* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download differential diagnosis of corpus callosum lesions
Survey
Document related concepts
Transcript
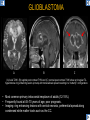
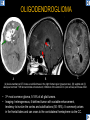
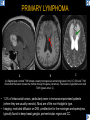
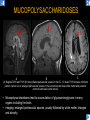
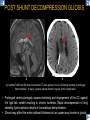
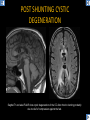
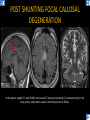
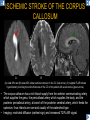
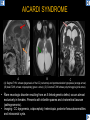
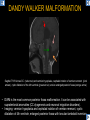
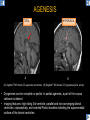
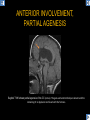
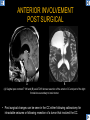
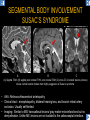
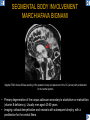
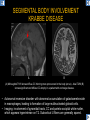
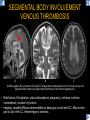
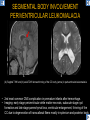
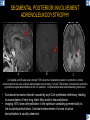
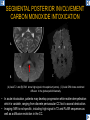
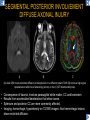
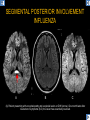
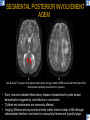
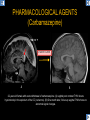
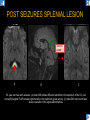
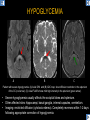
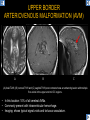
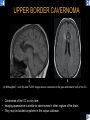
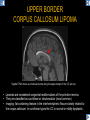
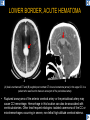
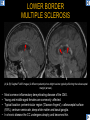
DIFFERENTIAL DIAGNOSIS OF CORPUS CALLOSUM LESIONS USING THE MIDLINE SAGITTAL PLANE Chapel Hill, NC Ames, Romy Amunugama, Monika Sepulveda, Francisco Causil, Lazaro Zamora, Carlos Castillo, Mauricio DISCLOSURES The authors have no pertinent disclosures. INTRODUCTION The corpus callosum (CC) is the largest white matter commissure between the cerebral hemispheres. It is traditionally divided into 4 segments (rostrum, genu, body and splenium) and 2 borders (upper and lower). Some authors consider the “isthmus” (between the posterior body and splenium) as a 5th segment. It is susceptible to involvement by a variety of conditions and diseases. Our aim is to demonstrate findings on sagittal MRI that may lead to a correct diagnosis. Lymphoma Diffuse DWM/DAI/Agenesis Oligodendroglioma/GB Mucopolysaccharidoses Aicardi Sd./Post shunt Ischemia Callosotomy Rostrum/ Genu Partial agenesis Krabbe Disease CORPUS CALLOSUM LESIONS, Predominant locations Venus thrombosis Body SEGMENTAL Marchiafava-Bignami Susac’s syndrome Periventricular leukomalacia ADEM Hypoglycemia Splenium Focal Carbamazepine MERS Adrenoleukodystrophy Lower Multiple Sclerosis BORDERS AVM/ Cavernoma Upper Lipomas Acute Hematoma LESIONS THAT AFFECT THE CORPUS CALLOSUM DIFFUSELY GLIOBLASTOMA A B C (A) Axial T2WI, (B) sagittal post contrast T1WI and (C) coronal post contrast T1WI show an irregular T2hyperintense ring enhancing lesion (arrows) with transcallosal spread resulting in a “butterfly” configuration. • Most common primary intracranial neoplasm of adults (12-15%). • Frequently found at 45–70 years of age, poor prognosis. • Imaging: ring enhancing lesions with central necrosis, preferential spread along condensed white matter tracts such as the CC. OLIGODENDROGLIOMA A B C (A) Axial unenhanced CT shows a calcified mass in the right frontal region (green arrows), (B) sagittal and (C) axial post contrast T1WI show minimal enhancement, infiltration of the anterior CC (red arrows) and mass effect. • 3rd most common glioma, 5-18% of all glial tumors. • Imaging: heterogeneous, ill-defined tumor with variable enhancement, tendency to involve the cortex and calcifications (50- 90%). It commonly arises in the frontal lobes and can