* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download EACA - Blood CME Center
Pharmacokinetics wikipedia , lookup
Discovery and development of direct Xa inhibitors wikipedia , lookup
Clinical trial wikipedia , lookup
Pharmaceutical industry wikipedia , lookup
Pharmacogenomics wikipedia , lookup
Theralizumab wikipedia , lookup
Discovery and development of direct thrombin inhibitors wikipedia , lookup
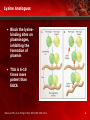
Hemostasis for the Surgeon: Treatment Practices Peter K. Smith, MD Professor and Chief Division of Cardiovascular and Thoracic Surgery Duke University Medical Center Durham, North Carolina Prohemostatic Agents • Antifibrinolytics Lysine analogues Aprotinin • Topical hemostatics • Desmopressin (DDAVP) • Recombinant activated factor VIIa (rVIIa) Levi M. Minerva Anestesiol. 2004;70:267-271. 2 Fibrinolysis Coagulation Cascade Fibrinolytic Cascade Prothrombin Plasminogen – APC TAFIa Thrombin – Plasmin PC TAFI Thrombin Thrombomodulin Fibrinogen Fibrin FDPs Antifibrinolytic Agents Tip the balance against fibrinolysis More clot Less bleeding Adapted from Nesheim M. Chest. 2003;124:33S-39S. 3 Antifibrinolytics • As implied by the name, these agents enhance hemostasis when fibrinolysis contributes to bleeding • Lysine analogues ε-aminocaproic acid (EACA) Tranexamic acid (TXA) • Aprotinin: Approved by FDA to reduce blood loss and transfusion in coronary artery bypass graft surgery (CABG) but marketing suspended 11/5/07 1. Levi M. Minerva Anestesiol. 2004;70:267-271. 2. US Food and Drug Administration. Available at: http://www.fda.gov/CDER/DRUG/infopage/ aprotinin/default.htm. Accessed May 7, 2008. 4 Lysine Analogues • Block the lysinebinding sites on plasminogen, inhibiting the formation of plasmin • TXA is 6-10 times more potent than EACA Mannucci PM, et al. N Engl J Med. 2007;356:2301-2311. 5 Lysine Analogues (cont) • Lysine analogues1-3: EACA and TXA Indicated for enhancing hemostasis when fibrinolysis contributes to bleeding Both competitively inhibit plasmin binding to fibrin Widely used in cardiac surgery, but data supporting safety and efficacy are limited EACA associated with increased incidence of certain neurologic deficits; concerns about rhabdomyolysis and renal dysfunction 1. Mannucci PM, et al. N Engl J Med 2007;356:2301-2311. 2. Levy JH. Am J Health-Syst Pharm. 2005;62(suppl 4):S15-S19. 3. Adams GL, et al. Hematol Oncol Clin North Am. 2007;21:13-24. 6 Lysine Analogues: EACA and TXA • Trial data have limitations1,2 Often only small numbers of patients studied Variable design ?Treatment criteria ?Factor reduction Most data are for TXA, not EACA TXA doses range from 2 g to 25 g Most EACA/TXA studies in lower-risk patients • Meta-analyses need to be cautiously interpreted • EACA removed from many European markets3 ?Safety data 1. Mannucci PM, et al. N Engl J Med 2007;356:2301-2311. 2. Levi M. Minerva Anestesiol. 2004;70:267-271. 3. European Medicines Agency. Available at: http://www.emea.europa.eu/pdfs/human/press/pr/Aprotinin_Q&A.pdf . Accessed May 13, 2008. 7 EACA: Study Findings • A study by Kikura and colleagues evaluated the efficacy of EACA • Study design: Double-blind, placebo-controlled, randomized trial 100 patients randomized to receive EACA (100 mg/kg before incision, 1 g/h infusion until chest closure, 10 g in CPB circuit) or placebo Postoperative thoracic-drainage volume (P=.003) EACA—649±261 mL Placebo—940±626 mL No significant between-group differences in: Need for RBC transfusion (P=.62) • EACA—24% • Placebo—18% Units of donor RBCs transfused (P=.29) • EACA—2.2±0.8 U • Placebo—1.9±0.8 U EACA did not reduce risk of RBC transfusion compared with placebo (OR: 1.2; 95% CI: 0.4 to 3.2; P=.63) • • • • EACA reduced postoperative thoracic-drainage volume by 30% but did not reduce need for allogeneic therapy CPB=cardiopulmonary bypass. Kikura M, et al. J Am Coll Surg. 2006;202:216-222. 