* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Eeva – the Early Embryo Viability Assessment Test
Survey
Document related concepts
Transcript
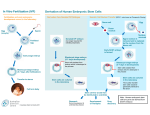
Eeva – the Early Embryo Viability Assessment Test INFERTILITY – A GLOBAL HEALTH ISSUE More than 72 million women around the world are estimated to be affected by infertility, a condition of the reproductive system1 Approx. 56% of women facing infertility issues seek medical assistance1 IVF OUTCOMES STRONGLY DEPEND ON ROUTINES AND TECHNOLOGIES APPLIED IN IVF LABORATORIES There are several factors during the IVF treatment journey that contribute to a successful live birth, including IVF drug treatment as well as technologies applied in the IVF laboratories. Among the latter, gamete assessment and preservation, fertilization and embryo cultivation, assessment and preservation are key steps. PREGNANCY TESTING CONSULTATION IVF DRUG TREATMENT OOCYTE & SPERM MGMT. & ANALYSIS OOCYTE & SPERM PRESERVATION FERTILIZATION EMBRYO CULTIVATION AND ASSESSMENT PRESERVATION (EMBRYO) OOCYTE AND SPERM RETRIEVAL EARLY PREGNANCY SUPPORT EMBRYO TRANSFER Due to the complex IVF workflow, it is essential to standardise and automate lab procedures and applied technologies, thereby ensuring consistency and objectivity in decision making, and maximizing the chances of IVF success.2 RH15-0029bf November 2015 1 of 3 Dedicated to the details that matter Eeva – the Early Embryo Viability Assessment Test EEVA The Early Embryo Viability Assessment (Eeva™) Early stage Eeva™ is designed to improve IVF outcomes at an early stage when used in conjunction with traditional morphology •innovative and non-invasive •used by IVF laboratories •analyses embryo development Quantitative data •on embryo’s potential development •optimizes IVF treatment path HOW DOES IT WORK? Eeva™ is a non-invasive embryo assessment test •enabled by a microscope equipped with a time-lapse camera with dark field illumination •fits into most standard incubators used in clinics The true innovation of Eeva™ is in its proprietary software: •automatically analyses embryo development •is built upon a unique predictive algorithm i Eeva™ provides medical staff with objective information on the development and viability of the embryos on day three after fertilisation The test helps identify embryos that have a high potential to become blastocysts*3 * an embryo that has developed for five to six days after fertilization EMBRYOS GROWING IN THE SPECIALLY DESIGNED EEVA DISH Embryo viability DAY 3-5 Embryoblast BLASTOCYST STAGE RH15-0029bf November 2015 2 of 3 Dedicated to the details that matter Eeva – the Early Embryo Viability Assessment Test The Eeva™ test was validated in a prospective, multicentre clinical trial with a total of 74 patients and 941 embryos* 3 Eeva™ used adjunctively to traditional methodology was able to predict blastocyst formation at the cleavage stage with 85% specificity* 3 *measurement of ability to predict embryos that became usable blastocysts Eeva used adjunctively to traditional methodology was able to reduce the false positive rate (test result falsely indicates that an embryo has a high viability) from 43% to 15% compared with traditional morphological methods alone4 Additionally, Eeva™ was able to increase the consistency of embryo assessment across embryologists3 1 Boivin J. et al. International estimates of infertility prevalence and treatment-seeking: potential need and demand for infertility medical care. Hum Reprod. 2007 Jun;22(6):1506-12. 2 Olofsson JI. et. al.Quality management systems for your in vitro fertilization clinic's laboratory: Why bother? J Hum Reprod Sci. 2013 Jan-Mar; 6(1): 3–8. available at http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3713573/ Accessed Nov 2015 3 Conaghan et al. Improving embryo selection using computer-automated time-lapse imaging plus day 3 morphology: results from a prospective multi-center trial. Fertility and Sterility: 2013;100:412-9. 4 Wong et al. Non-invasive imaging of human embryos before embryonic genome activation predicts development to the blastocyst stage. Nature Biotechnology, 2010;28:1115-21. The Eeva Test is available through Merck in the UK. The Eeva Test was in-licensed in 2014 from Auxogyn Inc. RH15-0029bf November 2015 3 of 3 Dedicated to the details that matter