* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Vancomycin dosing
Hormonal contraception wikipedia , lookup
Discovery and development of direct thrombin inhibitors wikipedia , lookup
Ciprofloxacin wikipedia , lookup
Electronic prescribing wikipedia , lookup
Pharmacokinetics wikipedia , lookup
Adherence (medicine) wikipedia , lookup
Dydrogesterone wikipedia , lookup
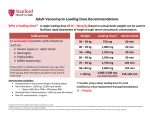
Clinical Guidelines for Use of Antibiotics VANCOMYCIN (Adult) Please always prescribe VANCOMYCIN in the Variable Dose Antibiotic section of the EPMA SUPPLEMENTARY drug chart (and add a ‘placeholder’ on the electronic drug chart). 1 Background Vancomycin is a glycopeptide antibiotic that is active against gram positive organisms, including methicillin resistant Staph. aureus (MRSA). The risk of renal toxicity (and ototoxicity) is increased when prescribed with other nephrotoxic drugs, such as gentamicin.1,2 2 Intravenous dose of VANCOMYCIN (intermittent bolus dosing) VANCOMYCIN requires determination and prescribing of both of the following: INITIAL LOADING DOSE ONGOING MAINTENANCE DOSE 2.1 BEFORE PRESCRIBING 1. Determine patient ACTUAL BODY WEIGHT 2. Determine patient Creatinine Clearance (see below) Creatinine Clearance (ml/min should be calculated manually using the Cockgroft Gault equation (below)or the online calculator function here. DO NOT use the eGFR. Cockcroft Gault equation: Notes for Cockcroft Gault equation: Age (years) Weight (kg) Serum Creatinine (micromols/litre) F = 1.23 in MEN F = 1.04 in WOMEN Compliance with these guidelines will be reviewed regularly by the Trust Prescribing Committee Please check the intranet to ensure you have the latest version. Note: This document is electronically controlled. The master copy is maintained by the owner department. If this document is printed it becomes uncontrolled. Contact Numbers: Microbiology Ext 4800; Antibiotics Pharmacist Ext 5033; 4PHA-GDL-031 Version 4 Adopted as Trust Policy: May 2015 Review Date: May 2017 Medicines Information Ext 5029 Page 1 of 8 Clinical Guidelines for Use of Antibiotics 2.2 PRESCRIBING INITIAL LOADING DOSE : Use Table 1 below to determine the loading dose: Use patient’s ACTUAL BODY WEIGHT1,6 Table 1: VANCOMYCIN LOADING DOSE Actual Body Weight (kg) Loading dose <60kg 60-90kg >90kg 1000mg 1500mg 2000mg (1g) (1.5g) (2g) Adapted from Thomson, AH et al, March 20096 2.3 MAINTENANCE DOSE: Based on CALCULATED Creatinine Clearance as indicator of RENAL FUNCTION using Cockcroft Gault equation: see here for online Cockcroft Gault calculator (via medicinescomplete). DO NOT use the eGFR. USE the calculated creatinine clearance for the patient in Table 2 below to determine the maintenance dose and dose interval for VANCOMYCIN The first maintenance dose should be given the same number of hours AFTER the LOADING DOSE as the ongoing dosing interval. Table 2: VANCOMYCIN MAINTENANCE DOSE SCALE Creatinine Clearance (mL/min) Dose (mg) Dosing Interval (hours) Dose point 8 >110 1500mg*see below 12 Dose point 7 90-110 1250mg 12 Dose point 6 75-89 1000mg 12 Dose point 5 55-74 750mg 12 Dose point 4 40-54 500mg 12 Dose point 3 30-39 750mg 24 Dose point 2 20-29 500mg 24 Dose point 1 <20 500mg 48 Adapted from Thomson, AH et al, March 20096 *UNLESS body weight <45kg: then use MAXIMUM dose of 1.25g (1250mg) until levels have been checked Compliance with these guidelines will be reviewed regularly by the Trust Prescribing Committee Please check the intranet to ensure you have the latest version. Note: This document is electronically controlled. The master copy is maintained by the owner department. If this document is printed it becomes uncontrolled. Contact Numbers: Microbiology Ext 4800; Antibiotics Pharmacist Ext 5033; 4PHA-GDL-031 Version 4 Adopted as Trust Policy: May 2015 Review Date: May 2017 Medicines Information Ext 5029 Page 2 of 8 3 Administration Clinical Guidelines for Use of Antibiotics The maximum concentration of vancomycin when prepared for infusion is 5mg/ml.3,4 Infusions should be given at a rate no greater than 10 mg/min is recommended.3,4 Rapid infusion may cause severe hypotension (including shock and cardiac arrest), wheezing, dyspnoea, urticaria, pruritus, flushing of the upper body (‘red man' syndrome), pain and muscle spasm of back and chest. Stop the infusion if they occur. A longer infusion time or premedication with an antihistamine may limit the reaction.3 4 VANCOMYCIN LEVEL MONITORING 4.1 Target VANCOMYCIN levels Standard dosing PRE-DOSE LEVELS Target range: 10-15mg/L1 High dose PRE-DOSE LEVELS for complicated infections Target range: 15-20mg/L 1,5,6,7 Complicated infections include: Bacteraemia (confirmed blood-stream infection) Endocarditis Osteomyelitis Meningitis HAP (Hospital acquired pneumonia) VAP (Ventilator associated pneumonia) Other infections caused by resistant organisms (microbiology will advise). 