cross to the contralateral hemisphere via the CC. PRIMARY LYMPHOMA A B C (A) Sagittal post contrast T1WI shows a nearly homogeneous enhancing lesion in the CC. (B) Axial T1WI shows that the lesion crosses the midline through the genu (red arrow). The lesion is hypointense on axial T2WI (green arrow, C) . • 1-2% of intracranial tumors, particularly seen in immunocompromised patients (where they are usually necrotic). Most are of the non Hodgkin's type. • Imaging: restricted diffusion on DWI, predilection for the meninges and ependyma, typically found in deep basal ganglia, periventricular region and CC. MUCOPOLYSACCHARIDOSES A B C (A) Sagittal T2WI and T1WI (B) show dilated perivascular spaces in the CC. (C) Axial T1WI shows a cribriform pattern (red arrow) of enlarged perivascular spaces in the subcortical and deep white matter and posterior centrum semiovale (white arrow). • Mucopolysaccharidoses lead to accumulation of glycosaminoglycans in many organs including the brain. • Imaging: enlarged perivascular spaces, usually followed by white matter changes and atrophy. POST SHUNT DECOMPRESSION GLIOSIS A B (A) Sagittal FLAIR and (B) axial unenhanced CT show gliosis in the CC following shunting of prolonged hydrocephalus. A “wavy” superior callosal border is typical of this complication. • Prolonged ventriculomegaly causes stretching and impingement of the CC against the rigid falx cerebri resulting in chronic ischemia. Rapid decompression of long standing hydrocephalus results in transcallosal demyelination. • Gliosis may affect the entire callosal thickness but an upper wavy border is typical. POST SHUNTING CYSTIC DEGENERATION Sagittal T1 and axial FLAIR show cystic degeneration of the CC after chronic shunting probably due to relief of compression against the falx. POST SHUNTING FOCAL CALLOSAL DEGENERATION In this patient, sagittal T1, axial FLAIR, and coronal T2 show post shunting CC involvement only in the body (arrow), unlike other cases in which the process is diffuse. ISCHEMIC STROKE OF THE CORPUS CALLOSUM A B C (A) Axial DWI and (B) axial ADC show restricted diffusion in the CC (red arrow). (C) Sagittal FLAIR shows hyperintensity involving the entire thickness of the CC in this patient with acute stroke (green arrow). • The corpus callosum has a rich blood supply from the anterior communicating artery which supplies the genu, the pericallosal artery which supplies the body, and the posterior pericallosal artery, a branch of the posterior cerebral artery, which feeds the splenium, thus infarcts are rare and usually of the watershed type. • Imaging: restricted diffusion (earliest sign) and increased T2/FLAIR signal. AICARDI SYNDROME A B C (A) Sagittal T1WI shows dysgenesis of the CC (red arrow) and pontocerebellar hypoplasia (orange arrow). (B) Axial T2WI, shows colpocephaly (green arrow). (C) Corornal T2WI shows polymicrogyria (pink arrow). • Rare neurologic disorder resulting from an X-linked genetic defect, occurs almost exclusively in females. Presents with infantile spasms and chorioretinal lacunae (pathognomonic). • Imaging: CC dysgenesis, colpocephaly, heterotopia, posterior fossa abnormalities and intracranial cysts. DANDY WALKER MALFORMATION Sagittal T1WI shows CC (red arrow) and vermian hypoplasia, cephalad rotation of vermian remnant (pink arrows), cystic dilatation of the 4th ventricle (green arrow) and an enlarged posterior fossa (orange arrow) • DWM is the most common posterior fossa malformation. It can be associated with supratentorial anomalies (CC dysgenesis and neuronal migration disorders). • Imaging: vermian hypoplasia and cephalad rotation of vermian