8 Blood Conservation Using Antifibrinolytics Trial (BART) • Design: Multicenter, triple-blind RCT Compared aprotinin, TXA, and epsilon-EACA • Population: 2330 high-risk cardiac surgical patients • Intervention: Aprotinin 2 million KIU bolus + 2 million KIU pump prime + 2 million KIU infusion on CPB TXA 30 mg/kg loading dose + 2 mg/kg pump prime + 16 mg/kg/hr on CPB Epsilon-EACA 10 g loading dose + 2 g/hr on CPB Fergusson DA, et al. N Engl J Med. 2008;358:2319-2331; Hebert PC, et al. Available at: http://www.ohri.ca/programs/clinical_epidemiology/thrombosis_group/studies/BART.asp. Accessed May 8, 2008. 9 BART Trial (cont) • Primary outcome Massive post-op bleeding • Secondary outcomes In-hospital death Death from any cause at 30 days Life-threatening or serious adverse clinical events • Trial terminated prematurely Enhanced relative risk (RR) of mortality for aprotinin compared to drug B (RR 1.5; P=.06) and drug C (RR 1.5; P=.08) Fergusson DA, et al. N Engl J Med. 2008;358:2319-2331; Hebert PC, et al. Available at: http://www.ohri.ca/programs/clinical_epidemiology/thrombosis_group/studies/BART.asp. Accessed May 8, 2008; US Food and Drug Administration. Available at http://www.fda.gov/Cder/drug/early_comm/aprotinin.htm. Accessed May 8, 2008. 10 Aprotinin • • A small protein isolated from bovine lung1 A non-specific serine protease inhibitor inhibits trypsin, plasmin, plasma/tissue kallikrein, etc1,2 • Inhibits contact phase activation of coagulation that both initiates coagulation and promotes fibrinolysis2 • In CPB, it reduces derangements in coagulation/fibrinolysis caused by negatively charged surface of CPB circuit2 • Indirectly preserves platelet function in extracorporeal circulation1 • Marketing suspended on 11/5/07 following FDA Advisory 2/8/063 • Available for “on-label” investigational use with IRB approval, see Bayer Web site • BART trial publication 5/084 1. Levi M. Minerva Anestesiol. 2004;70:267-271; 2. Mannucci PM, et al. N Engl J Med. 2007;356:2301-2311; 3. US Food and Drug Administration. Available at: http://www.fda.gov/CDER/DRUG/infopage/aprotinin/default.htm. Accessed May 7, 2008; 4. Fergusson DA, et al. N Engl J Med. 2008;358:2319-2331. 11 Topical Hemostatic Agents • Identified by FDA as “a device intended to produce hemostasis by accelerating the clotting process of blood”1 • Used to augment hemostasis in surgery/trauma • Available in a variety of forms (solutions, gels, granules, sprays) and used in conjunction with collagen, gelatin, cellulose matrices • Local thrombin and fibrinogen levels determine the rate of clot formation at wound site • Classification: Tissue/fibrin sealants (contain thrombin, fibrin, ? aprotinin, etc) Absorbable hemostatic agents (contain matrices) Combination products (contain both groups above) • Efficacy: Few RCTs1 • Safety: Associated with numerous adverse events2 1. Lawson JH, et al. Available at: www. fda.gov/ohrms/dockets/dockets/06n0362/06N0362_ECI-Attach-1.pdf. Accessed February 20, 2008. 2. Gabay M. Am J Health-Syst Pharm. 2006;63:1244-1253. 