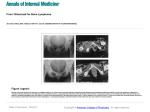
* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download 2.27 R-Fludarabine version1.2 Jul08
Survey
Document related concepts
Transcript
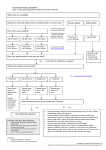
2.27 Protocol Name: Rituximab-Fludarabine Indications Advanced stage indolent and follicular lymphoma (especially useful for diabetic patients in whom the clinician wishes to avoid steroids) Pre-treatment Evaluation 582756765 Document histological sub-type of lymphoproliferative disorder according to WHO Classification. Document FBC (with film), U&E, creatinine, LFTs, calcium, glucose, serum protein electrophoresis, immunoglobulin levels and a direct antiglobulin test (DAT). If staging is relevant this should include documentation of B symptoms, CT of chest, abdomen & pelvis and bone marrow aspirate & trephine. Document WHO performance status of patient. A positive DAT is a relative contra indication to fludarabine therapy Document height, weight and body surface area. Consider ECG ± echocardiogram if clinical suspicion of cardiac dysfunction. Give adequate verbal and written information for patients and relatives concerning patient’s disease, treatment strategy and side effects. Obtain written consent from patient or guardian. If appropriate, discuss the possibility of pregnancy with female patients of child-bearing age and the need for contraception with both male and female patients. If appropriate, discuss potential risk of infertility with patient and relatives. Consider intravenous hydration in patients with bulk disease. Allopurinol should be given for the first 2 cycles of chemotherapy. Significant pleural effusions or ascites should be drained to a minimum prior to commencement of fludarabine. All cellular blood components should be irradiated to prevent the rare occurrence of transfusion associated graft versus host disease. Rituximab has been associated with potentially serious Hepatitis ‘flare’ in patients with chronic hepatitis B infection. All patients should have hepatitis B serology checked before therapy, and in those who have positive serology Page 1 of 4 lamivudine 100mg daily should be commenced 1-2 weeks prior to rituximab. Currently it is recommended that lamivudine is continued for 6 months after rituximab is discontinued. Drug Regimen (OPCS code: X70.4) Days 1 Drug Rituximab Dose 375mg/m² Route IV Comments See below for infusion speed 1–5 Fludarabine 40mg/ m² PO Once daily after breakfast Rituximab infusion speed: First infusion: Initiate at 50mg/h; if tolerated increase rate at 50mg/h increments every 30 minutes to a maximum of 400mg/h. Subsequent infusions: Initiate at 100 mg/h; if tolerated increase rate at 100mg/h increments every 30 minutes to a maximum of 400 mg/h. Fast rate infusion: Many units have implemented this locally as a safe method of reducing the infusion time Patients MUST have tolerated their first cycle of rituximab (at the standard recommended infusion rates as specified above) before being given the rapid infusion rate. All rituximab doses are prepared in 250mls 0.9% sodium chloride, with 50mls being given over the first 30 minutes and the remaining 200mls given over 1 hour. If a 500ml volume of 0.9% sodium chloride is used, then 100mls is given over the first 30 minutes and the remaining 400mls given over 1 hour. Considerations Oral fludarabine is only available as 10mg tablets so round doses up or down to the nearest whole tablet. Cycle Frequency Repeat every 28 days. Dose Modifications Reduce by up to 50% doses if renal impairment (creatinine clearance between 30-60ml/min). Do not give if creatinine clearance <30ml/min. Haematological dose reductions: 582756765 Page 2 of 4 Patients with good prognosis - consider maintaining dose intensity with GSCF. GCSF should be administered in doses sufficient to allow full dose of treatment on schedule. However use of GCSF should be in accordance with the ASCO guidance – therefore only for maintenance of full dose after an episode of febrile neutropenia or prolonged neutropenia that has led to a dose reduction. Neutropenia and thrombocytopenia may be due to disease. If a fall in counts is considered to be due to treatment (and GCSF is not appropriate) then, the following guidelines should be followed. At the time of the next cycle: If neutrophils <1 x 109/l or platelets <75 x 109/l, delay treatment for 1 week. If these values are not improved after a delay of 2 weeks treatment should proceed at 50% of the dose. If neutrophils are below 0.5 x 109/l or platelets below 50 x 109/l by the time a new course is due, delay treatment until counts rise to at least these levels, with dose modifications as above if necessary. Investigations prior to subsequent cycles FBC, U+E, Creat and LFTs. Clinical assessment of response to be documented in notes. Assuming clinical response, restage after cycle 3 and again after cycle 5. Treatment Duration Maximum 6 cycles. Concurrent Medication Allopurinol 300mg od PO (100mg if creatinine clearance <20mls/min) for first 2 cycles. Oral systemic PCP prophylaxis should be given according to local protocol throughout treatment and for six months after completion of chemotherapy. Consider Acyclovir antiviral prophylaxis if previous history of VZV or HSV reactivation. Anti-emetics This regimen has mild emetic potential - refer to local protocol. ** Issue patient with DOH/National Blood Service Irradiated Blood Products information sheet: ** 582756765 Page 3 of 4 References SPC Patient Information http://www.cancerbackup.org.uk/Treatments/Chemotherapy/Individualdrugs/Fludarabi ne http://www.cancerbackup.org.uk/Treatments/Biologicaltherapies/Monoclonalantibodies /Rituximab Written by: Dr Ed Kanfer Authorised by: WLCN Haematology TWG Date for review by Haematology TWG: 582756765 Page 4 of 4