* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Macrolide Antibiotics
Survey
Document related concepts
Transcript
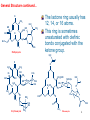
Macrolide Antibiotics Dr. Afshin Fassihi Department of Medicinal Chemistry MUI (Fall 1395) 1 Introduction Macrolide antibiotics are produced by species of Strptomyces. In 1950 the first drug of this class was isolated:Picromycin In 1952 Erythromycin and Carbomycin were introduced into clinic. O O CH3 HO HO CH3 H3C H3C H3C O H3C N CH3 CH3 R1 OH H3C OH CH3 CH3 O O O CH3 CH3 O OR2 CH3 CH3 Picromycin N H3C O O O CH3 CH3 HO O O H3C CH3 OH O Erythromycin CH3 2 General Structure They all contain three characteristic parts in the molecule: 1) A highly substituted macrocyclic lactone: Aglycone. 2) A ketone group on the aglycone ring (usually). 3) An amino desoxysugar, Glycon, Ketone group and, Aglycone In some of the macrolides, a neutral desoxysugar: Glycon Glycons are glycosidically desoxy attached to the aglycone ring. O H3C CH3 R1 OH CH3 CH3 H3C OH N HO O H3C O O CH3 CH3 CH3 O O OR2 CH3 CH3 OH O CH3 Erythromycin desoxy Glycon 3 General Structure continued… O CH3 HO H3 C HO CH3 H3C N CH3 O H3C O O O CH3 CH3 Methymycin The lactone ring usually has 12, 14, or 16 atoms. This ring is sometimes unsaturated with olefinic bonds conjugated with the ketone group. H3C O O O H3C CH3 R1 OH H3C CH3 CH3 OH HO H3C O CH3 O CH2CHO N O O O OR2 CH3 CH3 OH O Erythromycin CH3 CH3 CH3 HO H3CO O H 3C N(CH3)2 O O O O O H3C O O CH3 O O CH3 Miocamycin 4 Having a dimethyl amino group on the glycon part: Macrolide antibiotics are weak bases with different salts of pKa 6.0-9.0. As weak bases they are predominantly absorbed in the alkaline intestinal environment. O H3C CH3 R1 OH H3C CH3 CH3 OH N HO H3C O CH3 O O O CH3 CH3 O OR2 CH3 CH3 OH O Erythromycin CH3 5 General Structure continued… Macrolides are water-insoluble lipophilic molecules. Salts prepared by glucoheptonic acid (glucoheptonate=gluceptate) and lactobionic acid (lactobionate) are water soluble, COOH H OH H OH HO H H OH H OH COOH CHO H HO OH H HO H O H OH H O OH HO H CH2OH H OH H OH CH2OH CH2OH CH2OH Glucoheptonic acid HO OH Lactobionic acid Glucose 6 Stearic acid and laurylsulfuric acid salts are waterinsoluble. OH OH Stearic acid, C18H38O2 O O S O Laurylsulforic acid C12H26SO4 OH Macrolides are: • Stable in neutral aqueous solutions at room temperature. • Unstable in acidic conditions or at high temperatures. 7 Mechanism of Antimicrobial Activity Macrolides attach to the rRNA adjacent to the peptidyl transferase center in the 50s portion of bacterial ribosomes and inhibit the protein synthesis. They block the enzymes that catalyse the transfer of the new amino acid residue to the peptide chain, that is, prevent elongation in prokaryotic cells. 8 9 Erythromycin bound to 16s rRNA, E. coli 10 Spectrum of Antibacterial Activity Macrolides are similar to penicillins regarding their spectrum of activity: Macrolides are effective against most of the G(+) bacteria, cocci or bacillus. They are effective against penicillin-resistant strains. They are less effective against G(-) bacteria, though some strains of H. influenza and Brucella are sensitive to the antibacterial activity of this class of antibiotics. G(-) cocci, especially Neisseria Spp are also sensitive to macrolides. In contrast to penicillins, macrolide antibiotics are effective against Mycoplasma, Clamidia (G-), Campylobacter (G-) and Legionella (G-). 