* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Tuberculosis in the Setting of HIV Infection
Survey
Document related concepts
Hepatitis B wikipedia , lookup
Dirofilaria immitis wikipedia , lookup
Neonatal infection wikipedia , lookup
Hepatitis C wikipedia , lookup
Hospital-acquired infection wikipedia , lookup
Oesophagostomum wikipedia , lookup
Sexually transmitted infection wikipedia , lookup
Mycobacterium tuberculosis wikipedia , lookup
Diagnosis of HIV/AIDS wikipedia , lookup
Epidemiology of HIV/AIDS wikipedia , lookup
Microbicides for sexually transmitted diseases wikipedia , lookup
Transcript
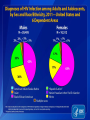
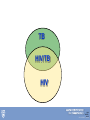
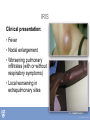
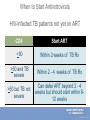
Tuberculosis in the Setting of HIV Infection Zelalem Temesgen, M.D. Tuberculosis Clinical Intensive Sept 9-11,2013 ©2013 MFMER | slide-1 Disclosures • Unrestricted educational grants • Gilead, Janssen, ViiV, Merck, CDC • Research grants • Gilead, Pfizer, NIH ©2013 MFMER | slide-2 Learning Objectives • Describe the epidemiology of TB/HIV co-infection • Describe the impact of HIV on the natural history of TB • Describe current guidelines for the diagnosis and treatment of LTBI in the setting of HIV infection • Explain treatment and management strategies for TB patients with HIV. ©2013 MFMER | slide-3 Epidemiology ©2013 MFMER | slide-4 Tuberculosis: 2011 Global Burden of Disease • 12 million prevalent cases • 1.4 million people died (10-13 million) from TB in 2011 • 1 million deaths among • 8.7 million incident cases HIV-negative (8.3-9 million) individuals • 125 cases per 100 000 • 430 000 among people population who were HIV-positive. • Asia (59%) • Africa (26%) • Eastern Mediterranean Region (7.7%) • Europe (4.3%) • Americas (3%) ©2013 MFMER | slide-5 ©2013 MFMER | slide-6 Tuberculosis: High-Burden Countries • Afghanistan • Myanmar • Bangladesh • Nigeria • Brazil • Pakistan • Cambodia • Philippines • China • Russian Federation • DR Congo • South Africa • Ethiopia • Thailand • India • Uganda • Indonesia • UR Tanzania • Kenya • Viet Nam • Mozambique • Zimbabwe ©2013 MFMER | slide-7 United States: Reported TB Cases United States, 1982–2012 30,000 2012 Data 9,951 Cases Rate 3.2/100,000 No. of Cases 25,000 20,000 15,000 10,000 5,000 0 Year Data are updated as of 2/22/13 and are provisional ©2013 MFMER | slide-8 Reported new tuberculosis cases, by county — United States, 2010–2012 DC Reported a new TB case Did not report a new TB case ©2013 MFMER | slide-9 Rate* of TB Cases by Race/Ethnicity — United States, 2012 2012 2012 2012 Race Race Rate Hispanic Asian Black White Race Rate Rate 5.2 Hispanic 5.2 19.8 Asian 19.8 5.7 Black 5.7 0.8 White 0.8 7.3x 6.6x Hispanic 5.2 Asian 19.8 Black 5.7 White 0.8 25.0x * Per 100,000 population Data are updated as of 2/22/13 and are provisional. ©2013 MFMER | slide-10 ©2010 MFMER | slide-11 ©2013 MFMER | slide-11 ©2013 MFMER | slide-12 ©2013 MFMER | slide-13 TB HIV/TB HIV ©2010 MFMER | slide-14 ©2013 MFMER | slide-14 TB and HIV Coinfection • Of the 8.7 million incident cases in 2011, 1.1 million (13%) were among people living with HIV • In Africa, 39% of TB cases were estimated to be coinfected • Africa accounted for 79% of TB cases among people living with HIV worldwide ©2013 MFMER | slide-15 ©2013 MFMER | slide-16 Estimated HIV Coinfection in Persons with HIV Test Results, United States, 1993–2012 30 % Coinfection 25 20 15 10 5 0 1993 1994 1995 1996 1997 1998 1999 2000 2001 2002 2003 2004 2005 2006 2007 2008 2009 2010 2011 2012 Aged 25-44 All Ages ©2013 MFMER | slide-17 Natural History ©2013 MFMER | slide-18 HIV Immune Deficiency • Loss of specific antibody responses • Reduced lymphocyte function decreased • Increased immune • Delayed hypersensitivity activation • Diminished T cell repertoire response to recall antigens • Phagocytosis • Chemotaxis • Intracellular killing • Natural killer cell-mediated killing • disruption of immunoregulatory cytokine expression and production • Decreased IL-2, γ interferon, and IL-12 • Increased IL-1, IL-6, TNFα ©2013 MFMER | slide-19 Natural History of M. tuberculosis infection. A Koul et al. Nature 469, 483-490 (2011) doi:10.1038/nature09657 ©2013 MFMER | slide-20 Tuberculous infection progressed to active disease within 106 days of acquiring the infection in 37 percent (11 of 30) of HIVinfected patients. Tuberculosis did not develop in any of 28 staff members with exposures, although there were 6 with documented tuberculin conversions and 8 others had positive tuberculin reactions of unknown duration. ©2013 MFMER | slide-21 7 of 18 (39%) HIV-infected inpatients developed active tuberculosis within 60 days of diagnosis of the index case ©2013 MFMER | slide-22 Natural History of M. tuberculosis infection. A Koul et al. Nature 469, 483-490 (2011) doi:10.1038/nature09657 ©2013 MFMER | slide-23 • Forty-nine HIV-seropositive subjects and 62 HIVseronegative subjects had a positive TST response. • Active tuberculosis developed in eight of the HIVseropositive subjects (4 percent) and none of the seronegative subjects during the study period (P<0.002). ©2013 MFMER | slide-24 HIV Epidemic State and Estimated Relative Risk of Developing Tuberculosis (TB) in HIVInfected Persons in Comparison with HIV-Uninfected Persons (Incidence Rate Ratio). Getahun H et al. Clin Infect Dis. 2010;50:S201-S207 ©2013 MFMER | slide-25 Diagnosis of LTBI in HIV-Infected Individuals ©2013 MFMER | slide-26 Am J Respir Crit Care Med Vol 175. pp 737–742, 2007 • • • • 294 HIV-infected subjects TST vs. QFT 89.3% concordance between QFT and TST only 28% subjects with positive test results by either TST or QFT had positive test results by both modalities. ©2013 MFMER | slide-27 • 2348 household contacts of sputum smear positive TB cases followed over 24 months • Twenty six contacts diagnosed with definite TB • TST sensitivity 56% • ELISPOT 52% • 71% were positive by one or other test • Those negative on either or both tests had the lowest rate of progression ©2013 MFMER | slide-28 J Acquir Immune Defic Syndr 2011;56:230–238 • 37 studies with 5736 HIV-infected individuals • Are IGRAs better than TST at predicting which HIV-infected individuals are at highest risk of progression to active TB? • Are IGRAs more sensitive than TST for diagnosis of MTB infection in HIV-infected individuals with advanced immunosuppression? • Insufficient evidence to conclude that either test is superior to the other. ©2013 MFMER | slide-29 • Testing for LTBI at the time of HIV diagnosis should be routine regardless of epidemiologic risk factors • Annual testing for LTBI only in those with ongoing exposure • If negative screen for LTBI while CD4 count is low, repeat testing post cART once CD4 is above 200. • Both TST and FDA-approved IGRAs are appropriate for TB screening in HIV-infected individuals • Routine use of both TST and IGRAs to screen for LTBI is not recommended ©2013 MFMER | slide-30 Natural History of M. tuberculosis infection. A Koul et al. Nature 469, 483-490 (2011) doi:10.1038/nature09657 ©2013 MFMER | slide-31 • 23,517 culture-positive, pulmonary tuberculosis patients from the California tuberculosis case registry from 1993 to 2007 who completed antituberculosis therapy. • 148 (0.63%) had a late recurrence. • Human immunodeficiency virus infection (adjusted hazard ratio, 1.81; p = 0.0149 ©2013 MFMER | slide-32 The Journal of Infectious