* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Drug Interaction with Immunosuppressants
Survey
Document related concepts
Transcript
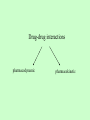
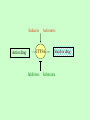
Drug Interaction with Immunosuppressants A Case Report and Review Immunospuppression in transplantation Aim of immunosuppression: Prevent acute rejection, while avoiding infection and toxicity. The primary efficacy end-points of immunosuppression: The incidence of clinical or biopsy-proven acute rejection in the first six months or one year. Graft survival and patient survival are also reported. Secondary end-points of immunosuppression: The incidence of side-effects, such as infection, renal dysfunction, diabetes, and lymphoproliferative disease. Calcineurin inhibitor (CNI) Cyclosporin: cyclic peptide Tacrolimus: macrolide Anti-metabolites Azathioprine Mycophenolate mofetil (MMF) Glucocorticoids Methylprednisolone Prednisolone TOR inhibitors Sirolimus: macrolide Everolimus ATG (anti-thymocyte globulin) or interleukin-2 receptor antagonists, such as basiliximab or daclizumab The combinations of drugs and variations in the doses, formulations, timing, and weaning schedules are almost endless. Adverse Effects of Immunosuppressants 1. the toxicity resulting from over-immunosuppression (eg, infection) 2. Drug-specific adverse reactions. Side effects of TOR inhibitors The main side effects are: hypercholesterolaemia infection acne mouth ulcers pneumonitis gastrointestinal side effects wound dehiscence Nephrotoxicity, neurotoxicity, hypertension and glucose intolerance are not usually observed. Case Report Age: two year-old Sex: female ABO-incompatible heart transplantation at four months of age. Maintenance immunosuppression included: tacrolimus (FK506) 0.8 mg p.o. q12 h mycophenolate mofetil 100 mg p.o. q12 h. prednisolone -3 weeks: routine echocardiography and EMBx were normal. Day -2 to day 0: recurrent fever with no focus progressive anemia renal dysfunction in a local hospital. Day 1: Transfered to HSC signs of congestive heart failure. ECG revealed ischemic changes. decreased LVEF with dilatation and severe right ventricular dysfunction. One small arterial vessel exhibited intimal hyperplasia consistent with CAD. Liver transaminases were mildly elevated (AST 69 umol/L, ALT 49 umol/L). INR was 1.64 and albumin was 24 g/L. Hemoglobin was low (72 g/ L) Renal dysfunction progressed. The patient was transfused with packed red blood cells. (Normalized hemoglobin) Methylprednisolone and Thymoglobulin® was discontinued. The patient was re-listed for another heart transplant. Day 2 to 6: Tacrolimus was held on day 2 for five days because of a reported trough blood concentration >30 ug/L [EMIT immunoassay, Viva Analyzer (Dade Behring, Deerfield, IL, USA)]. The patient required one-fourth of her pre-admission maintenance dose (0.02 mg/kg/day orally) to keep trough concentrations within the target range of 8–10 ug/L. Potential influencing factors such as liver dysfunction, decreased enteral intake (resulting in increased tacrolimus absorption) and drug interaction (concomitant pulse methylprednisolone) resulting in decreased calcineurin inhibitor metabolism and excretion were present. Day 5: Liver transaminases had normalized. Albumin was 27 g/L. Day 7: Nadolol and amiodarone were prescribed on day 7 to control arrhythmia. Day 8: Tacrolimus trough concentrations reported