4.2 Timing of level Levels should be taken within one hour before next dose is due (PRE-DOSE or ‘TROUGH’ levels)1 4.3 The intial PRE-DOSE level should be taken: TWICE DAILY (12 hourly) dosing: JUST BEFORE THE FOURTH DOSE ONCE DAILY (24 hourly) dosing: JUST BEFORE THE THIRD DOSE ALTERNATE DAY (48 hourly) dosing: JUST BEFORE THE SECOND DOSE Compliance with these guidelines will be reviewed regularly by the Trust Prescribing Committee Please check the intranet to ensure you have the latest version. Note: This document is electronically controlled. The master copy is maintained by the owner department. If this document is printed it becomes uncontrolled. Contact Numbers: Microbiology Ext 4800; Antibiotics Pharmacist Ext 5033; 4PHA-GDL-031 Version 4 Adopted as Trust Policy: May 2015 Review Date: May 2017 Medicines Information Ext 5029 Page 3 of 8 Clinical Guidelines for Use of Antibiotics 4.4 Do I wait for the level to give the dose or not? Do not wait for the results of the pre-dose (trough) levels before giving the next dose 4.5 How often to repeat blood samples for drug levels TWICE a week if VANCOMYCIN levels are within range and urine output and renal function are STABLE If the dosage has been adjusted, then recheck levels after 2 days: TWICE DAILY (12 hourly) dosing: FOUR DOSES ONCE DAILY (24 hourly)dosing: TWO DOSES ALTERNATE DAY (48 hourly) dosing: ONE DOSE 4.6 Practicalities of taking blood samples for drug levels Send 3.5mL blood in a yellow top (SST) tube Ensure that the following are recorded on the blood form: Time the sample was taken Current dose of VANCOMYCIN Time level taken/hours post last dose. Indication for treatment 4.7 Interpretation of levels: The trough (pre dose) level should be between 10–15mg/L or 15-20mg/L depending on the indication (see section 4.1) If level is outside the desired range, FIRST check for technical explanations; for example: LEVEL taken at INCORRECT TIME INCORRECT timing of DOSAGE Use Table 3 below for guidance on how to adjust VANCOMYCIN dose according to measured pre-dose levels. Compliance with these guidelines will be reviewed regularly by the Trust Prescribing Committee Please check the intranet to ensure you have the latest version. Note: This document is electronically controlled. The master copy is maintained by the owner department. If this document is printed it becomes uncontrolled. Contact Numbers: Microbiology Ext 4800; Antibiotics Pharmacist Ext 5033; 4PHA-GDL-031 Version 4 Adopted as Trust Policy: May 2015 Review Date: May 2017 Medicines Information Ext 5029 Page 4 of 8 Clinical Guidelines for Use of Antibiotics Table 3: VANCOMYCIN trough level interpretation and maintenance dose adjustment Pre-dose (trough level) Maintenance dose adjustments (Refer to maintenance dose ladder above) < 5 mg/L Move up 2 DOSE POINTS on maintenance dose scale (Table 2) 5 to 10 mg/L Move up 1 DOSE POINT on maintenance dose scale (Table 2) For HIGH DOSING of COMPLICATED INFECTIONS (see section 4.1) move up one step on maintenance dose ladder 10 to 15 mg/L For STANDARD DOSING indications stay at current dose 15 to 20 mg/L Stay at current dose 20 to 25 mg/L Move down 1 DOSE POINT on maintenance dose scale (Table 2) without omitting any doses More than 25 mg/L Omit next dose and decrease by 2 DOSE POINTS on maintenance dose scale (Table 2) > 30 mg/L HOLD VANCOMYCIN DOSING Seek advice from microbiology or an antimicrobial pharmacist Adapted from Vancomycin Prescribing Guidance for: Musgorve Park Hospital (Taunton) and Leeds Teaching Hospitals. Compliance with these guidelines will be reviewed regularly by the Trust Prescribing Committee Please check the intranet to ensure you have the latest version. Note: This document is electronically controlled. The master copy is maintained by the owner department. If this document is printed it becomes uncontrolled. Contact Numbers: Microbiology Ext 4800; Antibiotics Pharmacist Ext 5033; 4PHA-GDL-031 Version 4 Adopted as Trust Policy: May 2015 Review Date: May 2017 Medicines Information Ext 5029 Page 5 of 8 Clinical Guidelines for Use of Antibiotics 5 When to refer to the antibiotic team: Stable therapeutic levels cannot be achieved Patient has severe renal impairment or is on dialysis Patient is not clinically improving 6. Additional Monitoring 6.1 Renal function There is some evidence that high dose vancomycin is associated with a greater risk of renal toxicity.6,8 For patients with a high trough concentration target (15-20mg/L) renal function MUST be monitored a minimum of twice a week. Patients with new acute kidney injury (AKI) should be referred to the antibiotic team and/or Microbiology for review and/or advice. 