remnant, cystic dilatation of 4th ventricle, enlarged posterior fossa with torcular-lambdoid inversion. AGENESIS TOTAL A HYPOPLASIA B (A) Sagittal T1WI shows CC agenesis (red arrow). (B) Sagittal T1WI shows CC hypoplasia (pink arrow). • Dysgenesis can be complete or partial. In partial agenesis, a part of the corpus callosum is absent. • Imaging features: high-riding 3rd ventricle, parallel and non-converging lateral ventricles, colpocephaly, and inverted Probst bundles indenting the superomedial surface of the lateral ventricles. LESIONS THAT FOCALLY AFFECT THE CORPUS CALLOSUM ANTERIOR INVOLVEMENT, PARTIAL AGENESIS Sagittal T1WI shows partial agenesis of the CC (arrow). The genu and anterior body are absent and the remaining CC is dysplastic and fused with the fornices. ANTERIOR INVOLVEMENT POST SURGICAL A B (A) Sagittal post contrast T1WI and (B) axial T2WI shows resection of the anterior CC and part of the right frontal lobe secondary to brain tumor. • Post surgical changes can be seen in the CC either following callosotomy for intractable seizures or following resection of a tumor that involved the CC. SEGMENTAL BODY INVOLVEMENT SUSAC’S SYNDROME A B C (A) Sagittal T2WI, (B) sagittal post contrast T1WI, and coronal T2WI (C) show CC ‘snowball’ lesions (arrows) whose central location makes them highly suggestive of Susac’s syndrome. • AKA: Retinocochleocerebral arteriopathy • Clinical triad – encephalopathy, bilateral hearing loss, and branch retinal artery occlusion. Usually self-limited. • Imaging: Similar to MS/ transcallosal lesions/ gray matter microinfarctions but no demyelination. Unlike MS, lesions are not isolated to the callososeptal interface. SEGMENTAL BODY INVOLVEMENT MARCHIAFAVA BIGNAMI Sagittal T2WI shows diffuse swelling of the posterior body and splenium of the CC (arrow) with predilection for its central portion. • Primary degeneration of the corpus callosum secondary to alcoholism or malnutrition. (vitamin B deficiency). Usually men aged 40-60 years. • Imaging: callosal demyelination and necrosis with subsequent atrophy, with a predilection for the central fibers. SEGMENTAL BODY INVOLVEMENT KRABBE DISEASE A B (A) Mid sagittal T1WI shows diffuse CC thinning more pronounced in the body (arrow). Axial T2WI (B) shows significant and diffuse CC atrophy in a patient with end stage disease. • Autosomal recessive disorder with abnormal accumulation of galactocerebroside in macrophages, leading to formation of large multinucleated globoid cells. • Imaging: involvement of pyramidal tracts, CC and parieto-occipital white matter, which appears hyperintense on T2. Subcortical U fibers are generally spared. SEGMENTAL BODY INVOLVEMENT VENOUS THROMBOSIS A B C (A) Mid sagittal, (B) coronal and CC) axial T2 images show involvement of the CC body (arrow) and adjacent white matter in a patient with thrombosis of the inferior sagittal sinus. • Risk factors: Dehydration, oral contraceptives, pregnancy, cirrhosis, extrinsic compression, invasion by tumors. • Imaging: variable diffusion abnormalities in deep gray nuclei and CC. May involve part or all of the CC. Hemorrhage is common. SEGMENTAL BODY INVOLVEMENT PERIVENTRICULAR LEUKOMALACIA A B (A) Sagittal T1WI and (b) axial T2WI shows thinning of the CC body (arrow) in periventricular leukomalacia • 2nd most common CNS complication in premature infants after hemorrhage. • Imaging: early stage periventricular white matter necrosis, subacute stage cyst formation and late stage parenchymal loss, ventricular enlargement, thinning of the CC due to degeneration of transcallosal fibers mostly in splenium and posterior body. SEGMENTAL POSTERIOR