day 8 and day 10 were within the target range. Day 10: Conjugated bilirubin was zero and the unconjugated fraction was eight. Day 11: Sirolimus was prescribed on day 11 in an attempt to augment immunosuppression yet spare calcineurin inhibitor exposure given the patient’s renal dysfunction. The initial dose of 1 mg p.o. daily (0.06 mg/kg/day) was low in relation to our centre’s experience with children in this age group. Day 13: Given that tacrolimus was to be discontinued, sirolimus was then increased to 2 mg p.o. daily (0.12 mg/kg/day). Day 16: Sirolimus was held. The target trough concentration was 10 ug/L. The trough concentration (Ultima Tandem Mass Spectrometer, Waters Ltd., Centennial Park, Elstree, Hertfordshire, UK) was 52.7 ug/L. Tacrolimus was discontinued. Trough concentration was 12.9 ug/L Day 17-30: Sirolimus blood concentrations remained above the target range for an additional 14 days. Day 31: Once the trough concentration normalized, sirolimus was restarted at a smaller dose 0.5 mg p.o. daily (0.03 mg/kg/day). Day 32: The dose was further decreased to 0.3 mg p.o.daily (0.02 mg/kg/day). trough concentration = 17.2 ug/L. Day 37: The trough blood concentration was measured after five days of therapy and was reported as 13.4 lg/L, still above the target concentration. Day 42: The patient succumbed because of complications of ECMO. Triglycerides and cholesterol were not measured during the hospital course; however, blood samples were intermittently reported as lipemic. Amiodarone serum concentrations were not analyzed. Why immunosuppressants needs to be monitored? 1. Narrow therapeutic index Therapeutic index = toxic dose/minimal effective dose graft rejection adverse effects Desirable pharmacological effect only within narrow ranges of concentration in the blood. 2. Poor dose response relationship Highly variable absorption, distribution and metabolism between and within patients. Poor solubility in water Drug-drug interaction Polymorphism or defectiveness in key enzymes responsible for metabolizing immunosuppressants . 3. The clinical effect is difficult to measure Graft rejection may be the first indication of therapeutic ineffectiveness. The major adverse effect of the drug is hard to distinguish from a lack of therapeutic effects. Common medications taken by transplant recipients Immunosuppressants Antidepressants Antimicrobials Cholesterol-lowering Agents Antihypertensives Anti-ulcer Medications Drug-drug interactions pharmacodynamic pharmacokinetic Pharmacodynamic interactions Drug-drug interactions that modulate the drug's effect in the absence of a corresponding change in plasma concentration. Examples include interactions at the level of receptor binding or secondary messenger systems. For example, an increase in renal toxicity is observed with the coadministration of naproxen and CsA. Pharmacokinetic interactions Drug-drug interactions that alter the plasma concentration of 1 or both drugs. Drug-metabolizing enzymes Drug transporters Common Enzymes Involved in Immunosuppressant Metabolism Cytochrome P450s (CYPs), ie, 3A4/5, CsA, SIR, and TAC UDP-glucuronosyltransferases (UTGs) MPA Xanthine oxidase AZA Flavin mono-oxygenases, sulfotransferases, and N-acetyl transferases ??? UDP-glucuronosyltransferases (UTGs) in MPA Metabolism The UGT superfamily is composed of 2 families: UGT1 and UGT2. Conjugate substrates with the addition of a glucuronyl group. The