12 Some Topical Hemostatic Agents Sealants and Combination Products: Agent Topical Application Instructions Major Drawbacks or Comments Bovine thrombin Dry, spray, or mixed with isotonic saline applied to bleeding or oozing surfaces; may also be used with absorbable gelatin sponge or with FloSeal NT Prion disease transmission; autoantibodies may develop to impurities, potentially resulting in coagulopathy Recombinant human thrombin Released in 2008; similar to bovine thrombin Potentially less immunogenic than bovine thrombin FloSeal Hemostatic Matrix: bovine gelatin granules and human thrombin After reconstituting thrombin and mixing with gelatin granules, applied to bleeding wound and conforms to its shape Infectious disease transmission similar to that with other human blood products; bovine sensitization Virally inactivated aprotinin-free fibrin sealant (Crosseal): thrombin and fibrinogen (human) Stored frozen, then thawed and sprayed Contains no animal protein and is virally inactivated and highly purified; safety concerns minimized Reprinted from Voils S. Pharmacotherapy. 2007;27:69S-84S. 13 Some Topical Hemostatic Agents (cont) Agent Topical Application Instructions Major Drawbacks or Comments CoStasis: microfibrillar collagen-fibrin (bovine) Reconstituted mixture forms gel matrix Similar to other bovine preparations CoSeal Surgical Sealant: 2 synthetic polyethylene glycols Reconstituted mixture forms a hydrogel that is applied to bleeding or oozing surfaces; forms mechanical seal Swells up to 4 times its volume; may cause compression of anatomic structures Aprotinin and TXA Solutions containing 1 MU of aprotinin or 2.5 g of TXA in 250 mL of saline poured into pericardial cavity during CPB Single study with minimal effectiveness of aprotinin; TXA was less effective in reducing blood product usage Chitosan hemostatic bandage Bandage that binds electrostatically to red blood cells; considered a device; used in combat Floats off wound in severe hemorrhage Zeolite Powder applied to wounded tissue; considered a device; used in combat Local hyperthermia-induced tissue damage Reprinted from Voils S. Pharmacotherapy. 2007;27:69S-84S. 14 Some Topical Hemostatic Agents (cont) Cellulose-, Collagen-, and Gelatin-Based Topical Hemostatic Composition Approval Date Surgicel (J&J) Regenerated oxidized cellulose October 14, 1960 Gelfoam (Pfizer) Porcine gelatin molded into a sponge Available 1945; approved July 8, 1983 Surgifoam (J&J) Porcine gelatin sponge September 30, 1999 Avitene (Davol) Bovine collagen August 26, 1976 (as a drug) October 24, 1980 (as a device) Instat (J&J/Gateway) Bovine collagen October 10, 1985 Helistat (Integra LifeSciences) Bovine collagen November 8, 1985 Helitene (Integra LifeSciences) Bovine collagen November 8, 1985 Originally published in Gabay M. Am J Health-Syst Pharm. 2006;63:12441253. ©2006, American Society of Health-System Pharmacists, Inc. All rights reserved. Reprinted with permission. (R0821) 15 Indications Hemostatic Agent Labeled Indication(s) Surgicel For use in surgical procedures when conventional methods of hemostasis, such as pressure and ligature, are ineffective; for endoscopic procedures, may be used by cutting to size. Gelfoam For use in surgical procedures, including those that may result in calcellous bone bleeding, when conventional methods of hemostasis are ineffective or impractical. Surgifoam For use in surgical procedures, except urologic and ophthalmic procedures, when conventional methods of hemostasis are ineffective or impractical. Avitene For use in surgical procedures when conventional methods of hemostasis are ineffective or impractical. Instat For use in surgical procedures, except ophthalmic procedures, when conventional methods of hemostasis are ineffective or impractical; for endoscopic procedures, may be used by cutting to size. Helistat For use in surgical procedures, except urologic and ophthalmic procedures, when conventional methods of hemostasis are ineffective or impractical. Originally published in Gabay M. Am J Health-Syst Pharm. 