11 Chemical Instability of Macrolide Antibiotics Macrolides are unstable under acidic conditions and undergo an intramolecular reaction to form an inactive cyclic ketal. CH3 H3C The important C12-OH for binding to U2609 is gone HO The important C6-OH for binding to A2062 is gone 8 9 CH3 O H3C O 12 CH3 N 6 HO O O 1 CH3 CH3 O H3C CH3 1` 3 O O OR2 CH3 1`` CH3 OH O Anhydroerythromycin 6,9;9,12-spiroketal CH3 12 H+ O CH3 OH H3C CH3 9 OH HO OH 12 6 5 H3C O O 1 CH3 H 3O + O H3C 12 6 S H3C 1 O O 1 CH3 3 O O CH3 CH3 OH S1 3 O 8 9 HO CH3 H3C H H3C O CH3 S2 Erythromycin S2 Erythromycin 6,9-hemiketal -H3O+ CH3 CH3 H3C H+ H3C HO 8 9 O H3C O 12 CH3 H 3O 6 1 3 O O CH3 1 CH3 S2 Anhydroerythromycin 6,9;9,12-spiroketal CH3 6 OH H3C O O O CH3 H3C S1 H3C O 12 + 8 9 HO S1 O 3 O O CH3 S2 8,9-Anhydroerythromycin 6,9-hemiketal 13 Chemical Instability continued... The cyclic ketal is the cause of intestinal cramp which is reported after the use of erythromycin. Water-insoluble salts and enteric coated dosage forms of macrolides have less such a side effect. Water insoluble forms cannot take part in the reactions which occur in aqueous solutions. Stearate and laurylsulfate salts are insoluble salts of erythromycin. 14 Therapeutic Agents Erythromycin It is isolated from Streptomyces erythraeus in 1952. Erythronolide Aglycone: Erythronolide, Amino sugar: Desosamine, Neutral sugar: Cladinose. O H3C CH3 H3C OH HO CH3 H3C HO N CH3 OH Desosamine H3C O O O CH3 CH3 O O CH3 O Cladinose O CH3 H3C CH3 OH Erythromycin A 15 O Erythromycin H3C CH3 R1 OH CH3 CH3 H3C OH N HO O H3C O O CH3 CH3 CH3 O O OR2 CH3 CH3 OH O CH3 Erythromycin A: R1=OH, R2=CH3 Erythromycin B: R1=H, R2=CH3 Erythromycin C: R1=OH, R2=H Erythromycin D: R1=H, R2=H 16 Erythromycin Erythromycin has been the subject of chemical manipulations. The chemical manipulations are: 1. Preparation of acidic salts such as glucoheptonate, and stearate on the dimethyl amino group. 2. Ester formation on the 2`-OH of the amino sugar. Ethylsuccinate and propionate are examples. 17 Erythromycin These manipulations on erythromycin are aimed to: a) Increase the water solubility of the drug for parenteral dosage forms: • Hydrochloride, glucoheptonate and lactobionate salts. b) Increase the lipid solubility and hence chemical stability of the drug against aqueous acidic conditions as well as increase in oral absorption and masking the bitter taste of the drug: • stearate and laurylsulfate salts, • ethylsuccinate and propionate esters. 18 O Erythromycin esters and salts H3C CH3 HO OH CH3 CH3 H3C OH RO H3C O O O NH HA CH3 CH3 CH3 O O OCH3 CH3 CH3 OH O CH3 Water Soluble: Erythromycin Hydrochloride: R=H, Salt: HCl Erythromycin Gluceptate: R=H, Salt: structure A Erythromycin Lactobionate: R=H, Salt: structure B A: B: COOH COOH H OH H OH HO H H OH H OH CH2OH H HO OH HO H H O H OH OH O OH CH2OH CH2OH Water Insoluble: Erythromycin Stearate: R=H, Salt: CH3(CH2)16CO2H Erythromycin Ethylsuccinate: R=CO(CH2)2CO2C2H5 Erythromycin Estolat: R=COC2H5, Salt: CH3(CH2)11OSO3H 19 Clinical Application of Erythromycin It is used to treat The lower and upper part of the respiratory tract infections, Soft tissue G(+) infections, Mycoplasma pneumonia caused pneumonia, Campylobacter jejuni enteritis, Clamidia infections (G-). Gonorrhoea (G-). It is a good choice for penicillin-sensitive cases. 