Diseases 2010; 201:704–711 • Retrospective cohort study of South African gold miners, men with known dates of seroconversion to HIV (from 1991 to 1997) and HIV-negative men were followed up to 2004. • 342 HIV-positive and 321 HIV-negative men who had had1 previous episode of tuberculosis, • Rates of tuberculosis recurrence: • HIV-positive 19.7 cases per 100 person-years at risk (95% confidence interval [CI], 16.4–23.7) • 7.7 cases per 100 PYAR (95% CI, 6.1–9.8) ©2013 MFMER | slide-33 Diagnosis of Active Tuberculosis in the Setting of HIV ©2013 MFMER | slide-34 TB in the Setting of HIV: Clinical Presentation • In general, similar to that seen in HIV-uninfected patients • However, differential diagnosis broader • HIV itself • Other opportunistic infections • Extrapulmonary • HIV main risk factor • 35-80% vs. 15-50% • Bones and joints, lymph nodes, the pleura, peritoneum, CNS ©2013 MFMER | slide-35 Laboratory Diagnosis of Active TB • Sputum smear • HIV-infected patients are more likely to have smearnegative pulmonary TB • Range 31 – 81% • CXR • Individuals with advanced HIV are likely to have atypical presentations • Lower lobe locations • Less cavities • Consolidation • Intrathoracic LAD • May appear normal ©2013 MFMER | slide-36 Treatment of Tuberculosis ©2013 MFMER | slide-37 Treatment of LTBI Drugs Duration Interval Minimum doses Isoniazid 9 months Daily 270 Twice weekly* 76 Daily 180 Twice weekly 52 Isoniazid 6 months Isoniazid and Rifapentine 3 months Once weekly 12 Rifampin 4 months Daily 120 ©2013 MFMER | slide-38 Studies of the Effect of Human Immunodeficiency Virus (HIV) Coinfection on the Outcome of Tuberculosis Treatment. Swaminathan S et al. Clin Infect Dis. 2010;50:1377-1386 © 2010 by the Infectious Diseases Society of America ©2013 MFMER | slide-39 • Treatment of TB in HIV-infected individuals is the same as for those who are HIV uninfected • Treatment of TB in HIV-infected individuals should include an initial 4drug combination of isoniazid, rifampin, pyrazinamide, and ethambutol • DOT is recommended for all patients with suspected HIV-related TB • Daily therapy (5–7 days per week) given as DOT is recommended during the intensive phase • Daily (5–7 days per week) or thrice-weekly dosing is recommended during the continuation phase • Once- or twice-weekly dosing during the continuation phase assoc. with treatment failure/relapse with acquired rifamycin resistance ©2013 MFMER | slide-40 TB and HIV coinfection: Treatment Issues • Drug-drug interactions • Overlapping drug toxicities • Pill Burden • Immune-reconstitution inflammatory syndrome (IRIS) • Sequencing with antiretroviral therapy • Highly intermittent Dosing • TB treatment outcome in HIV • ART treatment effect on TB incidence ©2010 MFMER | slide-41 ©2013 MFMER | slide-41 TB/HIV Treatment Issues: Drug Interactions • Rifamycins induce hepatic cytochrome P450 (CYP3A4) enzymes, accelerating metabolism of: • Protease inhibitors (PIs), non-nucleoside reverse transcriptase inhibitors (NNRTIs), etc. • Rifampicin >> Rifabutin • For patients receiving PIs or NNRTIs, substitute rifabutin for rifampin, if available • Alternative non-rifamycin regimens less optimal, longer duration of therapy ©2010 MFMER | slide-42 ©2013 MFMER | slide-42 NNRTIs and Rifampicin Rifampicin decreases blood levels of NVP and EFV NNRTI Effect of Rifampicin Nevirapine 37–58% Efavirenz 22% ©2013 MFMER | slide-43 PIs and Rifamycins Rifampicin decreases blood levels of all PIs Protease Inhibitor Effect of Rifampicin Saquinavir by 84% Ritonavir by 35% Indinavir by 89% Nelfinavir by 82% Amprenavir by 81% Lopinavir/ritonavir by 75% ©2013 MFMER | slide-44 Rifamycins and Intermittent Therapy • Evidence for development of acquired