2006;63:12441253. ©2006, American Society of Health-System Pharmacists, Inc. All rights reserved. Reprinted with permission. (R0821) 16 Indications (cont) Hemostatic Agent Labeled Indication(s) Helitene For use in surgical procedures, except urologic and ophthalmic procedures, when conventional methods of hemostasis are ineffective or impractical. CoStasis For use in surgical procedures, except neurologic, ophthalmic, and urologic procedures, when conventional methods of hemostasis are ineffective or impractical. FloSeal For use in surgical procedures, except ophthalmic procedures, when conventional methods of hemostasis are ineffective or impractical. Thrombin-JMI For use as an aid to hemostasis whenever oozing blood and minor bleeding from capillaries and small venules are accessible; may be used in combination with absorbable gelatin sponge for hemostasis; may be used in conjunction with any other device that has been approved by FDA with a specified dosage of topical thrombin. Originally published in Gabay M. Am J Health-Syst Pharm. 2006;63:12441253. ©2006, American Society of Health-System Pharmacists, Inc. All rights reserved. Reprinted with permission. (R0821) 17 Considerations • Efficacy: Few RCTsstudies have shown beneficial effects in controlling capillary bleeding, achieving hemostasis in vascular surgery, controlling bleeding from fistulapuncture site in hemodialysis, etc • Cost: No published study of cost-effectiveness • Safety Gabay M. Am J Health-Syst Pharm. 2006;63:1244-1253. 18 Adverse Events • • • • • • • • • • • • • • • • Device failure (continued bleeding observed) Device deployment failure Infection Granuloma Abscess Foreign body reaction Allergic reaction Interference with wound healing Respiratory difficulty Bowel obstruction Hematoma Intermittent ischemia Stroke Tissue necrosis Erythema Edema US Food and Drug Administration. Available at: www.fda.gov/ohrms/dockets/ac/02/briefing/3876b1_06Absorbable%20Hemostatic%20Agent%20Reclass%20Memo.doc. Accessed May 9, 2008. 19 Adverse Events (cont) • In 2004, FDA issued a notification on possible development of paralysis following use of absorbable hemostatic agents • If agent used and left on or near a bony or neural space, when wetted, the material swelled and exerted pressure on neural structures, resulting in pain, numbness, or paralysis • Recommendations: Read labels carefully If used on or near bony/neural spaces, use the minimum amount necessary to achieve hemostasis and remove as much of the agent as possible after hemostasis is achieved US Food and Drug Administration. Available at: http://www.fda.gov/cdrh/safety/040204-hemostatics.html. Accessed May 9, 2008. 20 Desmopressin • Originally developed and licensed for the treatment of inherited defects of hemostasis1,2 • Several reviews suggest its effect is too small to influence the need for transfusion and reoperation1,2 • Most evidence of efficacy is in mild hemophilia A and von Willebrand’s disease1,2 • Not indicated for use in cardiac surgery patients1,2 Meta-analysis in cardiac patients: 2-fold increase in MI, a small decrease in perioperative blood loss, and no added benefits on clinical outcomes 1. Mannucci PM, et al. N Engl J Med. 2007;356:2301-2311. 2. Levy JH. Am J Health-Syst Pharm. 2005; 62(suppl 4):S15-S19. 