rate-limiting step in the metabolism of MPA (active form of MMF). Xanthine oxidase in AZA Metabolism AZA 6-mercaptopurine inactivation by xanthine oxidase allopurinol inhibits xanthine oxidase, co-administration of allopurinol and azathioprine can result in increased levels of 6mercaptopurine. CYPs The most prominent family of drug-metabolizing enzymes. Inducers Active drug Activators CYP3A Inhibitors Inactive drug Substrates Drug transporters and Immunosuppressant Major transporter P-glycoprotein (P-gp), the protein product of the multidrug resistance 1 gene (MDR1). CsA and TAC (substrates, inhibtors, inducers). SIR (inhibtor, inducer) Other transporters multidrug resistance gene 2/3, organic anion transporter proteins multidrug resistance-related proteins (MRP1-6) Sister of P-glycoprotein (S-Pgp) CYP3A and P-gp Substrates (S), Inhibitors (I), Inducers(In), and Activators (A) CYP3A Immunosuppressants Cyclosporine (S, I) Methylprednisolone (I) Sirolimus (S, I) Tacrolimus (S) P-gp Cyclosporine (S, I, In) Methylprednisolone (S) Prednisolone (S, In) Sirolimus (I, In) Tacrolimus (S, I, In) Antibiotics/antifungal agents Clarithromycin (S) Clotrimazole (I) Erythromycin (S, I) Itraconazole (S, I) Ketoconazole (S, I) Clarithromycin (I) Clotrimazole (In) Erythromycin (S, I, In) Itraconazole (S, I) Ketoconazole (I) Sparfloxacin (S) Antihistamines Astemizole (I) Chlorpromazine (I) Cimetidine (S) Fexofenadine (S) Ranitidine (S) Astemizole (S, I) Cimetidine (I) Loratidine (S) Terfenadine (PS, I) CYP3A P-gp Psychoactive agents/anticonvulsants Amitriptyline (S) Carbamazepine (In) Midazolam (I, In) Nefazodone (I) Nortriptyline (S) Phenobarbital (In) Phenytoin (S, In) Amitriptyline (S) Midazolam (S, I) Nefazodone (S, I) Phenobarbital (In) Hormonal agents Cortisol (S) Dexamethasone (S, I) Estradiol (S, I) Hydrocortisone (S) Progesterone (S, I) Testosterone (S, A) Aldosterone (S) Cortisol (I) Dexamethasone (S, In) Estradiol (In, MS) Hydrocortisone (S, I) Insulin (In) Progesterone (I) Testosterone (I) Other cytotoxics Colchicine (S) Colchicine (S, In) Mitoxantrone (S, In) CYP3A P-gp Cardiac agents Amiodarone (S) Atorvastatin (S) Digitoxin (PS) Diltiazem (S, I) Felodipine (S) Fluvastatin (S) Lidocaine (S) Lovastatin (S) Nicardipine (S, I) Nifedipine (S, I) Nitrendipine (S, I) Pravastatin (S) Quinidine (S, I) Simvastatin (S) Verapamil (S, I) Amiodarone (I, In) Atorvastatin (I) Celiprolol (S) Digoxin (S) Diltiazem (S, I, In) Felodipine (I) Lidocaine (I) Nicardipine (I, In) Nifedipine (In) Nitrendipine (I) Quinidine (S, I) Talinolol (S) Terfenadine (I) Verapamil (S, I, In) HIV agents Amprenavir (S, I) Indinavir (S, I) Nelfinavir (S, I) Ritonavir (S, I) Saquinavir (S, I) Amprenavir (S, In) Indinavir (S) Nelfinavir (S, I, In) Ritonavir (S, I, In) Saquinavir (S, I) CYP3A Anticancer drugs Busulfan (S) Docetaxel (S) Doxorubicin (S) Etoposide (S) Paclitaxel (S) Tamoxifen (S) Vinblastine (S, I) Vincristine (S, I) Vindesine (S) Benzodiazepines Alprazolam (A) Clonazepam (S) Midazolam (S) Triazolam (S) P-gp Actinomycin D (S) Cisplatin (S, In) Cytarabine (S) Daunorubicin (S, In) Docetaxel (S) Doxorubicin (S, In) Etoposide (S, In) Fluorouracil (S, In) Methotrexate (S, In) Mitomycin (S) Paclitaxel (S) Taxol (S) Tamoxifen (I, In) Vinblastine (S, I, In) Vincristine (S, In) Vindesine (S) CYP3A P-gp Opiates Methadone (S, I) Fentanyl (S) Loperamide (S) Methadone (S, I) Morphine (S, In) Others Grapefruit juice (I) Losartan (S) St. John's wort (In) Bepridil (I) Bromocriptine (In) Chlorambucil (S, In) Dipyridamole (I) Disulfiram (I) Grapefruit juice (A) Hydroxyurea (S, In) Losartan (S) Octreotide (S) Phenothiazine (I) Probenecid (In) Quinine (I) Reserpine (I, In) Rifampin (In) St. John's wort (In) Tetrabenazine (I) Topotecan (S) Valinomycin (I) Yohimbine (In) 1: Clin Pharmacokinet. 