INVOLVEMENT ADRENOLEUKODYSTROPHY A B C (A) Sagittal and (B) axial post contrast T1WI show the characteristic pattern of symmetric contrast enhancement at the zone of active demyelination (red arrows). (C) Axial T2WI shows confluent and symmetric hyperintense signal abnormalities in the CC splenium , occipital and parietal lobes bilaterally (pink arrow). • Autosomal recessive disorder caused by acyl-CoA synthetase deficiency, leading to accumulation of very long chain fatty acids in tissues/plasma. • Imaging: 80% show demyelination in the splenium spreading symmetrically to the occipital/parietal lobes. Contrast enhancement of zones of active demyelination is usually observed. SEGMENTAL POSTERIOR INVOLVEMENT CARBON MONOXIDE INTOXICATION A B C (A) Axial T2 and (B) DWI show high signal in the splenium (arrow). (C) Axial DWI shows restricted diffusion in the globus pallidi bilaterally. • In acute intoxication, patients may develop progressive white matter demyelination which is variable, ranging from discrete perivascular CC foci to axonal destruction. • Imaging: MRI is not specific, including high signal in T2 and FLAIR sequences as well as a diffusion restriction in the CC. SEGMENTAL POSTERIOR INVOLVEMENT DIFFUSE AXONAL INJURY A B C (A) Axial DWI shows restricted diffusion in the splenium. In a different patient T2WI (B) shows a high signal splenial lesion with loss of anisotropy (arrow) on the (C) DTI directionality map. • • • • Consequence of trauma, involves parasagittal white matter, CC and brainstem. Results from acceleration/deceleration that shear axons. Splenium and posterior CC are more commonly affected. Imaging: hemorrhage, hypointensity on T2/SWI images. Non hemorrhagic lesions show restricted diffusion. SEGMENTAL POSTERIOR INVOLVEMENT INFLUENZA RM 14/01/2008 RM 19/12/2007 A B C (A) Patient presenting with encephalopathy and a splenial lesion on DWI (arrow). One month later after resolution of symptoms (B-C) the lesion has essentially resolved. SEGMENTAL POSTERIOR INVOLVEMENT ADEM A B (A & B) Axial T2 images show typical patchy white and gray matter ADEM lesions with little mass effect. Note bilateral splenial involvement in A (arrows). • Rare, immune mediated inflammatory disease characterized by wide spread demyelination triggered by viral infection or vaccination. • Children and adolescents are commonly affected. • Imaging: Bilateral and asymmetrical white matter lesions similar to MS although callososeptal interface involvement is unusual and lesions are typically larger. TRANSIENT LESIONS IN THE SPLENIUM OF THE CORPUS CALLOSUM • Various causes: patients receiving antiepileptic drugs, hypoglycemia, post seizures, high-altitude and infective agents (influenza, rotavirus, mumps, Escherichia coli, adenovirus). • Possibly due to intramyelinic edema. • Needs to be considered to avoid unnecessary invasive diagnostic and therapeutic measures. PHARMACOLOGICAL AGENTS (Carbamazepine) 1 MONTH LATER A B 22-year-old female with acute withdrawal of carbamazepine. (A) sagittal post contrast T1WI shows hypointensity in the splenium of the CC (red arrow). (B) One month later, follow-up sagittal T1WI shows no abnormal signal changes. POST SEIZURES SPLENIAL LESION 1 MONTH LATER A B C 30 -year-old male with seizures. (A) Axial DWI shows diffusion restriction in the splenium of the CC (red arrow)(B) Sagittal FLAIR shows high-intensity in the splenium (green arrow). (C) Axial DWI one month later shows resolution of the signal abnormalities. HYPOGLYCEMIA A B C Patient with severe hypoglycemia. (A) Axial DWI and (B) ADC map show diffusion restriction in the splenium of the CC (red