* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download trend lab
Survey
Document related concepts
Transcript
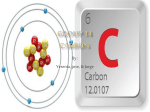
Name: Partner: Block The Periodic Table Background: The present organization of the elements is a product of the table Mendeleev created in 1869. The Periodic Law states that the chemical and physical properties of the elements are periodic functions of their atomic numbers. Each of the elements known today has its own set of characteristic properties. These range from the state of matter, color, conductivity, density, size, and a variety of other properties. The elements are arranged within the periodic table into groups (vertical columns) or families and horizontal periods. This arrangement reflects the repeating nature of the properties of the elements. Purpose: You will arrange the code elements in groups 1&2 (s block) and 13-18 ( the p block) (the entire d and f block is not present )according to atomic number, symbol, oxidation number, atomic radii, and electronegativity. Materials: Scissors, glue, tables Procedure: 1. Cut out all the blocks of the mystery elements arrange them into their vertical groups listed below. 2. Following the clues to place them onto the table. Using your knowledge of periodic trends re-orangized the elements within each group according to atomic radii and electronegativity. Place unlettered code boxes in according to oxidation number or size. 3. Glue them into the position you believe they are and identify them. 4. Fill in the atomic number, symbol, and oxidation state for all the mystery elements. THE CLUES: 1. The following elements are in the same group. Once in the group do not split them up! LKD, SNE, AWIXT, YMB, QCF, POH, RJV, ZGU 2. I is an alkali metal 3. M has 8 valence electrons 4. H is a halogen 5. P is the only liquid 6. N is an alkaline metal 7. S ions build strong bones and teeth it will lose 2 electrons to become stable 8. R is used as a poison 9. D is a metal that will melt in your hand! 10. R us a metalloid 11. U has six protons 12. O is a gas 13. Q has six valence electrons 14. E is the least reactive in its group 15. I is more reactive than S 16. X has the smallest atomic mass 17. C has 8 electrons 18. N was used in flash bulbs because it burns with a bright white light 19. BIRQPZDS ARE ALL IN THE SAME PERIOD 20. C is used in respiration of animals 21. K is used in soda cans 22. H is the most reactive non-metal Lab Analysis: Define the following terms and looking at your table describe the following trends you observe and explain why each trend is occurring 1. Atomic Radii- Down a group Across a period left to right 2. Electronegativity Down a Group Across a Period 3. Oxidation Number How is the electron configuration related to the oxidation number of a group? Post Lab Questions: 1. Explain how the size of a metal influences the reactivity of the metal? 2. Explain how the size of a halogen influences the reactivity of the halogen? 3. What is the oxidation number for the noble gases? Explain. 4. How could you use your table to predict the atomic radii for the element Iodine? What would you predict it to be? 5. How could you use your table to predict the value for electronegativity for carbon? What would you predict it to be?