6.3 Hearing and balance Where patients are prescribing vancomycin alongside other nephrotoxic drugs (especially aminoglycosides such as GENTAMICIN or AMIKACIN) consider monitoring for signs of ototoxicity (changes to or loss of hearing, changes in balance).1 6.4 Full blood count Blood dyscrasias have been reported due to VANCOMYCIN therapy, including neutropenia and thrombocytopenia.4 It is recommended that the FULL BLOOD COUNT is monitored weekly for patients on vancomycin therapy where the duration of the course is greater than 1 week. Compliance with these guidelines will be reviewed regularly by the Trust Prescribing Committee Please check the intranet to ensure you have the latest version. Note: This document is electronically controlled. The master copy is maintained by the owner department. If this document is printed it becomes uncontrolled. Contact Numbers: Microbiology Ext 4800; Antibiotics Pharmacist Ext 5033; 4PHA-GDL-031 Version 4 Adopted as Trust Policy: May 2015 Review Date: May 2017 Medicines Information Ext 5029 Page 6 of 8 Clinical Guidelines for Use of Antibiotics 7. Dialysis patients Please liaise with the Dialysis Unit (5286) for all cases when drugs are administered on dialysis. For Haemodialysis (HD) patients usual dosing is a 1g IV loading dose then a single 500mg IV dose after every dialysis unless the previous level is above 20mg/L. Due to logistics trough vancomycin levels are used from the previous dialysis to decide if the next dose is given. Only in this circumstance is there no need to await the level from the current day before administering the dose. If a trough vancomycin level is above 20mg/L, omit further doses and wait until subsequent trough level is below 20mg/L. Vancomycin will be administered, in the usual case, on the dialysis unit and not on the ward. Supplies of vancomycin will be provided by the ward. Infuse a 1g vancomycin dose over 100 minutes. Type of dialysis Haemodialysis (HD) patients Vancomycin dose 1g IV loading dose and then 500mg IV stat after each HD unless previous level at dialysis is above 20mg/L. The level is recorded from the previous dialysis before each dose is given. On peritoneal dialysis or acute haemodialysis 1g IV loading dose monitor levels every 24 hours. When level is 20mg/L or under give another 1g IV dose. For patients with PD peritonitis, the treatment protocol involves administration of intraperitoneal vancomycin. Please seek advice from the Renal PD Nurses (Ext 5288 / 5286 / 7346). Compliance with these guidelines will be reviewed regularly by the Trust Prescribing Committee Please check the intranet to ensure you have the latest version. Note: This document is electronically controlled. The master copy is maintained by the owner department. If this document is printed it becomes uncontrolled. Contact Numbers: Microbiology Ext 4800; Antibiotics Pharmacist Ext 5033; 4PHA-GDL-031 Version 4 Adopted as Trust Policy: May 2015 Review Date: May 2017 Medicines Information Ext 5029 Page 7 of 8 8 References Clinical Guidelines for Use of Antibiotics 1. Rybak et al. Therapeutic monitoring of vancomycin in adult patients: A consensus review of the American Society of Health-System Pharmacists, the Infectious Diseases Society of America, and the Society of Infectious Diseases Pharmacists. Am J HealthSyst Pharm 2009; 66: 82-88 2. Rybak et al. Nephrotoxicity of vancomyrin , alone and with an aminoglycoside. Journal of Antimicrobial Chemotherapy 1990; 25: 679-687 3. ‘Medusa’ IV monograph for VANCOMYCIN IV. Accessed via http://medusa.wales.nhs.uk/IVGuideDisplay.asp (Feb 2015). 4. Summary of Product Characteristic: Vancocin®, date of last revision of the text 23 October 2008 (Flynn). Accessed via http://www.medicines.org.uk/emc/medicine/21291 (Feb 2015). 5. American Thoracic Society, Infectious Diseases Society of America. Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcareassociated pneumonia. Am J Respir Crit Care Med 2005; 171: 388–416 6. Thomson et al. Development and evaluation of vancomycin dosage guidelines designed to achieve new target concentrations. Journal of Antimicrobial Chemotherapy 2009; 63: 1050-1057. 7. Tunkel AR, Hartman BJ, Kaplan SL et al. Practice guidelines for the management of bacterial meningitis. Clin Infect Dis 2004; 39:1267–84. 8. Jeffres MN, Isakow W, Doherty JA et al. A retrospective analysis of possible renal toxicity associated with vancomycin in patients with healthcare-associated methicillinresistant Staphylococcus aureus pneumonia. Clin Ther 2007; 29: 1107–15. Compliance with these guidelines will be reviewed regularly by the Trust Prescribing Committee Please check the intranet to ensure you have the latest version. Note: This document is electronically controlled. The master copy is maintained by the owner department. If this document is printed it becomes uncontrolled. Contact Numbers: Microbiology Ext 4800; Antibiotics Pharmacist Ext 5033; 4PHA-GDL-031 Version 4 Adopted as Trust Policy: May 2015 Review Date: May 2017 Medicines Information Ext 5029 Page 8 of 8