21 Desmopressin and Surgical Bleeding in Patients With Inherited Coagulation Disorders • • Study data limitations1-3 Small numbers of patients Mostly retrospective analyses Multimodal approaches Antifibrinolytics used concomitantly with other factor concentrates2 • • Bleeding depends on types of surgical procedure1 Superficial vs major Vascular, cardiac, neurosurgical Monitoring of effects is limited—especially with platelet function tests1 1. Mannucci PM, et al. N Engl J Med. 2007;356:2301-2311. 2. Berntorp E. Haemophilia. 2006;12:62-66. 3. Levy JH. Am J Health-Syst Pharm. 2005; 62(suppl 4):S15-S19. 22 Recombinant Factor VIIa • Vitamin K-dependent glycoprotein structurally similar to human plasma-derived FVIIa1 • Approved in United States for treatment of bleeding in patients with hemophilia A or B with inhibitors to FVIII or FIX1 • Multiple reports of off-label use in cardiac surgery, trauma, liver transplantation to secure hemostasis2 • Promotes hemostasis by activating the coagulation cascade1 • Believed to cause local thrombin generation and platelet recruitment at sites of vascular and microvascular injury2 1. Levy JH. Am J Health-Syst Pharm. 2005; 62(suppl 4):S15-S19. 2. Mannucci PM, et al. N Engl J Med. 2007;356:2301-2311. 23 Recombinant Factor VIIa (cont) • A central factor in coagulation1 • A trypsin-like serine protease (characterized by a serine residue in the active side of the enzyme)2 • Initiates coagulation in a complex with TF2 • Once bound to TF, it is activated (rVIIa) by different proteases2 • Produced in liver3 TF=tissue factor. 1. Levy JH. Am J Health-Syst Pharm. 2005; 62(suppl 4):S15-S19. 2. Lawson JH, et al. Semin Hematol. 2004;41(suppl 1):55-64. 3. Levy JH. Transfusion. 2006; 46:919-933. 24 Mechanism of Action Remember the mechanism… • FIXa and FVIIIa aid FVIIa in activating X • If FIXa or FVIIIa is missing (or inhibited), rFVIIa can replace their function by converting more FX to FXa Mannucci PM, et al. N Engl J Med. 2007;356:2301-2311. 25 Thrombotic Adverse Events • From March 1999 to December 2004, 431 adverse events (AEs) were reported to the FDA 185 (43%) were thrombotic AEs • In 36 (72%) of 50 reported deaths, thrombotic AE was probable cause • 73 TBEs (52%) occurred within 24 hours of rVIIa therapy • Concomitant use of other hemostatic agents was noted in 64 cases (38%) O’Connell KA, et al. JAMA. 2006;295:293-298. 26 Thrombotic Adverse Events (cont) • Critical safety data obtained from 13 pharmaceutical-sponsored clinical trials of rVIIa Patients with coagulopathy due to: • Anticoagulant therapy • Cirrhosis • Severe traumatic injury • • Thrombotic AEs reported Placebo: 5.3% (23/430) Active treatment: 6.0% (45/748) No significant differences between placebo-treated and rVIIatreated patients for thrombotic AEs—either on individual trial basis or for combined trial populations (P=.57) Levy JH, et al. Transfusion. 2006;46:919-933. 27 “Off-label” Uses of rVIIa • Increasingly being considered for: Reversal of oral anticoagulation Reversal of heparin, lepirudin, and fondaparinux Thrombocytopenia and thrombocytopathy Bleeding with impaired liver function Gastrointestinal bleeding Trauma Surgery: Non-traumarelated (hepatic resection, prostatectomy, cardiac, spinal) • These off-label uses are mostly based on anecdotal case reports Need better evidence Franchini M. Thromb Haemost. 2005;93:1027-1035. 28 Comparison of Available Randomized Patient Data: Cardiac Surgery 4500 Enrolled On-label 4000 Number of Patients 3500 3000 2500 2000 1500 1000 500 0 Aprotinin BART Factor VIIa Silver DA, et al. Crit Care Med. 2007;35:1782-1783. 