20 Clarithromycin 6-Methyl ether of erythromycin (semisynthetic), so: cannot undergo cyclic ketal formation: no GI cramp. More lipophylicity so: • Higher blood concentrations. • Longer intervals: every 12 hours OH is important to attach to A2062 by hydrogen bonding O H3C CH3 9 CH3 O HO Less active than erythromycin: H C Lacks a good binding site. H C 250 and 500 mg EC tablets. Its hepatic metabolism produces the more active 6-OH analogue, another reason for longer intervals. CH3 CH3 OH 3 N 6 HO 3 CH3 O O O CH3 O H3C O OH CH3 CH3 OH O Clarithromycin CH3 21 Azithromycin Azalide, a semisynthetic macrolide with a 15 membered ring. Stable under acidic conditions, because it doesn’t form cyclic ketal. Longer half-life, due to the nitrogen atom in the macrocyclic lactone ring, longer intervals: every 24 hours. H3C The important C12-OH for binding to U2609 Capable for necessary interactions The important C6-OH for binding to A2062 N CH3 H3C OH HO 12 H3C OH N HO 5 H3C In the treatment of urogenital infections caused by: N. gonorrhoeae and Chlamidia trachomatis. CH3 CH3 O O O 1 CH3 1` CH3 CH3 3 O O CH3 OCH3 1`` CH3 OH O 22 Azithromycin CH3 Erythromycyclamine Another semisynthetic derivative of erythromycin with 9-amino instead of 9-keto group. Lacks the GI cramp as a side effect. The same antibacterial effects as erythromycin. The important C12-OH for binding to U2609 NH2 H3C The important C6-OH for binding to A2062 CH3 OH HO CH3 CH3 H3C OH N HO O H3C O O CH3 CH3 CH3 O O OCH3 CH3 O Erythromycylamine OH 23 Dirithromycin A more lipophyl prodrug with high oral absorption. For infections of the upper and lower parts of respiratory system with only one daily oral dose. O HN H3C 9 CH3 OH HO CH3 O CH3 CH3 11 H3C O N HO O H3C O O CH3 CH3 CH3 O O OCH3 CH3 O Dirithromycin OH CH3 24 Dirithromycin Metabolism Unstable 9N,11O oxazine ring is easily hydrolyzed to yield erythromyclamine. O HN H3C 9 NH2 H3C OH HO CH3 OH H3C O O H3C O O HO CH3 O H3C CH3 O O Metabolism CH3 CH3 OCH3 CH3 O O Dirithromycin CH3 O O OCH3 CH3 CH3 O O N N HO CH3 CH3 CH3 11 CH3 H3C CH3 OH HO CH3 O OH OH CH3 Erythromycylamine 25 Troleandomycin Oleandomycin Oleandomycin is isolated from Streptomyces antiboticus. Troleandomycin is a prodrug of the former (acetyl of C2’-OH). Lacks the GI cramp due to the lack of 6-OH. The same antibacterial effects as erythromycin. O H3C 9 CH3 CH3 H3C OH N 6 HO H3C H3C O O O CH3 CH3 O O CH3 CH3 O Oleandomycin, R=H CH3 Troleandomycin R=COCH3 O OR 26 Mechanism of Microbial Resistance Methylation of a guanine residue on ribosomal RNA leads to lower affinity toward macrolides. Mutation of the Adenine 2058 in 23 rRNA causes a 10000 fold decrease in the attachment of the drug. 27 Mechanism of …continued The macrolide producing microorganism uses the same way to protect its rRNAs against this antibiotic. Maybe the antibiotic producing microorganisms are the source of R factors that cause the microbial resistance In some bacterial strains, an active efflux system is responsible for the resistance against macrolides. 28