 tons) Composition of Continental Crust, Weight percent Oxygen (O) 46.6% Silicon (Si) 27.7% Aluminum (Al) 8.1% Iron (Fe) 5% Magnesium (Mg) 2.1% Calcium (Ca) 3.6% Potassium (K) 2.6% Sodium (Na) 2.8% All others <1.5% O + Si= 74.3% Composition of Whole Earth, Weight percent Iron (Fe) 35% Oxygen (O) 30% Silicon (Si) 15% Magnesium (Mg) 13% Nickel (Ni) 2.4% Sulfur (S) 1.9% Calcium (Ca) 1.1% Aluminum (Al) 1.1% All others <1% Earth’s Internal Structure Layers defined by composition (P-waves vs. S-wave and the Earth’s Interior) 1. Crust Continental-30-50 km thick, thickest beneath mts.; mostly granitic Oceanic- 7 km thick; basalt Base is defined by abrupt change in seismic characteristics of rx 2. Mantle 3. Core Earth’s Interior by waves Mohorovičić discontinuity=“Moho” Mantle shows increase in velocity to depths 70 km below ocean 120 km below continents Sudden decrease may be due to partial melting Low velocity separates: Lithosphere=crust and uppermost mantle Asthenosphere=mantle below low velocity zone 2900 km=abrupt increase in velocity Layers of the Earth by Physical Properties 1. Lithosphere – Upper most top of Mantle – Includes crust (less dense than rocks beneath) e.g., Continental and Oceanic Oceanic thinner than Continental 1. Asthenosphere – Hot, slowly flowing, weak rock – Near top of mantle/beneath lithosphere – Plastic (solid, but jello-like) 2. Mesosphere (Mantle) – Solid – Rocks=mostly iron, magnesium silicates – Moho marks boundary between mantle, crust 3. Core – Outer-liquid, mostly iron – Inner-solid, comp sim. to outer Composition of minerals and matter •Elements •Basic building blocks of minerals •100+ are known (92 naturally occurring) •Periodic Table •Atoms •Smallest particles of matter •Retains all the characteristics of element •Atomic structure •Central region = nucleus •Consists of protons (positive charges), neutrons (neutral charges) •Electrons –Negatively charged particles that surround nucleus –Located in discrete energy levels called shells •Formation of compound by combining two or more elements •Ionic bonding •Atoms gain or lose outermost (valence) electrons to form ions •Ionic compounds consist of orderly arrangement of oppositely charged ions •Covalent bonding •Atoms share electrons to achieve electrical neutrality •Covalent compounds are generally stronger than ionic bonds •Van der Waals or Molecular bonding •Usually strong covalent bonds with weaker electro-statically charged layers between. •Metallic bonding •Valence electrons are free to migrate among atoms •Weaker and less common than ionic or covalent bonds