* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Chem 110 2014 - University of KwaZulu
Survey
Document related concepts
Chemical element wikipedia , lookup
California Green Chemistry Initiative wikipedia , lookup
Drug discovery wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Institute of Chemistry Ceylon wikipedia , lookup
Nuclear chemistry wikipedia , lookup
Chemistry: A Volatile History wikipedia , lookup
History of molecular theory wikipedia , lookup
Computational chemistry wikipedia , lookup
Analytical chemistry wikipedia , lookup
Green chemistry wikipedia , lookup
Inorganic chemistry wikipedia , lookup
History of chemistry wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Transcript
CHEM 110
QUANTITATIVE CHEMISTRY
Chapter One
Introduction to Matter and Measurement
Dr V Paideya
2014
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
• Intranet
http://cheminnerweb.ukzn.ac.za/Firstyear/
oneten.aspx
• Mastering Chemistry
https://secure.ecollege.com/ukznmlp/inde
x.learn?action=welcome
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
School of Chemistry,
University of KwaZulu-Natal, Westville Campus, Durban
CHEM110: General Principles of Chemistry
Worksheet 1
Matter, Measurements and Molecules
AIM:
• To provide a background to understanding the properties of
matter in terms of atoms, molecules and ions including scientific
measurements
CONTENT: Units, significant figures and scientific notation, basic
nomenclature, atoms and molecules, elements and compounds,
atomic structure and isotopes
LEARNING OBJECTIVES - You should be able to:
• Distinguish between elements, compounds and mixtures
• Recognise symbols of common elements and common prefixes for
units
• Use significant figures, scientific notation and SI units.
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
CHEMISTRY
“study of matter & changes it undergoes”
“matter is anything that has mass and takes up space”
- study of physical & chemical properties of matter
- what changes occur in these properties, in the course of/as the result of a
chemical reaction, & how these changes may be observed
- why the reaction involved does (or doesn’t…) occur
be able to understand & explain such
macroscopic changes from an atomic/molecular
(submicroscopic) perspective
States (Phases) of Matter
- solid, H2O(s); liquid, H2O(l); gas, H2O(g)
- phase transitions occur @ specific P/T values,
governed by properties of atoms/molecules
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Matter
- atoms are building blocks of matter
- each element is made of same kind of atom/molecules*
- compounds made of two or more different kinds of elements bonded together
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Pure Substances, Elements & Compounds
pure substance
-has distinct properties & unvarying/constant composition
eg. NaCl(s), H2O(l), HCN(g)
element
-substance that cannot be decomposed into simpler substances
eg. Cl2(g), Br2(l), I2(s); Ne(g), Hg(l), Au(s)
compound
-substance composed of 2 or more different elements
2 or more different kinds of atoms
eg. UF6(g), H2O(l), CaCO3(s)
Law of Constant Composition/Definite Proportions (Joseph Proust ca 1800)
“...elemental composition of pure substance is always the same…”
- different samples of pure compound have the same elemental composition
- elements present in such samples have same proportion by mass
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
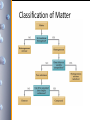
Classification of Matter
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Mixtures
- combination of 2 or more substances, in which each substance retains own
chemical identity & can thus be separated from each other
- 2 types:
heterogeneous:
- mixture of visibly different composition, properties or appearance
eg. sand in H2O(l) (s, l), sand & NaCl (s, s), petrol & H2O(l) (l, l)
homogeneous:
- mixture of visibly uniform composition, properties & appearance throughout
eg. NaCl(aq) (s,l), air (g,g), stainless steel (s,s), soda water (g,l)
Properties of Matter:
- physical: measurement without changing identity/composition eg. Change in
state, temperature, volume
- chemical: must involve change in chemical identity eg. Combustion, oxidation
- extensive: dependent on quantity of sample involved eg. mass, volume
- intensive: independent of quantity eg. , colour, m.p.; useful for identification
of substances
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
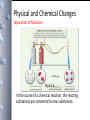
Physical and Chemical Changes
Separation of Mixtures
Figure 1.6
In the course of a chemical reaction, the reacting
substances are converted to new substances.
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Separation of Mixtures
1. Distillation
Separates a
homogeneous
mixture on the
basis of differences
in boiling point.
Figure 1.8
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Separation of Mixtures
2. Filtration
Separates solid substances from liquids and solutions.
3.
Chromatography
Separates substances on the basis of differences in solubility
in a solvent.
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
The Scientific Method
A systematic approach to solving problems
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
SI Units
Système International d’Unités
Uses a different base unit for each quantity
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Metric System
Prefixes convert the base units into units that are
appropriate for the item being measured.
