* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Leishmaniasis Paul R. Earl Facultad de Ciencias Biológicas
Survey
Document related concepts
Transcript
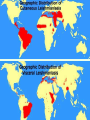
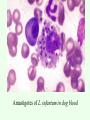
Leishmaniasis Paul R. Earl Facultad de Ciencias Biológicas Universidad Autónoma de Nuevo León San Nicolás, NL 66451, Mexico Policies and generalities. The complete management of diseases such as leishmaniasis, trypanosomiasis and malaria throughout the subtropical and tropical world awaits the much improved management of poverty which is the lack of resources, knowledge and industrial skills, and should include birth control. Diseases like the ones noted keep populations down, or they used to before the antibiotics arose in the 1940s. A country like landcleared treefree Haiti without decent water & sewage facilities punctuated by electricity failures is an example of Malthusian misery. That is what can happen if rural ignorance prevails. Will many tropical diseases be soon eradicated, and will their demise be accompanied by higher standards of living in much of the world? What will the planet be like after eradication? Cutaneous leishmaniasis usually divided into: Old World leishmaniasis caused primarily by L. donovani, L. tropica, L. infantum, L. major, L. aethiopica and New World leishmaniasis caused primarily by L. mexicana and L. braziliensis. Diffuse cutaneous leishmaniasis is caused primarily by L. aethiopica or L. mexicana. Mucocutaneous leishmaniasis (espundia) is caused primarily by L. braziliensis. The trypanosomatids of the genus Leishmania are the etiological agents of a variety of diseases, collectively known as leishmaniasis. It is prevalent throughout the tropical and subtropical regions of Africa, Asia, the Mediterranean, Southern Europe (Old World) and South and Central America (New World). The impact of the leishmaniasis on public health is modest, since many cases go unreported or misdiagnosed. About 12 million people are currently infected, and a further 367 million are at risk of acquiring leishmaniasis in 88 nations, 72 of which are developing countries. A little geography. The geographic distribution of leishmaniasis is cosmopolitan. Thus L. tropica and L. major, causing Oriental sore are found in Russia, Indonesia, equitorial Africa, in the west and east of the Mediterranean (Italy, Spain, Greece, Bulgaria and Romania). For instance, L. mexicana is found in the southeast of México in Tabasco, Campeche and other states, Central America and some countries of South America like Venezuela. Leishmania brasiliensis and its subspecies cause mucocutaneous leishmaniasis or espundia localized in South and Central American countries and the southeast of Mexico, principally Tabasco and Quintana Roo. L. peruviana causes Uta found in the Peruvian slopes of the Andes and Argentine highlands. L. donovani with its subspecies and closely related ones like L. infantum are found in the Mediterranean basin, Africa, regions of Asia and in America as L. donovani chagasi localized mainly in Brasil, Venezuela, Colombia, El Salvador, Guatemala and in Mexico in the Balsas valley. History. In 1900, Major William B. Leishman (1865-1926) performed a postmortem on an English soldier returning from Bengal who had died of "fever." He described finding enormous numbers of oval bodies 2-3 m in diameter in the splenic smears of this patient. In 1903, Captain James Donovan (18631915) in Madras, described similar findings in the splenic smears taken from the enlarged spleens of Indian patients who had died, presumably from malaria. It was Ronald Ross who named the new protozoa the Leishman Donovan body. Ninety percent of cases are found in Bangladesh, India, Nepal, Sudan and Brazil. There are 3 distinct species: L. infantum found in the Mediterranean basin, Central Asia and China, L. donovani in India and Eastern Africa, and L. chagasi in South and Central America. Leishmania ssp. are members of the family Trypanosomatidae, order Kinetoplastida. L. donovani and L. infantum are often geographically associated, and dogs worldwide are reservoirs for L. infantum. It is not especially important if other good names like L. mexicana are applied, because DNA sequencing has not yet proceeded far enough to differentiate species as in an efficient routine taxonomy. Leishmania needs a complete molecular revision. It was greatly helped by isozyme plus cluster analysis, but this is not enough. The list of 18 species now given requires scrutiny. PCR amplification of kinetoplast DNA minicircles using general kinetoplastid primers, for all Leishmania species and other kinetoplastids (k) was followed by the identification of the L. species complexes by hybridisation of the PCR products with specific kDNA probes. The polymorphic PCR-products were analysed by electrophoresis and the banding patterns compared with multilocus enzyme electrophoresis data. Life cycle. The reservoir of infection is the amastigote form of the parasite, present in animal reservoir hosts such as rodents, dogs, foxes, jackals and humans. Dogs are especially common reservoirs in the Mediterranean basin. They