2006;45(2):169-75. Pharmacokinetic interaction between levofloxacin and ciclosporin or tacrolimus in kidney transplant recipients: ciclosporin, tacrolimus and levofloxacin in renal transplantation. Federico S, Carrano R, Capone D, Gentile A, Palmiero G, Basile V. 2: Transplantation. 2006 Jan 15;81(1):136-7. Interaction between tacrolimus and fumagillin in two kidney transplant recipients. Arzouk N, Michelon H, Snanoudj R, Taburet AM, Durrbach A, Furlan V. 3: Pharmacotherapy. 2006 Feb;26(2):289-95. Links Sirolimus-itraconazole interaction in a hematopoietic stem cell transplant recipient. Said A, Garnick JJ, Dieterle N, Peres E, Abidi MH, Ibrahim RB. 4: Transplantation. 2006 Feb 15;81(3):487-8. Interaction between tacrolimus and omeprazole in a pediatric liver transplant recipient. Moreau C, Debray D, Loriot MA, Taburet AM, Furlan V. 5: J Clin Pharmacol. 2006 Mar;46(3):328-36. Interaction of single-dose ezetimibe and steady-state cyclosporine in renal transplant patients. Bergman AJ, Burke J, Larson P, Johnson-Levonas AO, Reyderman L, Statkevich P, Maxwell SE, Kosoglou T, Murphy G, Gottesdiener K, Robson R, Paolini JF. 6: Biol Blood Marrow Transplant. 2006 Mar;12(3):325-34. Pharmacokinetic evaluation of the drug interaction between intravenous itraconazole and intravenous tacrolimus or intravenous cyclosporin A in allogeneic hematopoietic stem cell transplant recipients. Leather H, Boyette RM, Tian L, Wingard JR. 7: Int J Clin Pharmacol Ther. 2006 Apr;44(4):163-71. Evaluation of the pharmacokinetic interaction between fluvastatin XL and cyclosporine in renal transplant recipients. Holdaas H, Hagen E, Asberg A, Lund K, Hartman A, Vaidyanathan S, Prasad P, He YL, Yeh CM, Bigler H, Rouilly M, Denouel J. 8: Transplant Proc. 2006 May;38(4):1138-40. Pharmacokinetic interaction between Amprenavir/Ritonavir and FosAmprenavir on cyclosporine in two patients with human immunodeficiency virus infection undergoing orthotopic liver transplantation. Guaraldi G, Cocchi S, Codeluppi M, Di Benedetto F, Bonora S, Motta A, Luzi K, Pecorari M, Gennari W, Masetti M, Gerunda GE, Esposito R. 9: Med Monatsschr Pharm. 2006 May;29(5):179-82. [Interaction between an antiinfective agent and an immunosuppressant after liver transplantation] [Article in German] Swoboda S, Meyer-Massetti C, Hoppe-Tichy T. 10: Clin Pharmacol Ther. 2006 Nov;80(5):509-21. Rifampin induces alterations in mycophenolic acid glucuronidation and elimination: implications for drug exposure in renal allograft recipients. Naesens M, Kuypers DR, Streit F, Armstrong VW, Oellerich M, Verbeke K, Vanrenterghem Y. 11: Pharmacol Res. 2006 Sep;54(3):181-5. Co-administration of sirolimus alters tacrolimus pharmacokinetics in a dose-dependent manner in adult renal transplant recipients. Baldan N, Rigotti P, Furian L, Margani G, Ekser B, Frison L, De Martin S, Palatini P. 12: Transplantation. 2006 Jun 27;81(12):1750-2. Clinically relevant drug interaction between voriconazole and tacrolimus in a primary renal allograft recipient. Kuypers DR, Claes K, Evenepoel P, Vanrenterghem Y.