Tera T 1012
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
SI Units - Temperature
• The Kelvin is the SI unit of
temperature.
• It is based on the
properties of gases.
• There are no negative
Kelvin temperatures.
• K = C + 273.15
Figure 1.10
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Derived SI Units Volume
• The most commonly used metric
units for volume are the litre (L)
and the millilitre (mL).
– A litre is a cube 1 dm long on
each side.
– A millilitre is a cube 1 cm long
on each side.
1dm3 = (1 dm) x (1 dm) x (1 dm)
= 10 cm x 10 cm x 10 cm
= 1000 cm3 = 1 L
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Derived SI Units Density
Density is a physical property of a substance and
is determined through the following formula:
mass
density = volume
or symbolically
m
= V
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Common Volumetric glassware
Different measuring devices have different uses
and different degrees of accuracy.
Figure 1.12
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Uncertainty in Measurement
Precision and Accuracy
• Accuracy refers to the proximity of a measurement to
the true value of a quantity.
• Precision refers to the proximity of several
measurements to each other.
Figure 1.15
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Significant Figures
• All digits of a measured quantity,
including the uncertain, are called
significant figures.
• The greater the number of significant
figures, the greater the certainty of the
measurement.
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Significant Figures
To determine s.f in a measurement read no. from left to right, counting
the digits starting with first digit that is not zero
All nonzero digits are significant, e.g. 123.45
Zeros between two significant figures are themselves significant,
e.g. 103.405
Zeros at the beginning of a number are never significant,
e.g. 00123.45 = 123.45
Zeros at the end of a number are significant if a decimal point is written
in the number, e.g. 123.450 has six significant figures but If no. ends in
zero but contains no decimal can be problem – exponential notation
used to indicate if zeros at the end are significant, e.g. 1.03 x 104 g
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Handling Significant Figures in Calculations
Addition and Subtraction
The answer has the least no. of digits to
the right of the decimal pt .in comparison
to the original nos. e.g.(a) 89.332 + 1.1
= 90.432 = 90.4
e.g. (b) 2.097 – 0.12 = 1.977 = 1.98
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Rounding off Procedure
1.
Drop off the digit that follows if it is less
than 5
e.g. 8.724 -- 8.72
2.
Add 1 to the preceding digit if it is equal
or greater than 5
e.g. 8.727 -- 8.73
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Multiplication and Division
The number of significant figures in the
final product or quotient is determined by
the original number that has the smallest
no. of significant figures
e.g. 1. 2.8 x 4.5039 = 12.61092 = 13
e.g. 2. 6.85 ÷ 112.04 = 0.0611388789
= 0.0611
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Infinite number of significant figures
Exact numbers by definition or counting nos. of
objects can be considered to have an infinite no. of
significant figures.
If an object has a mass of 0.2786, then the mass
of 8 such objects
= 0.2786 x 8 = 2.229 g
NB. We don’t round off this product to one significant
fig. because 8 is 8. 0000 .....
Similarly the average of two measured lengths
6.64 cm and 6.68cm = (6.64 + 6.68) ÷ 2 = 6.66 cm
Bec. 2 is 2.0000 .......
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Significant Figures
1. any figure that is not zero is significant:
845 mL _____ s.f.1243.29 mg _____ s.f.
2. zeroes between non-zero figures are significant:
1906 mL _____ s.f.40501.09 J _____ s.f.
3. exact (“counting”) numbers by definition have an number of s.f., so physical
constants defined to be exact numbers do so also...:1 atm 101.325 kPa 760
mmHg;
0 OC 32 OF 273.15 K
all _____ s.f.
4. leading zeroes (to the left of the first non-zero figure) are not significant:
0.008 kg _____ s.f.
0.003798 L _____ s.f.
5. trailing zeroes (to the right of the last non-zero figure) are significant only if the
number has a d.p.: 300.0 mm _____ s.f.0.0300 mm _____ s.f.
6. in measurements without a d.p., the number of s.f. is ambiguous:
1200mm ??
OR
either:
i)
use scientific notation
ii)
1200.0
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Using Significant Figures in Calculations
all calculations governed by two fundamental rules
multiplication/division
number of s.f. in final answer is the LEAST of numbers of s.f. in each of
original measurements
addition/subtraction
number of d.p. in final answer is the LEAST of numbers of d.p. in each of
original measurements
Eg. 1
Calculate
i)
volume, in mm3, of a box of length 6.741 cm, breadth 2.441 x 10-1 m, & height
4.2 mm
i) 6.9 x 104 mm3;
ii)
density () of a pure liquid, in g cm-3, if 103.67 g of it is needed to fill the box
completely
ii) 1.5 g cm-3
Eg. 2
An empty container of mass 23.29 g has a mass of 86.1 g when filled with 0.5000
dm3 of a pure liquid. Determine the of this liquid in g cm-3.