are infected worldwide and particularly in the Mediterranean basin as in Italy with L. infantum which is like L. donovani. Amastigotes of L. infantum in dog blood In a very simple way, some amastogotes remain in the skin causing cutaneous leishmaniasis, whereas others are carried by macrophages into internal organs thereby causing visceral leishmantiasis. Many different strains of leishmania can cause disease in humans with each strain differing in its reservoir, vector, geographic location and in the pathological lesions that it gives rise to. This makes creating a passable taxonomy difficult. The reservoir of infection is the amastigote form of the parasite, present in animal reservoir hosts such as rodents, dogs, foxes, jackals and humans. Dogs are especially common reservoirs in the Mediterranean basin such as in southern Italy. The infection is usually transmitted by the bite (blood feed) of the female sandfly, although human infection has been reported from blood transfusion, congenital transmission, and by sexual intercourse. In the sandfly vectors and on culture, the parasite takes up the promastigote form. Promastogotes, Courtesy of Tsehay Atlaw Rosette of promastigotes, Courtesy of Duncan Kennedy The sandfly vector, Phlebotomus, Lutzomyia or another similar one Visceral leishmaniasis. Nicolle in 1908 reported that mammals including dogs could act as reservoir hosts for the leishmania parasite. Using human volunteers, Swaminath and coworkers in 1942 proved that the leishmania parasite was transmitted by the phlebotomus sandflies. The only proven vector of the leishmania parasite is the blood-sucking female of the genus Phlebotomus in the Old World and Lutzomyia a copy in the New World. These genera are extremely similar. Visceral leishmaniasis is also known as Kala-azar (Hindi: kala black, azar sickness). The etiological agents belong to the Leishmania donovani complex like L. d. donovani, L. d. infantum and L. d. archibaldi in the Old World and L. d. chagasi in the New World. The Old World species are transmitted by the sandfly vector Phlebotomus sp. and Lutzomyia sp. in the New World. The acceptibility of this taxonomy depends on the reader. Mucocutaneous leishmaniasis has a clinical picture dominated by great destruction of the nasal mucosa, sometimes with respiratory complications. In visceral leishmaniasis or kala-azar, the parasites multiply abundantly in the medula of the spleen and bone marrow. The infection causes fever, headache, anorexia, loss of weight, splenomegly, hepatomegly, lymphadenopathy, pancytopenia, hypergammaglobinemia, anemia and darkening of the skin, tending towards a chronic state. Diagnosis. Laboratory diagnosis by direct and indirect methods can begin with the microscopy of the ulcer, using Giemsa stain. Cultivation of the parasite’s cells is an indirect method, true also for the infection of rodents that develop typical lessions. The antibody titers are low in cutaneus, high in mucocutaneous and very high in disseminated cutaneous or visceral leishmaniasis. Treatment. The great stumbling block is cost of both medication and hospitalization. The traditional pentavalent antimony compounds like pentamidine were the chosen medicaments over many decades, but they are no longer recommended, because of resistence. Amphotericin B especially with lysosomes and miltefosin give excellent curative results. Oral miltefosine for 28 days, daily injections of aminosidine for 21 days, infusions of conventional amphotericin B given daily for 20 days or on alternate days for 30 days, and short courses of infusions of a liposome formulation of amphotericin B have cure rates of over 90%. Treatments are not really affordable. US average wholesale prices for the various lipid formulations of amphotericin B are $188 per 50 mg vial for AmBisome (liposomal), $93 per 50 mg for Amphotec (cholesterol dispersion), and $194 per 100 mg for Abelcet (lipid complex). The regimen for liposomal amphotericin B consists of 3 mg/kg given on days 1-5, 14 & 21 (total dose: 21 mg/kg). AmBisome currently costs about $173 per 50 mg. This amounts to roughly $500-2000 per patient. Eradication. The eradication of various tropical and other infectious diseases will be almost commonplace in the first half of this century, yet leishmaniasis may not be one of these. Sandfly vector control in the Old and New Worlds is never mentioned just as research is simply not done with these creatures. Will DDT eradication of malaria vectors also eliminate sandflies? Will Brazil eradicate its leishmaniasis? Many infectious diseases from malaria on have the same geography, which is to say that certain diseases are wellknown in certain countries like India. The tropical parasitoses have to be eradicated in their strongholds like subSahara Africa and India. All of these planned eradications are exceedingly difficult, because disease endemicity correlates to poverty much of which is rural. On a lesser scale, the same thing occurs in South America. However, the poverty is not as severe, and how poverty correlates to disease is not as clear. Are given diseases causing poverty? Chronic leishmaniasis is one of these.