rifamycin resistance with intermittent therapy • Intermittent therapy not recommended during initial phase of TB treatment in patients with HIV infection • No twice-weekly continuation phase with advanced immune suppression ©2013 MFMER | slide-45 Overlapping Side Effects Side Effect Skin rash* Nausea, vomiting TB Drug PZA, RIF, INH PZA, RIF, INH ARV Nevirapine Efavirenz Abacavir Zidovudine Ritonavir Amprenavir Indinavir * May also see rash with cotrimoxazole Burman et al, Am J Respir Crit Care Med 2001 ©2013 MFMER | slide-46 Overlapping Side Effects Side Effect Hepatitis Leukopenia, anemia TB Drug ARV Nevirapine Protease inhibitors PZA, RIF, INH IRIS (with chronic hepatitis) RIF Zidovudine Burman et al, Am J Respir Crit Care Med 2001 ©2013 MFMER | slide-47 TB/HIV: Barriers to Adherence • Dual social stigma • Higher pill burden • Greater number of potential drug side effects • Additional illness (opportunistic infections) • Difficult medical access, drug-supply interruptions ©2013 MFMER | slide-48 ©2013 MFMER | slide-49 IRIS Immune Reconstitution Inflammatory Syndrome (IRIS) Clinical worsening in the setting of an adequate response to ART • “Paradoxical” worsening of previously known treated (completed or ongoing) opportunistic pathogen • “Unmasking” of subclinical opportunistic pathogen ©2013 MFMER | slide-50 Incidence of Tuberculosis Immune Reconstitution Inflammatory Syndrome (IRIS) in Human Immunodeficiency Virus (HIV)–Tuberculosis Coinfection. Swaminathan S et al. Clin Infect Dis. 2010;50:1377-1386 © 2010 by the Infectious Diseases Society of America ©2013 MFMER | slide-51 IRIS • Risk factors • Disseminated TB • Shorter delay between onset of TB and ART drugs • Low baseline CD4, higher baseline viral load • Greater CD4 or viral load response to ART • Timing of onset • Usually within first 6 weeks of ART (often 2–3 weeks, but can be months after ART started) ©2013 MFMER | slide-52 IRIS Clinical presentation: • Fever • Nodal enlargement • Worsening pulmonary infiltrates (with or without respiratory symptoms) • Local worsening in extrapulmonary sites ©2013 MFMER | slide-53 IRIS Differential Diagnosis • TB treatment failure • Drug-resistant TB • ART failure • Other opportunistic (or non-opportunistic) infections • Lymphoma, Kaposi’s sarcoma • Hypersensitivity drug reactions ©2013 MFMER | slide-54 IRIS Management • Continue TB treatment • Continue ART • Exclude TB treatment failure • Adherence • Drug resistance • Exclude additional/new diagnosis • Consider NSAIDS, steroids • Drainage of lesions ©2013 MFMER | slide-55 Studies of the Timing of Antiretroviral Therapy (ART) in Human Immunodeficiency Virus (HIV)–Infected Patients with Tuberculosis Undergoing Antituberculosis Therapy. Swaminathan S et al. Clin Infect Dis. 2010;50:1377-1386 © 2010 by the Infectious Diseases Society of America ©2013 MFMER | slide-56 When to Start Antiretrovirals HIV-infected TB patients not yet on ART CD4 Start ART <50 Within 2 weeks of TB Rx >50 and TB severe Within 2 - 4 weeks of TB Rx >50 but TB not severe Can defer ART beyond 2 - 4 weeks but should start within 812 weeks ©2013 MFMER | slide-57 Antiretroviral Therapy (ART) Significantly Reduces TB Incidence Decrease in TB incidence after starting ART in resource-limited, high-burden area Lawn SD, et al, Am J Respir Crit Care Med, 2008;177:680-685 ©2013 MFMER | slide-58 Tuberculosis/HIV Coinfection: Summary • Substantial global disease burden, individually and as coinfection • Bidirectional impact on natural history • Difficulties in diagnosis and treatment • TB, even in the setting of HIV, is curable • ART has tremendous impact on TB incidence in the population ©2010 MFMER | slide-59 ©2013 MFMER | slide-59 ©2013 MFMER | slide-60