0.126 g cm-3
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
CHAPTER TWO
ATOMS, MOLECULES AND IONS
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Early Atomic Theory
John Dalton 1803 - 1807
-each element is composed of very small, indestructible, particles called atoms*
-all atoms* of given element are physically & chemically identical to each other,
but atoms of a particular element are different from atoms of all other elements
Law of Conservation of Mass
-atoms are neither created or destroyed in chemical reactions
- mass reactants present @ start = mass products formed @ completion*
Law of Constant Composition
-different samples of a pure compound have the same elemental composition
-elements present in such samples have same proportion by mass at the end of a
chemical process as before the process took place.
Law of Multiple Proportions
-if 2 elements (C & O) can combine to form 2 or more different compounds (CO &
CO2), the different masses of one element (O) combining with a fixed mass of the
other (C) can be expressed as a simple integral ratio
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Atomic Theory
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
The Law of Multiple Proportions
• Was deduced by Dalton from the preceding four
postulates and states that:
• If two elements A and B combine to form more than
one compound, the masses of B that can combine
with a given mass of A are in the ratio of small whole
numbers.
Examples
• H2O consists of 2 hydrogens and 1 oxygen
• H2O2 consists of 1 hydrogen and 1 oxygen
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
The Discovery of Atomic Structure
The Electron (JJ Thomson, 1897)
- electrical discharges from cathode originally thought to be new form of radiation
- showed that radiation emitted was
- independent of cathode material used
- deflected by magnetic/electric fields
- findings consistent with model in which “beam” /”rays” composed of negatively
charged “particles”(-) with charge/mass ratio of 1.78 x 108 C g-1
Electron Charge & Mass (Robert Millikan, 1909)
- oil drop experiment
- (-) charge on oil drops found always to be a multiple of
minimum value of 1.6(02) x 10-19 C ie. 1.602 x 10-19 C must
be charge of single electron (e-)
-mass of single e- determined to be 9.109 x 10-28 g
- only 1/1836 of mass of an H atom
- first subatomic particle
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Radioactivity
The spontaneous emission of radiation by
an atom was first observed by Henri
Becquerel. It was also studied by Marie and
Pierre Curie.
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Radioactivity
Three types of radiation were discovered by Ernest
Rutherford
– particles
– particles
– rays
Figure 1.21
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Discovery of the Nucleus
Ernest Rutherford shot particles at a thin sheet of gold foil and observed
the pattern of scatter of the particles
.
The Nuclear Atom
Some particles were deflected at large angles.
This led Rutherford to postulate that the
atom had a nucleus with positively charged
Particles called protons.Protons
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Modern Atomic Structure
- more than 99.99 % of atom mass & entire Q+ centred in atomic nucleus,
where nucleons (protons, p+ (Rutherford, 1919) & neutrons, nO (Chadwick,
1932) are collectively bound together by strong nuclear force
- atomic nucleus surrounded by much larger atomic volume, containing as
many e- as p+, so atom is electrically neutral & held together by force of
Coulombic/ electrostatic attraction
-
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Atomic (Z) & Mass (A) Numbers
- atoms of different elements have different numbers of p+ in their nuclei
mass number (A)
number of p+ & nO
Z
AE
element symbol
atomic number (Z)
number of p +
(number of e - in neutral atom)
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Isotopes
Atoms with identical atomic numbers (Z)
but different mass numbers (A), or atoms
with the same number of protons which
differ only in the number of neutrons are
called isotopes.
Examples:
11
6C
12
6C
carbon-12
isotope
13
6C
14
6C
carbon-14
isotope
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Isotopes
- atoms of same element having d different numbers of nO in their nuclei
ie. same Z, different A, or same Z, different N
- chemical properties largely similar, but physical properties, & particularly the ones
involving radioactive “nuclei”, can be very different
- each Mg atom is one of three naturally occurring isotopes
25Mg;
26Mg
- 24Mg;
Mg-24
Mg-25;
Mg-26
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Complete the following table:
Element
Eg. 3 CompleteSymbol
the table below:
name
p+ (Z)
79
No
e-
A
118
138
Ba
143
Pb
92
126
35
Krypton
46
36
Experimentally..
