* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download star model
Survey
Document related concepts
Transcript
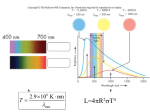
1B11 Foundations of Astronomy Sun (and stellar) Models Silvia Zane, Liz Puchnarewicz [email protected] www.ucl.ac.uk/webct www.mssl.ucl.ac.uk/ 1B11 Physical State of the stellar (Sun) interior Fundamental assumptions: Although stars evolve, their properties change so slowly that at any time it is a good approximation to neglect the rate of change of these properties. Stars are spherical and symmetrical about their centre; all physical quantities depend just on r, the distance from the centre.. 1B11 1) Equation of hydrostatic equilibrium. Concept 1): Stars are self-gravitating bodies in dynamical equilibrium balance of gravity and internal pressure forces. Consider a small volume element at distance r from the centre, cross section S=2r, thickness dr Pr dr Pr S GM r Sdr / r dP GM r 2 dr r 2 0 (1) 1B11 2) Equation of distribution of mass. Consider the same small volume element at distance r from the centre, cross section S=2r, thickness dr M r dr M r dM r / dr dr 4r 2 dr dM r 2 4r dr (2) 1B11 First consequence: upper limit on central P From (1) and (2): dPr / dM r GM r / 4r 2 2 dM r dP / dM / dM P P GM / 4 r r C S r r r MS MS 0 0 At all points within the star r<Rs; hence 1/r4>1/RS4: GM MS 0 r / 4r dM r 2 GM / 4R dM MS r 22 S r GM S2 / 8RS4 0 PC PS GM S2 / 8RS4 GM S2 / 8RS4 For the Sun: Pc>4.5 1013 Nm-2=4.5 108 atm 1B11 Toward the E-balance equation: The virial theorem dP G 3 dP GM r 4r dP GM dr r 4 r Pr 3 r RS , P PS r 0, P PC RS r 2 RS dr r2 RS 3 Pr 4r 2 dr GM r / r 4r 2 dr 0 RS 0 2 2 3 P 4 r dr GM / r 4 r dr r r 0 0 • Thermal energy/unit volume u=nfkT/2=(/mH)fkT/2 • Ratio of specific heats =cP/cV=(f+2)/f (f=3:=5/3) u 1/( 1)kT / mH P / 1 3 1U 0 • U= total thermal Energy; = total gravitational energy 1B11 Toward the E-balance equation: The virial theorem For a fully ionized gas =5/3 and 2U+=0 Total Energy of the star: E=U+ E U / 2 • E is negative and equal to /2 or –U • A decrease in E leads to a decrease in but an increase in U and hence T. • A star, with no hidden energy sources, c.omposed of a perfect gas, contracts and heat up as it radiates energy Stars have a negative “heat capacity” = they heat up when their total energy decreases. 1B11 Toward the E-balance equation Sources of stellar energy: since stars lose energy by radiation, stars supported by thermal pressure require an energy source to avoid collapse. • Energy loss at stellar surface as measured by stellar luminosity is compensated by energy release from nuclear reactions through the stellar interior. RS L r 4r 2 dr 0 r=nuclear energy released per unit mass per s. Depends on T, and chemical composition • During rapid evolutionary phases (contraction/expansion): dL dS TdS/dt accounts for the 4r 2 r T gravitational energy term dr dt 1B11 The equations of Stellar structure Summary: dP GM r dr r2 dM r 4r 2 dr dL dS 4r 2 r T dr dt dT 3kL dr 16acr 2T 3 • P,k,r are functions of ,T, chemical composition (basic physics provides these expressions) • In total: 4, coupled, non-linear partial differential equations (+ 3 constitutive relations) for 7 unknowns: P, ,T, M, L, k, r as a function of r. • These completely determine the structure of a star of given composition, subject to suitable boundary conditions. • in general, only numerical solutions can be obtained (=computer). 1B11 The equations of Stellar structure Using mass as independent variable (better from a theoretical point of view): • Boundary conditions: M=0, L=0 and r=0; M=Ms L=4RS 2Teff4 and P=2/3g/k •These equations must be solved for specified Ms and composition. dP dM dr dM dL dM dT dM GM 4r 4 1 /( 4r 2 ) r T dS dt 3kL /( 64 2 acr 4T 3 ) Uniqueness of solution: the Vogt Russel “theorem”: “For a given chemical composition, only a single equilibrium configuration exists for each mass; thus the internal structure is fixed”. This “theorem” has not been proven and is not rigorously true; there are unknown exceptions (for very special cases) 1B11 Last ingredient: Equation of State Perfect gas: P NkT m H kT • N=number density of particles; =mean particle mass in units of mH. • Define: • X= mass fraction of H (Sun=0.70) • Y= mass fraction of He (Sun =0.28) • Z= mass fraction of heavy elements (metals) (Sun=0.02) • X+Y+Z=1 1B11 Last ingredient: Equation of State If the material is assumed to be fully ionized: ELEMENT Hydrogen NO OF ATOMS X/mH NO OF ELECTRONS X/mH Helium Metals Y/(4mH) Z/(AmH) 2Y/(4mH) (1/2)AZ/(AmH) • A=average atomic weight of heavier elements; each metal atom contributes ~A/2 electrons • Total number of particles: • N=(2X+3Y/4+Z/2) /mH • (1/) = 2X+3Y/4+Z/2) • Very good approximation is “standard” conditions! 1B11 Deviations from a perfect gas The most important situations in which a perfect gas approximation breaks down are: 1) When radiation pressure is important (very massive stars): 4 P kT /( mH ) aT / 3 2) In stellar interiors where electrons becomes degenerate (very compact stars, with extremely high density): here the number density of electrons is limited by the Pauli exclusion principle)