* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Section: 2-1
Survey
Document related concepts
Transcript
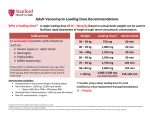
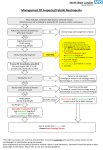
Medications – Vancomycin Administration Strength of Evidence Level: 3 PURPOSE: To provide accurate and safe administration of intravenous vancomycin. CONSIDERATIONS: 1. Vancomycin is used to treat serious or severe infections when other antibiotics are ineffective or contraindicated, including those caused by susceptible organisms, particularly gram positive organisms including Staphylococci, Methicillinresistant Staph aureus, Staph epidermidis and diptheroid organisms; Group A β-hemolytic strep, Streptococcus pneumoniae, Enterococci, Corynbacterium and Clostridium. 2. Vancomycin is administered intravenously for treatment of infections. Recommended dosing is as follows: a. Adults: 1 g IV every 12 hours. b. Children: 10 mg/kg IV every 6 hours. c. Infants: Initial dose 15 mg IV followed by 10 mg/kg IV every 12 hours. 3 Patient-specific dosing is adjusted based on weight, serum levels, clearance of the drug, and the volume of distribution of administered drug. 4. Vancomycin is primarily excreted via the renal pathway. Patients with renal impairment will likely need their dose and dose interval adjusted to reduce toxic effects of the drug. 5. Adverse Effects: a. Renal: Geriatric and neonatal patients are at a greater risk of nephrotoxicity; increased serum Cr and BUN, decreased Cr Clearance and (rare) interstitial nephritis. b. Ototoxicity: Damage to eighth cranial nerve. Hearing loss and permanent deafness has occurred. Rarely, vertigo, dizziness and tinnitus. c. Redman’s Syndrome: Presenting with sudden decrease in blood pressure and accompanied by flushing and/or maculopapular rash or erythematous rash on the face, neck, chest and upper extremities. Redman’s syndrome is usually associated with too rapid of infusion of medication and extending the infusion period may eliminate or reduce the extent of the reaction. Wheezing, dyspnea, angio-edema, urticaria and pruritis may also occur. d. Hematological: Leukopenia, eosinophilia and rarely, thrombocytopenia. e. Miscellaneous: Anaphylaxis, drug fever, chills, nausea, phlebitis. 6. Vancomycin should not be given IM due to the risk of tissue necrosis. Administer via the IV route with particular attention to preventing infiltration or extravasation of drug into the surrounding tissue. 7. Vancomycin doses should be infused over at least 60 minutes to decrease the likelihood of adverse effects. SECTION: 16.31 __RN__LPN/LVN__HHA 8. Concomitant use of other ototoxic and/or nephrotoxic drugs should be avoided due to the risk of additive toxicity. 9. Concomitant administration of Vancomycin with anesthetic agents in children has been associated with erythema and histamine-like flushing. 10. Vancomycin trough levels, BUN and creatinine should be obtained periodically as necessary. 11. Levels should not be drawn from the same vascular access device through which the dose was administered without flushing with a volume sufficient to clear the drug from the line. EQUIPMENT: None PROCEDURE: 1. Verify orders with physician: a. Route (IV only), dose and duration of therapy. b. Recommend laboratory orders as necessary. 2. Review patient’s medical record and prior history. 3. Adhere to Standard Precautions. 4. Identify patient and explain procedure. 5. Initiate therapy using appropriate vascular access device and administer drug solution over at least 60 minutes. Utilize appropriate control device based on dosage. 6. During therapy, closely monitor the patient for the following problems and contact the physician promptly for: a. Increases in serum Cr and/or BUN, and increases in Vancomycin trough levels. b. Any reported signs of ototoxicity. c. Any signs of hypersensitivity. 7. Teach patient to check access site daily for phlebitis and irritation and to report any pain at infusion site to avoid extravasation. 8. Review and discuss actions and side effects with the patient and/or caregiver and document on the patient visit record. 9. Dispose of supplies properly. AFTER CARE: 1. Document in patient’s medical record: a. Procedure and observations. b. Patient’s response to procedure. c. Instructions given to patient and/or caregiver. d. Communication with physician.