High Resolution Mass Spectrometry (p. 39) used for very precise (4-6 d.p.; 7-10 s.f. in total)
measurements of the masses of an element’s isotopes & their naturally occurring abundances
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
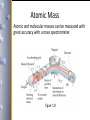
Atomic Mass
Atomic and molecular masses can be measured with
great accuracy with a mass spectrometer.
Figure 1.23
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Average Atomic Mass
(commonly called Atomic Mass)
• We use average masses in calculations, because we use large
amounts of atoms and molecules in the real world.
• Average atomic mass is calculated from the fractional abundance
of each isotope and mass of that isotope.
For example, the average atomic mass of C made up mostly of 12C (98.93%) and 13C (1.07%) - is 12.01 u.
• extremely small SI masses of individual atoms (~4 x 10-22 g) too awkward
for everyday usage, so masses expressed in unified atomic mass units
(amu, u):
1 amu (u) = 1.66054 x 10-24 g
1 g = 6.02214 x 1023 amu (u)
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Average Atomic Masses of Naturally Occurring Elements
-use average masses in “real world” calculations, as even smallest weighable sample
(~1 g 10-6 g) involves large (~1015, or 10 quadrillion) numbers of atoms
-no AAMs calculated as “weighted average” of an element’s isotopic masses (IMs) &
naturally occurring abundances
AAM
= (IM x % ab/100)
or
(IM x fr ab)
Eg. 4
a)Naturally occurring Mg has three isotopes:
Calculate its AAM.
b) Naturally occurring Pb has four isotopes:
Calculate its AAM.
24Mg
78.99 %, 23.9850 u
25Mg 10.00 %, 24.9858 u
26Mg 11.10 %, 25.9826 u
204Pb
1.40 %, 203.973037 u
206Pb 24.10 %,205.974455 u
207Pb 22.10 %, 206.975885 u
208Pb 52.40 %, 207.976641 u
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Eg. 4
a)
Naturally occurring Mg has three isotopes:
Calculate its AM.
24Mg
25Mg
26Mg
78.90 %, 23.9850 u
10.00 %, 24.9858 u
11.10 %, 25.9826 u
AM = (IM x % ab/100)
= {[23.9850 x (78.90/100)] + [24.9858 x (10.00/100)] + [25.9826 x (11.10/100)] }
= 18.924 + 2.4986 + 2.8841
= 24.306 u
b)
Naturally occurring Pb has four:
204Pb 1.40 %, 203.973037 u
206Pb 24.10 %, 205.974455 u
207Pb 22.10 %, 206.975885 u
208Pb 52.40 %, 207.976641 u
Calculate its AM.
AM
= (IM x % ab/100)
= 2.856 + 49.640 + 45.742 + 108.98
= 207.22 u
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Eg. 5
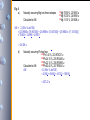
Chlorine has two naturally occurring stable isotopes: 35Cl
37Cl
34.968853 u
36.965803 u
If the (average) atomic mass of naturally occurring elemental Cl is 35.453 u, what are
the % abundances of the two isotopes?
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
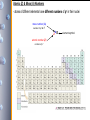
The Periodic Table
- rapid post-Dalton growth in experiment-based chemical
knowledge showed very quickly that many elements could be
grouped together on basis of similarities in their physical &
chemical properties
-
arrangement of elements in order of Z showed that these
similarities occurred in repetitive/periodic patterns, & agreed
so closely with experimentally acquired data, that phys/chem
properties of 2 “missing” elements were accurately predicted
before their being reported as formally discovered, &/or
phys/chem properties characterized
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
The Periodic Table
When one looks at the chemical properties of elements, one notices a repeating
pattern of reactivities.
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Periodic Table
• The rows are called periods.
• The columns are called groups.
• Elements in the same group have similar chemical properties.
• Nonmetals are on the right side of the periodic table (with the
exception of H).
• Metalloids border the stair-step line (with the exception of Al
and Po).
• Metals are on the left side of the chart.
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Groups
Table 1.7
The above five groups are
known by their names.
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
The Periodic Table
Metals, Non-Metals, & Metalloids
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Molecules and Chemical Formulae
The subscript to the right of the
symbol of an element tells the
number of atoms of that
element in one molecule of the
compound.
Notice how the composition of
each compound is given by its
chemical formula.
Figure 1.29
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Diatomic Molecules
Figure 1.28
These seven elements occur naturally as
molecules containing two atoms.
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Molecular Compounds
Molecular compounds are composed of molecules and almost always
contain only nonmetals.
Types of Formulae
Empirical formulae give the lowest whole-number ratio of atoms of
each element in a compound, e.g. HO.