29 Current Ongoing Global Trials Phase Total Enrollment Expected Completion Phase 3 40 June 2008 ?? ?? December 2010 Randomized, DoubleBlind (Subject, Caregiver, Investigator) PlaceboControl, Parallel Assignment, Efficacy Study Phase 3 52 December 2009 Randomized, Controlled, OpenLabel, InvestigatorBlinded Pilot Study Phase 2 30 January 2008 Trial Title Study Design "Salvage Use" of Recombinant Activated Factor VII After Inadequate Haemostatic Response to Conventional Therapy in Complex Cardiac Surgery Multicenter, Treatment, Randomized, DoubleBlind, PlaceboControl, Parallel Assignment, Safety/Efficacy Study Evaluation of the Quality of the NovoSeven (rVIIa) Treatment Practice at Rigs hospital, Copenhagen University Hospital Natural History, Longitudinal, Defined Population, Retrospective/ Prospective Study Effect of Recombinant Coagulation Factor VIIa on PeriOperative Blood Loss in Patients Undergoing Major Burn Excision and Grafting Efficacy and Safety of Factor VIIa (Eptacog Alfa) on Rebleeding After Surgery for Spontaneous Supratentorial Intracerebral Hemorrhage US National Institutes of Health. ClinicalTrials.gov. Available at: www.clinicaltrials.gov. Accessed February 10, 2008. 30 Current Ongoing Global Trials (cont) Phase Total Enrollment Multicenter, Treatment, Randomized, DoubleBlind, PlaceboControl, Parallel Assignment, Safety/Efficacy Study Phase 3 40 June 2008 Effect of Recombinant Coagulation Factor VIIa on PeriOperative Blood Loss in Patients Undergoing Major Burn Excision and Grafting Randomized, DoubleBlind (Subject, Caregiver, Investigator) PlaceboControl, Parallel Assignment, Efficacy Study Phase 3 52 December 2009 Efficacy and Safety of Factor VIIa on Rebleeding After Surgery for Spontaneous Intracerebral Hemorrhage (ICH) (PRE-SICH) Randomized, Controlled, OpenLabel, InvestigatorBlinded Pilot Study Phase 2 30 January 2008 Trial Title Study Design "Salvage Use" of Recombinant Activated Factor VII After Inadequate Haemostatic Response to Conventional Therapy in Complex Cardiac Surgery US National Institutes of Health. ClinicalTrials.gov. Available at: www.clinicaltrials.gov. Accessed February 10, 2008. Expected Completion 31 Current Ongoing Global Trials (cont) Trial Title Study Design Recombinant Human Activated Factor VII as Salvage Therapy in Women With Severe Postpartum Hemorrhage Randomized, OpenLabel, Active Control, Parallel Assignment, Safety/Efficacy Study The Use of rVIIa in Trauma Patients: A Multi-Center Case Registry Natural History, Cross-Sectional, Case Control, Retrospective/ Prospective Study Assessment of rVIIa in Controlling Bleeding in Patients With Severe Trauma Injuries Treatment, Randomized, DoubleBlind, Placebo Control, Parallel Assignment, Safety/Efficacy Study US National Institutes of Health. ClinicalTrials.gov. Available at: www.clinicaltrials.gov. Accessed February 10, 2008. Phase Total Enrollment Expected Completion Phase 4 84 December 2009 ?? 1000 Not recruiting yet Phase 3 1502 Recruiting 32 The Role of Recombinant Factor VIIa in On-pump Cardiac Surgery: Proceedings of the Canadian Consensus Conference • Panel recommendation against prophylactic or routine use of rVIIa • Weak recommendation for use of rVIIa as rescue therapy for blood loss refractory to standard hemostasis Tx 1-2 doses of 35-70 µg/kg Karkouti K, et al. Can J Anesth. 2007;54:573-582. 33 Final Thoughts • Prohemostatic agents fall into multiple categories • Significant surgical bleeding with fibrinolysis can be controlled using antifibrinolytic agents • Topical hemostatic agents play an important role in the management of significant surgical bleeding • Recombinant factor VIIa is an intriguing, potential new agent, the role of which will be better defined for different types of surgery when results of multiple RCTs are published in near future 34