arrow). (C) Axial FLAIR shows mild high-intensity in the splenium (green arrow). • • • Severe hypoglycemia usually affects the occipital lobes and splenium. Other affected sites: hippocampi, basal ganglia, internal capsules, cerebellum. Imaging: restricted diffusion (cytotoxic edema). Completely reverses within 1-2 days following appropriate correction of hypoglycemia. LESIONS THAT PREFERENTIALLY AFFECT THE BORDERS OF THE CORPUS CALLOSUM UPPER BORDER ARTERIOVENOUS MALFORMATION (AVM) A B C (A) Axial T2WI, (B) coronal T1WI and (C) sagittal T1WI post contrast shows an enhancing lesion with multiple flow voids in the upper and mid CC regions. • In this location: 10% of all cerebral AVMs. • Commonly present with intraventricular hemorrhage. • Imaging: shows typical signal voids and tortuous vasculature. UPPER BORDER CAVERNOMA A B (A) Mid sagittal T1 and (B) Axial FLAIR images show a cavernoma in the genu and anterior body of the CC. • Cavernoma of the CC is very rare. • Imaging appearance is similar to cavernomas in other regions of the brain. • They may be located anywhere in the corpus callosum. UPPER BORDER CORPUS CALLOSUM LIPOMA Sagittal T1WI shows a curvilinear lipoma along the upper margin of the CC (arrow). • Lipomas are considered congenital malformations of the primitive meninx. • They are classified as curvilinear or tubulonodular (most common). • Imaging: fat-containing lesions in the interhemispheric fissure closely related to the corpus callosum. In curvilinear types the CC is normal or mildly dysplastic. LOWER BORDER, ACUTE HEMATOMA A B (A) Axial unenhanced CT and (B) sagittal post contrast CT show a hematoma (arrow) in the upper CC in a patient who was found to have an aneurysm of the pericallosal artery. • Ruptured aneurysms of the anterior cerebral artery or the pericallosal artery may cause CC hemorrhage. Hemorrhage in this location can also be associated with ventriculostomies. Other less frequent etiologies: isolated cavernoma of the CC or microhemorrhages occurring in severe, non-lethal high-altitude cerebral edema. LOWER BORDER MULTIPLE SCLEROSIS A B (A) & (B) Sagittal FLAIR images (2 different patients) show bright lesions typically affecting the callososeptal margin (arrows). • Most common inflammatory demyelinating disease of the CNS. • Young and middle aged females are commonly affected. • Typical location: periventricular region (“Dawson fingers”), callososeptal surface (93%), centrum semiovale, deep white matter and basal ganglia. • In chronic disease the CC undergoes atrophy and becomes thin. CONCLUSION MRI in the sagittal plane is useful in the evaluation of the CC and may provide clues with respect to the pattern and extent of different diseases narrowing the differential diagnosis. REFERENCES 1. 2. 3. 4. 5. B Battal, M Kocaoglu, V Akgun et al. Corpus callosum: Normal imaging appearance, variants and pathologic conditions. Journal of Medical Imaging and Radiation Oncology 54 (2010) 541–549. S. A. Friese, M. Bitzer, D. Freudenstein et al. Classification of acquired lesions of the corpus callosum with MRI. Neuroradiology 2000) 42: 795±802. A Fitsiori, D Nguyen, A Karentzos et al. The corpus callosum: white matter or terra incognita. The British Journal of Radiology, 84 (2011), 5–18. Aamish Z Kazi, Priscilla C Joshi, Abhimanyu B Kelkar et al. MRI evaluation of pathologies affecting the corpus callosum: A pictorial essay. Indian J Radiol Imaging. 2013 Oct-Dec; 23(4): 321–332. David L. Kasow, Sylvie Destian, Carl Braun, John C et al. Corpus Callosum Infarcts with Atypical Clinical and Radiologic Presentations. AJNR Am J Neuroradiol 21:1876–1880, Nov/Dec 2000.