Molecular formulae give the exact number of atoms of each element
in a compound, e.g. H2O2.
Structural formulae show which atoms are attached to which within
the molecule, e.g. H-O-O-H.
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Picturing Molecules
Different
representations of
the methane (CH4)
molecule.
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Ions and Ionic Compounds
•
When atoms lose or gain electrons, they become ions.
–
Cations are positive and are formed by elements on the left side of the
periodic chart.
–
Anions are negative and are formed by elements on the right side of
the periodic chart.
Anion formation
+ ee.g. Cl atom ----------------------> Cl ion (anion)
17 protons
17 protons
17 electrons
18 electrons
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Ionic Compounds
Ionic compounds (such as NaCl) are generally formed
between metals and nonmetals.
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Using Ionic charge to write empirical
Formulae
• Because compounds are electrically neutral, one can determine
the formula of a compound by:
– writing the value of the charge on the cation as the
subscript on the anion.
– writing the value of the charge on the anion as the
subscript on the cation.
Note: if the subscripts are not in the lowest whole number
ratio, simplify it, e.g. Ca2O2 would become CaO.
Ex. What is the empirical formula of the compound formed by
(a) Al3+ and S2- and (b) Zn2+ and PO42Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Chemical Nomenclature
Positive Ions (Cations)
a) Cations formed from metal atoms have the same
name as the metal, e.g. Na+ is the sodium ion.
b) If a metal can form different cations, the positive
charge is indicated by a Roman numeral in
parentheses following the name of the metal, e.g.
Au+ is the gold(I) ion and Au3+ is the gold(III) ion.
c) Cations formed from nonmetal atoms have names
that end in -ium, e.g. NH4+ is the ammonium ion.
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Chemical Nomenclature
Common Cations
Table 2.4
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Chemical Nomenclature
Negative Ions (Anions)
a) The names of the monatomic anions are formed by
replacing the ending of the name of the element with ide, e.g. O2- is the oxide ion.
b) Polyatomic anions containing oxygen (called oxyanions)
have names ending in -ate or -ite, e.g. SO42- is the
sulfate ion and SO32- is the sulfite ion.
c) Anions derived by adding H+ to an oxyanion are named
by adding the prefix hydrogen or dihydrogen, e.g. HCO3is the hydrogen carbonate ion.
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Chemical Nomenclature
Common Anions
Table 2.5
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Chemical Nomenclature
More on naming oxyanions
Examples:
ClO4perchlorate ion (one more O atom than chlorate)
ClO3
chlorate
ClO2
chlorite ion (one less O atom than chlorate)
ClO
hypochlorite ion (one O atom less than chlorite)
Names of ionic compounds consist of the cation followed by the
anion name, e.g. Cu(ClO4)2 is copper(II) perchlorate, and CaCO3
is calcium carbonate.
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Chemical Nomenclature
Name and Formulae of Acids
1. Acids containing anions whose names end in -ide are named by
changing the -ide ending to -ic, adding the prefix hydro- to this anion
name, and then following with the word acid.
2. Acids containing anions whose names end in -ate or -ite are named
by changing the -ate ending to -ic and the -ite ending to -ous and
then adding the word acid.
Figure 2.22
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Chemical Nomenclature
Binary Molecular Compounds
1.
The name of the element farther to the left in the
periodic table is written first.
2.
If both elements are in the same group in the
periodic table, the one having the higher atomic
number is written first.
3.
The name of the second element is given an -ide
ending.
4.
Greek prefixes are used to indicate the number of
atoms of each element.
Examples
N2O4 is dinitrogen tetroxide
P4S10 is tetraphosphorus decasulfide.
Table 2.6
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Derivatives of Alkanes
Alcohol
An alcohol is an example of an alkane that has
some hydrocarbon groups replaced with an –OH
group.
Methanol (CH3OH)
Ethanol (C2H5OH)
Propanol (C3H7OH)
Octanol (C8H17OH)
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
Naming of simple Organic Compounds
Alkanes
Compounds contain only carbon and hydrogen and are
called hydrocarbons.
Simplest class of hydrocarbons are alkanes- names of
compounds end in -ane
Methane (CH4)
Ethane (C2H6)
Propane (C3H8)
Octane (C8H18)
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia
EXERCISE
• Name the following ionic compounds:
(a) MgO
(b) AlCl3
(c) (NH4)2SO4
• Write Chemical Formulae for the following
compounds:
(a) Copper(I) oxide
(b) Sodium hydoxide
(c) Zinc sulphate
Brown, LeMay, Bursten, Murphy, Langford, Sagatys: Chemistry 2e © 2010 Pearson Australia