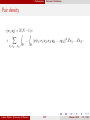* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Lecture on DFT in English is available on the web page
Atomic orbital wikipedia , lookup
Density matrix wikipedia , lookup
Hydrogen atom wikipedia , lookup
Atomic theory wikipedia , lookup
Tight binding wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
Electron configuration wikipedia , lookup
Density Functional Theory Łukasz Rajchel Interdisciplinary Center for Mathematical and Computational Modeling [email protected] Warsaw, 2010 Lecture available online: http://tiger.chem.uw.edu.pl/staff/lrajchel/ Questions, comments, mistakes in the Lecture — don’t hesitate to write me! Outline of the Lecture Part I 1 2 3 4 DFT — A Real Celebrity Preliminaries Basic Concepts of Quantum Chemistry Electronic Distribution Approximate Methods Hartree-Fock Variation in HF Equations Correlation and exchange Self-Interaction in HF Fermi and Coulomb Holes Definitions Outline of the Lecture Part II 5 6 7 8 Density and Energy Remarks and Problems Historical Models Results Hohenberg-Kohn Theorems Definitions The Theorems Representability of the Density Kohn-Sham Approach Introductory Remarks KS Determinant and KS Energy xc Functionals Is There a Road Map? Adiabatic Connection Outline of the Lecture Part III 9 Approximate xc Functionals Introduction LDA and LSD GGA Hybrid Functionals Beyond GGA Problems of Approximate Functionals Part I The Road to DFT. Recapitulation of Basic Concepts of Quantum Chemistry Outline of the Talk 1 DFT — A Real Celebrity 2 Preliminaries 3 Hartree-Fock 4 Fermi and Coulomb Holes DFT — A Real Celebrity DFT vs. CC vs. nano 9000 density functional theory coupled cluster nanotechnology 8000 number of publications 7000 6000 5000 4000 3000 2000 1000 0 20 20 20 20 20 20 20 20 20 20 19 09 08 07 06 05 04 03 02 01 00 99 year Number of publications returned by the Web of Science for the respective topics Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 6 / 101 Outline of the Talk 1 DFT — A Real Celebrity 2 Preliminaries Basic Concepts of Quantum Chemistry Electronic Distribution Approximate Methods 3 Hartree-Fock 4 Fermi and Coulomb Holes Preliminaries Basic Concepts of Quantum Chemistry Symbols used in the lecture: system composed of N electrons and M nuclei, Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 8 / 101 Preliminaries Basic Concepts of Quantum Chemistry Symbols used in the lecture: system composed of N electrons and M nuclei, atomic units used (melectron = 1, ~ = 1, e = 1), Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 8 / 101 Preliminaries Basic Concepts of Quantum Chemistry Symbols used in the lecture: system composed of N electrons and M nuclei, atomic units used (melectron = 1, ~ = 1, e = 1), xi Xα ri = yi , Rα = Yα — positions electrons, nuclei, zi Zα Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 8 / 101 Preliminaries Basic Concepts of Quantum Chemistry Symbols used in the lecture: system composed of N electrons and M nuclei, atomic units used (melectron = 1, ~ = 1, e = 1), xi Xα ri = yi , Rα = Yα — positions electrons, nuclei, zi Zα rij = |ri − rj | — interelectron distance, Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 8 / 101 Preliminaries Basic Concepts of Quantum Chemistry Symbols used in the lecture: system composed of N electrons and M nuclei, atomic units used (melectron = 1, ~ = 1, e = 1), xi Xα ri = yi , Rα = Yα — positions electrons, nuclei, zi Zα rij = |ri − rj | — interelectron distance, Zα — nuclear charge, Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 8 / 101 Preliminaries Basic Concepts of Quantum Chemistry Symbols used in the lecture: system composed of N electrons and M nuclei, atomic units used (melectron = 1, ~ = 1, e = 1), xi Xα ri = yi , Rα = Yα — positions electrons, nuclei, zi Zα rij = |ri − rj | — interelectron distance, Zα — nuclear charge, q = (r; σ) — spatial (r ∈ R3 ) and spin (σ = ± 21 ) variable, Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 8 / 101 Preliminaries Basic Concepts of Quantum Chemistry Symbols used in the lecture: system composed of N electrons and M nuclei, atomic units used (melectron = 1, ~ = 1, e = 1), xi Xα ri = yi , Rα = Yα — positions electrons, nuclei, zi Zα rij = |ri − rj | — interelectron distance, Zα — nuclear charge, q = (r; σ) — spatial (r ∈ R3 ) and spin (σ = ± 21 ) variable, ∆ = ∇2 = ∂2 ∂x2 + Łukasz Rajchel (University of Warsaw) ∂2 ∂y 2 + ∂2 ∂z 2 — Laplacian, DFT Warsaw, 2010 8 / 101 Preliminaries Basic Concepts of Quantum Chemistry Symbols used in the lecture: system composed of N electrons and M nuclei, atomic units used (melectron = 1, ~ = 1, e = 1), xi Xα ri = yi , Rα = Yα — positions electrons, nuclei, zi Zα rij = |ri − rj | — interelectron distance, Zα — nuclear charge, q = (r; σ) — spatial (r ∈ R3 ) and spin (σ = ± 21 ) variable, ∆ = ∇2 = ∂2 ∂x2 + ∂2 ∂y 2 + ∂2 ∂z 2 — Laplacian, P̂ij — operator exchanging particles i and j (permutator). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 8 / 101 Preliminaries Basic Concepts of Quantum Chemistry Born-Oppenheimer approximation We can separate nuclear and electronic motions because mproton ≈ mneutron ≈ 1836 × melectron . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 9 / 101 Preliminaries Basic Concepts of Quantum Chemistry Born-Oppenheimer approximation We can separate nuclear and electronic motions because mproton ≈ mneutron ≈ 1836 × melectron . We restrict our attention to electronic Hamiltonian only, Ĥel = T̂ |{z} kinetic energy Łukasz Rajchel (University of Warsaw) + V̂ne |{z} nuclear-electron attraction DFT + V̂ee . |{z} electron-electron repulsion Warsaw, 2010 9 / 101 Preliminaries Basic Concepts of Quantum Chemistry Born-Oppenheimer approximation We can separate nuclear and electronic motions because mproton ≈ mneutron ≈ 1836 × melectron . We restrict our attention to electronic Hamiltonian only, Ĥel = T̂ |{z} kinetic energy + V̂ne |{z} nuclear-electron attraction + V̂ee . |{z} electron-electron repulsion N N X M N −1 X N X X 1X Zα −1 ∆ri , V̂ne = − , V̂ee = rij T̂ = − . 2 |ri − Rα | i=1 Łukasz Rajchel (University of Warsaw) i=1 α=1 DFT i=1 j=i+1 Warsaw, 2010 9 / 101 Preliminaries Basic Concepts of Quantum Chemistry Born-Oppenheimer approximation We can separate nuclear and electronic motions because mproton ≈ mneutron ≈ 1836 × melectron . We restrict our attention to electronic Hamiltonian only, Ĥel = T̂ |{z} kinetic energy + V̂ne |{z} + nuclear-electron attraction V̂ee . |{z} electron-electron repulsion Clamped nuclei ⇒ nuclear-nuclear repulsion is a constant, so we can skip it now, but remember to add it to the result: Ĥ = Ĥel + V̂nn = Ĥel + M −1 X M X Zα Zβ . Rαβ α=1 β=α+1 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 9 / 101 Preliminaries Basic Concepts of Quantum Chemistry Schrödinger equation Spectrum of the Hamiltonian — wavefunctions and energies for the electronic states: Ĥψk (q1 ; q2 ; . . . ; qN ) = Ek ψk (q1 ; q2 ; . . . ; qN ). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 10 / 101 Preliminaries Basic Concepts of Quantum Chemistry Schrödinger equation Spectrum of the Hamiltonian — wavefunctions and energies for the electronic states: Ĥψk (q1 ; q2 ; . . . ; qN ) = Ek ψk (q1 ; q2 ; . . . ; qN ). The Schrödinger equation is an eigeinequation in which the input is the Hamiltonian itself. As output, we obtain ψk ’s and Ek ’s. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 10 / 101 Preliminaries Basic Concepts of Quantum Chemistry Schrödinger equation Spectrum of the Hamiltonian — wavefunctions and energies for the electronic states: Ĥψk (q1 ; q2 ; . . . ; qN ) = Ek ψk (q1 ; q2 ; . . . ; qN ). The Schrödinger equation is an eigeinequation in which the input is the Hamiltonian itself. As output, we obtain ψk ’s and Ek ’s. Solving the Schrödinger equation is not a trivial issue → analytical solution known only for H and H-like systems. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 10 / 101 Preliminaries Basic Concepts of Quantum Chemistry Schrödinger equation Spectrum of the Hamiltonian — wavefunctions and energies for the electronic states: Ĥψk (q1 ; q2 ; . . . ; qN ) = Ek ψk (q1 ; q2 ; . . . ; qN ). The Schrödinger equation is an eigeinequation in which the input is the Hamiltonian itself. As output, we obtain ψk ’s and Ek ’s. Solving the Schrödinger equation is not a trivial issue → analytical solution known only for H and H-like systems. From now on we are interested in the ground state only: ψk → ψ0 . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 10 / 101 Preliminaries Basic Concepts of Quantum Chemistry Wavefunction ψ: a complex function of 4N variables, no physical meaning. But: P (q1 ; q2 ; . . . ; qN ) = |ψ(q1 ; q2 ; . . . ; qN )|2 is the density probability of finding the electrons at positions q1 , q2 , . . . , qN . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 11 / 101 Preliminaries Basic Concepts of Quantum Chemistry Wavefunction ψ: a complex function of 4N variables, no physical meaning. But: P (q1 ; q2 ; . . . ; qN ) = |ψ(q1 ; q2 ; . . . ; qN )|2 is the density probability of finding the electrons at positions q1 , q2 , . . . , qN . Indistinguishable particles → the exchange of any two particles can’t change the density probabily, so P̂ij P (q1 ; . . . ; qi ; . . . ; qj ; . . . ; qN ) = P (q1 ; . . . ; qj ; . . . ; qi ; . . . ; qN ). more on P̂ij Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 11 / 101 Preliminaries Basic Concepts of Quantum Chemistry Wavefunction ψ: a complex function of 4N variables, no physical meaning. But: P (q1 ; q2 ; . . . ; qN ) = |ψ(q1 ; q2 ; . . . ; qN )|2 is the density probability of finding the electrons at positions q1 , q2 , . . . , qN . Indistinguishable particles → the exchange of any two particles can’t change the density probabily, so P̂ij P (q1 ; . . . ; qi ; . . . ; qj ; . . . ; qN ) = P (q1 ; . . . ; qj ; . . . ; qi ; . . . ; qN ). This yields the two possibilities: ( ψ → bosons: photons, gluons, W, Z, Higgs?, . . . P̂ij ψ = −ψ → fermions: electrons, protons, neutrons, quarks, . . . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 11 / 101 Preliminaries Basic Concepts of Quantum Chemistry Wavefunction ψ: a complex function of 4N variables, no physical meaning. But: P (q1 ; q2 ; . . . ; qN ) = |ψ(q1 ; q2 ; . . . ; qN )|2 is the density probability of finding the electrons at positions q1 , q2 , . . . , qN . Indistinguishable particles → the exchange of any two particles can’t change the density probabily, so P̂ij P (q1 ; . . . ; qi ; . . . ; qj ; . . . ; qN ) = P (q1 ; . . . ; qj ; . . . ; qi ; . . . ; qN ). For electrons, the wavefunctions must be antisymmetric with respect to electron permutation: P̂ij ψ = −ψ. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 11 / 101 Preliminaries Basic Concepts of Quantum Chemistry Some remarks on the wavefunction Wavefunction is a fairly complicated object! For N -electron system it depends on 4N variables. For systems of biological importance this may boil down to several thousand variables . . . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 12 / 101 Preliminaries Basic Concepts of Quantum Chemistry Some remarks on the wavefunction Wavefunction is a fairly complicated object! For N -electron system it depends on 4N variables. For systems of biological importance this may boil down to several thousand variables . . . But Hamiltionian contains only one- and two-electron operators, since electrons don’t have internal structure (no many-body contributions). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 12 / 101 Preliminaries Basic Concepts of Quantum Chemistry Some remarks on the wavefunction Wavefunction is a fairly complicated object! For N -electron system it depends on 4N variables. For systems of biological importance this may boil down to several thousand variables . . . But Hamiltionian contains only one- and two-electron operators, since electrons don’t have internal structure (no many-body contributions). So, do we really need the state of the art, but incredibly expensive wavefunction? Is there something cheaper that would do the job we want, i.e. yield the energy and other properties? Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 12 / 101 Preliminaries Basic Concepts of Quantum Chemistry Some remarks on the wavefunction Wavefunction is a fairly complicated object! For N -electron system it depends on 4N variables. For systems of biological importance this may boil down to several thousand variables . . . But Hamiltionian contains only one- and two-electron operators, since electrons don’t have internal structure (no many-body contributions). So, do we really need the state of the art, but incredibly expensive wavefunction? Is there something cheaper that would do the job we want, i.e. yield the energy and other properties? Fortunately, the answer is yes. It’s the electron density — it’s simple, cheap, and you can buy it in Walmart. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 12 / 101 Preliminaries Electronic Distribution Electron density The probability of finding any electron anywhere must be 1, so a proper wavefunction should be normalized, giving 1 upon full integration: Z X Z ... |ψ(q1 ; . . . ; qN )|2 d3 r1 . . . d3 rN = 1. σ1 ,...,σN R3 R3 more on integration Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 13 / 101 Preliminaries Electronic Distribution Electron density The probability of finding any electron anywhere must be 1, so a proper wavefunction should be normalized, giving 1 upon full integration: Z X Z ... |ψ(q1 ; . . . ; qN )|2 d3 r1 . . . d3 rN = 1. σ1 ,...,σN R3 R3 If we perform the integration over all the spatial coordinates but one (arbitrarily chosen, say one) and over all spin variables, we get the well-known density distribution (the quantity measured in crystallography!): Z X Z ρ(r) = N ... |ψ(r; σ1 ; q2 ; . . . ; qN )|2 d3 r2 . . . d3 rN . σ1 ,σ2 ,...,σN Łukasz Rajchel (University of Warsaw) R3 R3 DFT Warsaw, 2010 13 / 101 Preliminaries Electronic Distribution Electron density The probability of finding any electron anywhere must be 1, so a proper wavefunction should be normalized, giving 1 upon full integration: Z X Z ... |ψ(q1 ; . . . ; qN )|2 d3 r1 . . . d3 rN = 1. σ1 ,...,σN R3 R3 ρ(r) is a 3D function and as such can’t be presented by a 3D graph. However, its isosurfaces, i.e. implicit functions ρ(r) = const > 0 may be plotted, e.g. for water: Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 13 / 101 Preliminaries Electronic Distribution Ground-state electron density: vanishes at infinity: lim ρ(r) = 0, r→∞ Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 14 / 101 Preliminaries Electronic Distribution Ground-state electron density: vanishes at infinity: lim ρ(r) = 0, r→∞ Z integrates to the number of electrons, ρ(r) d3 r = N , R3 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 14 / 101 Preliminaries Electronic Distribution Ground-state electron density: vanishes at infinity: lim ρ(r) = 0, r→∞ Z integrates to the number of electrons, ρ(r) d3 r = N , R3 has a finite value at nuclei positions and cusps in their neigbourhood (r → Rα ), Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 14 / 101 Preliminaries Electronic Distribution Ground-state electron density: vanishes at infinity: lim ρ(r) = 0, r→∞ Z integrates to the number of electrons, ρ(r) d3 r = N , R3 has a finite value at nuclei positions and cusps in their neigbourhood (r → Rα ), the cusp steepness keeps the information on nuclear charge: ∂ ρ(r)|Rα = −2Zα ρ(Rα ). ∂r Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 14 / 101 Preliminaries Electronic Distribution Ground-state electron density: vanishes at infinity: lim ρ(r) = 0, r→∞ Z integrates to the number of electrons, ρ(r) d3 r = N , R3 has a finite value at nuclei positions and cusps in their neigbourhood (r → Rα ), the cusp steepness keeps the information on nuclear charge: ∂ ρ(r)|Rα = −2Zα ρ(Rα ). ∂r The electron density already provides all the information on the molecule! Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 14 / 101 Preliminaries Electronic Distribution Expectation values Dynamical variable A (energy, momentum, velocity, time, . . . ) → operator  → expectation (mean) value of that operator: hAi = hψ|Â|ψi more on Dirac braket notation Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 15 / 101 Preliminaries Electronic Distribution Expectation values Dynamical variable A (energy, momentum, velocity, time, . . . ) → operator  → expectation (mean) value of that operator: hAi = hψ|Â|ψi ψ eigenfunction of  ⇒ Âψ = Aψ ⇒ hAi = A. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 15 / 101 Preliminaries Electronic Distribution Expectation values Dynamical variable A (energy, momentum, velocity, time, . . . ) → operator  → expectation (mean) value of that operator: hAi = hψ|Â|ψi ψ eigenfunction of  ⇒ Âψ = Aψ ⇒ hAi = A. Energy (E) operator → Hamiltonian (Ĥ). In practice we don’t know ψ satisfying Schrödinger equation, Ĥψ = Eψ, we only know Ĥ! Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 15 / 101 Preliminaries Electronic Distribution Expectation values Dynamical variable A (energy, momentum, velocity, time, . . . ) → operator  → expectation (mean) value of that operator: hAi = hψ|Â|ψi ψ eigenfunction of  ⇒ Âψ = Aψ ⇒ hAi = A. Energy (E) operator → Hamiltonian (Ĥ). In practice we don’t know ψ satisfying Schrödinger equation, Ĥψ = Eψ, we only know Ĥ! But there are cures for that (more — later on) . . . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 15 / 101 Preliminaries Electronic Distribution One-matrix Density function (one-matrix) is a generalization of the electronic density: Z X Z 0 ... ψ(r; σ1 ; q2 ; . . . ; qN ) × ρ(r; r ) = N σ1 ,σ2 ,...,σN R3 R3 × ψ ∗ (r0 ; σ1 ; q2 ; . . . ; qN ) d3 r2 . . . d3 rN . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 16 / 101 Preliminaries Electronic Distribution One-matrix Density function (one-matrix) is a generalization of the electronic density: Z X Z 0 ... ψ(r; σ1 ; q2 ; . . . ; qN ) × ρ(r; r ) = N R3 σ1 ,σ2 ,...,σN R3 × ψ ∗ (r0 ; σ1 ; q2 ; . . . ; qN ) d3 r2 . . . d3 rN . We need it to calculate expectation values of operators which are not simply multiplicative, e.g. kinetic energy: Z X Z T = hψ|T̂ |ψi = ... ψ ∗ (q1 ; . . . ; qN )× σ1 ,σ2 ,...,σN R3 R3 ! N 1X × − ∆ri ψ(q1 ; . . . ; qN ) d3 r1 . . . d3 rN = 2 Z i=1 1 =− ∆r ρ(r; r0 ) r0 =r d3 r. 2 R3 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 16 / 101 Preliminaries Electronic Distribution One-matrix Density function (one-matrix) is a generalization of the electronic density: Z X Z 0 ... ψ(r; σ1 ; q2 ; . . . ; qN ) × ρ(r; r ) = N R3 σ1 ,σ2 ,...,σN R3 × ψ ∗ (r0 ; σ1 ; q2 ; . . . ; qN ) d3 r2 . . . d3 rN . We need it to calculate expectation values of operators which are not simply multiplicative, e.g. kinetic energy: Z X Z T = hψ|T̂ |ψi = ... ψ ∗ (q1 ; . . . ; qN )× σ1 ,σ2 ,...,σN R3 R3 ! N 1X × − ∆ri ψ(q1 ; . . . ; qN ) d3 r1 . . . d3 rN = 2 Z i=1 1 =− ∆r ρ(r; r0 ) r0 =r d3 r. 2 R3 Electronic density is the diagonal part of one-matrix: ρ(r) = ρ(r; r). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 16 / 101 Preliminaries Electronic Distribution Pair density γ(r1 ; r2 ) = N (N − 1)× Z X Z ... |ψ(r1 ; σ1 ; r2 ; σ2 ; q3 ; . . . ; qN )|2 d3 r3 . . . d3 rN : × σ1 ,σ2 ,...,σN R3 Łukasz Rajchel (University of Warsaw) R3 DFT Warsaw, 2010 17 / 101 Preliminaries Electronic Distribution Pair density γ(r1 ; r2 ) = N (N − 1)× Z X Z ... |ψ(r1 ; σ1 ; r2 ; σ2 ; q3 ; . . . ; qN )|2 d3 r3 . . . d3 rN : × σ1 ,σ2 ,...,σN R3 R3 gives the probabilty distribution of any of two electrons being at r1 and r2 at the same time. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 17 / 101 Preliminaries Electronic Distribution Pair density γ(r1 ; r2 ) = N (N − 1)× Z X Z ... |ψ(r1 ; σ1 ; r2 ; σ2 ; q3 ; . . . ; qN )|2 d3 r3 . . . d3 rN : × σ1 ,σ2 ,...,σN R3 R3 gives the probabilty distribution of any of two electrons being at r1 and r2 at the same time. normalized to the number of non-distinct pairs, N (N − 1). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 17 / 101 Preliminaries Electronic Distribution Pair density γ(r1 ; r2 ) = N (N − 1)× Z X Z ... |ψ(r1 ; σ1 ; r2 ; σ2 ; q3 ; . . . ; qN )|2 d3 r3 . . . d3 rN : × σ1 ,σ2 ,...,σN R3 R3 gives the probabilty distribution of any of two electrons being at r1 and r2 at the same time. normalized to the number of non-distinct pairs, N (N − 1). yields density if integrated over one variable: Z γ(r1 ; r2 ) d3 r2 = (N − 1)ρ(r1 ). R3 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 17 / 101 Preliminaries Electronic Distribution Pair density γ(r1 ; r2 ) = N (N − 1)× Z X Z ... |ψ(r1 ; σ1 ; r2 ; σ2 ; q3 ; . . . ; qN )|2 d3 r3 . . . d3 rN : × σ1 ,σ2 ,...,σN R3 R3 gives the probabilty distribution of any of two electrons being at r1 and r2 at the same time. normalized to the number of non-distinct pairs, N (N − 1). yields density if integrated over one variable: Z γ(r1 ; r2 ) d3 r2 = (N − 1)ρ(r1 ). R3 is a measure of electron correlation, i.e. mutual interaction between electrons. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 17 / 101 Preliminaries Electronic Distribution Pair density γ(r1 ; r2 ) = N (N − 1)× Z X Z ... |ψ(r1 ; σ1 ; r2 ; σ2 ; q3 ; . . . ; qN )|2 d3 r3 . . . d3 rN : × σ1 ,σ2 ,...,σN R3 R3 gives the probabilty distribution of any of two electrons being at r1 and r2 at the same time. normalized to the number of non-distinct pairs, N (N − 1). yields density if integrated over one variable: Z γ(r1 ; r2 ) d3 r2 = (N − 1)ρ(r1 ). R3 is a measure of electron correlation, i.e. mutual interaction between electrons. but don’t confuse γ(r1 ; r2 ) (pair density) with ρ(r; r0 ) (one-matrix). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 17 / 101 Preliminaries Approximate Methods Variational principle We take any function ψ̃ depending on the same variables that the function ψ we are looking for and satisfying the usual boundary and antisymmetry conditions. Then hψ̃|Ĥ|ψ̃i = Ẽ ≥ E0 , where E0 — ground-state energy. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 18 / 101 Preliminaries Approximate Methods Variational principle We take any function ψ̃ depending on the same variables that the function ψ we are looking for and satisfying the usual boundary and antisymmetry conditions. Then hψ̃|Ĥ|ψ̃i = Ẽ ≥ E0 , where E0 — ground-state energy. This is the recipe for the quest for our wavefunction — find the function yielding the smallest energy. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 18 / 101 Preliminaries Approximate Methods Variational principle We take any function ψ̃ depending on the same variables that the function ψ we are looking for and satisfying the usual boundary and antisymmetry conditions. Then hψ̃|Ĥ|ψ̃i = Ẽ ≥ E0 , where E0 — ground-state energy. This is the recipe for the quest for our wavefunction — find the function yielding the smallest energy. Schematically, E0 = min hψ̃|Ĥ|ψ̃i . ψ̃→N Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 18 / 101 Outline of the Talk 1 DFT — A Real Celebrity 2 Preliminaries 3 Hartree-Fock Variation in HF Equations Correlation and exchange Self-Interaction in HF 4 Fermi and Coulomb Holes Hartree-Fock Variation in HF Hartree-Fock wavefunction The wavefunction assumed to be a single determinant functions (spinorbitals, φi ): φ1 (q1 ) φ2 (q1 ) 1 φ1 (q2 ) φ2 (q2 ) ψHF (q1 ; q2 ; . . . ; qN ) = √ . .. N ! .. . φ1 (qN ) φ2 (qN ) Łukasz Rajchel (University of Warsaw) DFT built of one-electron φN (q1 ) φN (q2 ) . .. . . . . φN (qN ) ... ... .. . Warsaw, 2010 20 / 101 Hartree-Fock Variation in HF Hartree-Fock wavefunction The wavefunction assumed to be a single determinant functions (spinorbitals, φi ): φ1 (q1 ) φ2 (q1 ) 1 φ1 (q2 ) φ2 (q2 ) ψHF (q1 ; q2 ; . . . ; qN ) = √ . .. N ! .. . φ1 (qN ) φ2 (qN ) Constraint: spinorbitals orthonormal, i.e. ( 1, hφi |φj i = δij = 0, Łukasz Rajchel (University of Warsaw) DFT built of one-electron φN (q1 ) φN (q2 ) . .. . . . . φN (qN ) ... ... .. . i = j, i 6= j. Warsaw, 2010 20 / 101 Hartree-Fock Variation in HF Hartree-Fock wavefunction The wavefunction assumed to be a single determinant functions (spinorbitals, φi ): φ1 (q1 ) φ2 (q1 ) 1 φ1 (q2 ) φ2 (q2 ) ψHF (q1 ; q2 ; . . . ; qN ) = √ . .. N ! .. . φ1 (qN ) φ2 (qN ) Constraint: spinorbitals orthonormal, i.e. ( 1, hφi |φj i = δij = 0, built of one-electron φN (q1 ) φN (q2 ) . .. . . . . φN (qN ) ... ... .. . i = j, i 6= j. Then, the wavefunction is antisymmetrical: P12 ψHF = −ψHF , and normalised: hψHF |ψHF i = 1. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 20 / 101 Hartree-Fock Variation in HF Orbitals and spinorbitals Spinorbitals and orbitals in the closed-shell restricted HF (RHF): spinorbital = orbital × spin function. ( φ2i−1 (r; σ) = ϕi (r)α(σ) . φ2i (r; σ) = ϕi (r)β(σ) Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 21 / 101 Hartree-Fock Variation in HF Orbitals and spinorbitals Spinorbitals and orbitals in the closed-shell restricted HF (RHF): spinorbital = orbital × spin function. ( φ2i−1 (r; σ) = ϕi (r)α(σ) . φ2i (r; σ) = ϕi (r)β(σ) Density function for ψ = ψHF : 0 ρ(r; r ) = 2 N/2 X ϕi (r)ϕ∗i (r0 ). i=1 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 21 / 101 Hartree-Fock Variation in HF Optimization of Hartree-Fock energy HF energy of a system: EHF = hψHF |Ĥ|ψHF i , and being a very diligent audience, we remember very well that Ĥ = T̂ + V̂ne + V̂ee + V̂nn . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 22 / 101 Hartree-Fock Variation in HF Optimization of Hartree-Fock energy HF energy of a system: EHF = hψHF |Ĥ|ψHF i , and being a very diligent audience, we remember very well that Ĥ = T̂ + V̂ne + V̂ee + V̂nn . HF energy in terms of density and density function: EHF [ρ] = T [ρ] + |{z} kinetic energy − Vne [ρ] | {z } + nuclear-electron attraction K[ρ] |{z} J[ρ] |{z} classical electrostatic electron-electron repulsion + non-classical electron-electron exchange interaction + Vnn |{z} . nuclear-nuclear repulsion (constant) what is a functional? how do these terms look like? Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 22 / 101 Hartree-Fock Variation in HF Optimization of Hartree-Fock energy HF energy of a system: EHF = hψHF |Ĥ|ψHF i , and being a very diligent audience, we remember very well that Ĥ = T̂ + V̂ne + V̂ee + V̂nn . HF energy in terms of density and density function: EHF [ρ] = T [ρ] + |{z} kinetic energy − Vne [ρ] | {z } + nuclear-electron attraction K[ρ] |{z} J[ρ] |{z} classical electrostatic electron-electron repulsion + non-classical electron-electron exchange interaction + Vnn |{z} . nuclear-nuclear repulsion (constant) Goal: minimize HF energy varying the orbitals ϕi (spatial parts of spinorbitals φi ) while keeping them orthonormal. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 22 / 101 Hartree-Fock Variation in HF Optimization of Hartree-Fock energy HF energy of a system: EHF = hψHF |Ĥ|ψHF i , and being a very diligent audience, we remember very well that Ĥ = T̂ + V̂ne + V̂ee + V̂nn . HF energy in terms of density and density function: EHF [ρ] = T [ρ] + |{z} kinetic energy − Vne [ρ] | {z } + nuclear-electron attraction K[ρ] |{z} J[ρ] |{z} classical electrostatic electron-electron repulsion + non-classical electron-electron exchange interaction + Vnn |{z} . nuclear-nuclear repulsion (constant) Result: HF equations for the best orbitals, i.e. orbitals yielding minimum HF energy. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 22 / 101 Hartree-Fock Equations Fock operator HF equations for best orbitals: fˆϕi = i ϕi . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 23 / 101 Hartree-Fock Equations Fock operator HF equations for best orbitals: fˆϕi = i ϕi . Fock operator: 1 fˆ(r) = − ∆r + v̂ne (r) + v̂HF (r). 2 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 23 / 101 Hartree-Fock Equations Fock operator HF equations for best orbitals: fˆϕi = i ϕi . Fock operator: 1 fˆ(r) = − ∆r + v̂ne (r) + v̂HF (r). 2 Nuclear potential: v̂ne (r) = − M X α=1 Łukasz Rajchel (University of Warsaw) DFT Zα . |r − Rα | Warsaw, 2010 23 / 101 Hartree-Fock Equations Fock operator HF equations for best orbitals: fˆϕi = i ϕi . Fock operator: 1 fˆ(r) = − ∆r + v̂ne (r) + v̂HF (r). 2 HF potential: the average repulsive potential experienced by the electron from to the remaining N − 1 electrons. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 23 / 101 Hartree-Fock Equations Fock operator HF equations for best orbitals: fˆϕi = i ϕi . Fock operator: 1 fˆ(r) = − ∆r + v̂ne (r) + v̂HF (r). 2 −1 The complicated two-electron repulsion operator rij in the Hamiltonian is replaced by the simple one-electron operator v̂HF (r), but now the electronelectron repulsion is taken into account only in an average way. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 23 / 101 Hartree-Fock Equations Coulomb and echange operators HF potential: Coulomb − exchange, v̂HF = ̂ − |{z} k̂ . |{z} Coulomb Łukasz Rajchel (University of Warsaw) DFT exchange Warsaw, 2010 24 / 101 Hartree-Fock Equations Coulomb and echange operators HF potential: Coulomb − exchange, ̂ − |{z} k̂ . |{z} v̂HF = Coulomb Z ̂(r)f (r) = R3 Łukasz Rajchel (University of Warsaw) exchange ρ(r0 ) 3 0 d r f (r) : |r − r0 | DFT Warsaw, 2010 24 / 101 Hartree-Fock Equations Coulomb and echange operators HF potential: Coulomb − exchange, ̂ − |{z} k̂ . |{z} v̂HF = Coulomb Z ̂(r)f (r) = R3 exchange ρ(r0 ) 3 0 d r f (r) : |r − r0 | the classical electrostatic interaction between electron at postion r with the charge density ρ. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 24 / 101 Hartree-Fock Equations Coulomb and echange operators HF potential: Coulomb − exchange, ̂ − |{z} k̂ . |{z} v̂HF = Coulomb Z ̂(r)f (r) = R3 exchange ρ(r0 ) 3 0 d r f (r) : |r − r0 | the classical electrostatic interaction between electron at postion r with the charge density ρ. its action on f (r) requires the knowledge of f value at r only ⇒ ̂(r) is local . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 24 / 101 Hartree-Fock Equations Coulomb and echange operators HF potential: Coulomb − exchange, v̂HF = ̂ − |{z} k̂ . |{z} Coulomb 1 k̂(r)f (r) = 2 Łukasz Rajchel (University of Warsaw) Z R3 exchange ρ(r; r0 ) f (r0 ) d3 r0 : |r − r0 | DFT Warsaw, 2010 24 / 101 Hartree-Fock Equations Coulomb and echange operators HF potential: Coulomb − exchange, v̂HF = ̂ − |{z} k̂ . |{z} Coulomb 1 k̂(r)f (r) = 2 Z R3 exchange ρ(r; r0 ) f (r0 ) d3 r0 : |r − r0 | non-classical and entirely due to the antisymmetry of the Slater determinant. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 24 / 101 Hartree-Fock Equations Coulomb and echange operators HF potential: Coulomb − exchange, v̂HF = ̂ − |{z} k̂ . |{z} Coulomb 1 k̂(r)f (r) = 2 Z R3 exchange ρ(r; r0 ) f (r0 ) d3 r0 : |r − r0 | non-classical and entirely due to the antisymmetry of the Slater determinant. its action on f (r) requires the knowledge of f value at all points in space (because of the integration) ⇒ k̂(r) is non-local . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 24 / 101 Hartree-Fock Correlation and exchange Pair densities and exchange in Hartree-Fock HF pair density function for the electron with opposite spins: γαβ (r1 ; r2 ) = ρα (r1 )ρβ (r2 ), Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 25 / 101 Hartree-Fock Correlation and exchange Pair densities and exchange in Hartree-Fock HF pair density function for the electron with opposite spins: γαβ (r1 ; r2 ) = ρα (r1 )ρβ (r2 ), and for the electron with the same spin: γαα (r1 ; r2 ) = ρα (r1 )ρα (r2 ) − ρα (r1 ; r2 )ρα (r2 ; r1 ), Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 25 / 101 Hartree-Fock Correlation and exchange Pair densities and exchange in Hartree-Fock HF pair density function for the electron with opposite spins: γαβ (r1 ; r2 ) = ρα (r1 )ρβ (r2 ), and for the electron with the same spin: γαα (r1 ; r2 ) = ρα (r1 )ρα (r2 ) − ρα (r1 ; r2 )ρα (r2 ; r1 ), Conclusions: Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 25 / 101 Hartree-Fock Correlation and exchange Pair densities and exchange in Hartree-Fock HF pair density function for the electron with opposite spins: γαβ (r1 ; r2 ) = ρα (r1 )ρβ (r2 ), and for the electron with the same spin: γαα (r1 ; r2 ) = ρα (r1 )ρα (r2 ) − ρα (r1 ; r2 )ρα (r2 ; r1 ), Conclusions: probability density of two electrons with opposite spins occupying some regions in space is just the product of probability densities of each of the two events occuring independently: no correlation. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 25 / 101 Hartree-Fock Correlation and exchange Pair densities and exchange in Hartree-Fock HF pair density function for the electron with opposite spins: γαβ (r1 ; r2 ) = ρα (r1 )ρβ (r2 ), and for the electron with the same spin: γαα (r1 ; r2 ) = ρα (r1 )ρα (r2 ) − ρα (r1 ; r2 )ρα (r2 ; r1 ), Conclusions: probability density of two electrons with opposite spins occupying some regions in space is just the product of probability densities of each of the two events occuring independently: no correlation. but probability density of two electrons with same spins occupying some regions in space is correlated and that density vanishes for r2 → r1 — this prevents the two electrons with like spins occupy the same region of space: it’s called Fermi correlation or exchange. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 25 / 101 Hartree-Fock Correlation and exchange Electron correlation ψHF is the best function within the one-electron approximation (each electron is ascribed to a spinorbital). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 26 / 101 Hartree-Fock Correlation and exchange Electron correlation ψHF is the best function within the one-electron approximation (each electron is ascribed to a spinorbital). But it is not the true wavefunction of the system (i.e. the one from Ĥψ = Eψ), and in most cases we don’t know that true function. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 26 / 101 Hartree-Fock Correlation and exchange Electron correlation ψHF is the best function within the one-electron approximation (each electron is ascribed to a spinorbital). But it is not the true wavefunction of the system (i.e. the one from Ĥψ = Eψ), and in most cases we don’t know that true function. Thus, due to the variational principle, HF energy of the system is always higher than its true energy, EHF > E. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 26 / 101 Hartree-Fock Correlation and exchange Electron correlation ψHF is the best function within the one-electron approximation (each electron is ascribed to a spinorbital). But it is not the true wavefunction of the system (i.e. the one from Ĥψ = Eψ), and in most cases we don’t know that true function. Thus, due to the variational principle, HF energy of the system is always higher than its true energy, EHF > E. The error introduced throgh the HF scheme is called the correlation energy: Ecor = E − EHF , and is always negative. We assume HF method misses any electron correlation (though it properly accounts for the exchange). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 26 / 101 Hartree-Fock Correlation and exchange Electron correlation ψHF is the best function within the one-electron approximation (each electron is ascribed to a spinorbital). But it is not the true wavefunction of the system (i.e. the one from Ĥψ = Eψ), and in most cases we don’t know that true function. Thus, due to the variational principle, HF energy of the system is always higher than its true energy, EHF > E. The error introduced throgh the HF scheme is called the correlation energy: Ecor = E − EHF , and is always negative. We assume HF method misses any electron correlation (though it properly accounts for the exchange). energy 6 EHF E Łukasz Rajchel (University of Warsaw) DFT 6 −Ecor ? Warsaw, 2010 26 / 101 Hartree-Fock Self-Interaction in HF Self-interaction problem Hydrogen atom has only one electron, so obviously there is no electron-electron interaction of any kind. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 27 / 101 Hartree-Fock Self-Interaction in HF Self-interaction problem Hydrogen atom has only one electron, so obviously there is no electron-electron interaction of any kind. Now, we all remember (because we are a diligent audience!) that EHF [ρ] = T [ρ] + Vne [ρ] + J[ρ] + Ex [ρ] + Vnn . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 27 / 101 Hartree-Fock Self-Interaction in HF Self-interaction problem Hydrogen atom has only one electron, so obviously there is no electron-electron interaction of any kind. Now, we all remember (because we are a diligent audience!) that EHF [ρ] = T [ρ] + Vne [ρ] + J[ρ] + Ex [ρ] + Vnn . Thus, the electron-electron repulsion in HF is J[ρ] + Ex [ρ]: Coulomb and exchange. What happens in hydrogen atom in HF picture? Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 27 / 101 Hartree-Fock Self-Interaction in HF Self-interaction problem Hydrogen atom has only one electron, so obviously there is no electron-electron interaction of any kind. Now, we all remember (because we are a diligent audience!) that EHF [ρ] = T [ρ] + Vne [ρ] + J[ρ] + Ex [ρ] + Vnn . Thus, the electron-electron repulsion in HF is J[ρ] + Ex [ρ]: Coulomb and exchange. What happens in hydrogen atom in HF picture? Let’s look at the numbers (from Molpro, QChem, Gaussian, . . . ): Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 27 / 101 Hartree-Fock Self-Interaction in HF Self-interaction problem Hydrogen atom has only one electron, so obviously there is no electron-electron interaction of any kind. Now, we all remember (because we are a diligent audience!) that EHF [ρ] = T [ρ] + Vne [ρ] + J[ρ] + Ex [ρ] + Vnn . Thus, the electron-electron repulsion in HF is J[ρ] + Ex [ρ]: Coulomb and exchange. What happens in hydrogen atom in HF picture? Let’s look at the numbers (from Molpro, QChem, Gaussian, . . . ): T [ρ] + Vne [ρ] = −0.49999, Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 27 / 101 Hartree-Fock Self-Interaction in HF Self-interaction problem Hydrogen atom has only one electron, so obviously there is no electron-electron interaction of any kind. Now, we all remember (because we are a diligent audience!) that EHF [ρ] = T [ρ] + Vne [ρ] + J[ρ] + Ex [ρ] + Vnn . Thus, the electron-electron repulsion in HF is J[ρ] + Ex [ρ]: Coulomb and exchange. What happens in hydrogen atom in HF picture? Let’s look at the numbers (from Molpro, QChem, Gaussian, . . . ): T [ρ] + Vne [ρ] = −0.49999, J[ρ] = 0.31250, Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 27 / 101 Hartree-Fock Self-Interaction in HF Self-interaction problem Hydrogen atom has only one electron, so obviously there is no electron-electron interaction of any kind. Now, we all remember (because we are a diligent audience!) that EHF [ρ] = T [ρ] + Vne [ρ] + J[ρ] + Ex [ρ] + Vnn . Thus, the electron-electron repulsion in HF is J[ρ] + Ex [ρ]: Coulomb and exchange. What happens in hydrogen atom in HF picture? Let’s look at the numbers (from Molpro, QChem, Gaussian, . . . ): T [ρ] + Vne [ρ] = −0.49999, J[ρ] = 0.31250, Ex [ρ] = −0.31250, Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 27 / 101 Hartree-Fock Self-Interaction in HF Self-interaction problem Hydrogen atom has only one electron, so obviously there is no electron-electron interaction of any kind. Now, we all remember (because we are a diligent audience!) that EHF [ρ] = T [ρ] + Vne [ρ] + J[ρ] + Ex [ρ] + Vnn . Thus, the electron-electron repulsion in HF is J[ρ] + Ex [ρ]: Coulomb and exchange. What happens in hydrogen atom in HF picture? Let’s look at the numbers (from Molpro, QChem, Gaussian, . . . ): T [ρ] + Vne [ρ] = −0.49999, J[ρ] = 0.31250, Ex [ρ] = −0.31250, so, J[ρ] + Ex [ρ] = 0. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 27 / 101 Hartree-Fock Self-Interaction in HF Self-interaction problem Hydrogen atom has only one electron, so obviously there is no electron-electron interaction of any kind. Now, we all remember (because we are a diligent audience!) that EHF [ρ] = T [ρ] + Vne [ρ] + J[ρ] + Ex [ρ] + Vnn . Thus, the electron-electron repulsion in HF is J[ρ] + Ex [ρ]: Coulomb and exchange. What happens in hydrogen atom in HF picture? Let’s look at the numbers (from Molpro, QChem, Gaussian, . . . ): T [ρ] + Vne [ρ] = −0.49999, J[ρ] = 0.31250, Ex [ρ] = −0.31250, so, J[ρ] + Ex [ρ] = 0. There is no self-interaction in HF! The unphysical self-interaction of electron with itself contained in J[ρ] is removed by Ex [ρ]. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 27 / 101 Outline of the Talk 1 DFT — A Real Celebrity 2 Preliminaries 3 Hartree-Fock 4 Fermi and Coulomb Holes Definitions Fermi and Coulomb Holes Definitions Electron-electron repulsion The source of all troubles and misfortunes (but also a nice grant-generator) in quantum chemistry: electron-electron repulsion operator, V̂ee = N −1 X N X 1 . rij i=1 j=i+1 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 29 / 101 Fermi and Coulomb Holes Definitions Electron-electron repulsion The source of all troubles and misfortunes (but also a nice grant-generator) in quantum chemistry: electron-electron repulsion operator, V̂ee = N −1 X N X 1 . rij i=1 j=i+1 Exact wavefunction ψ → exact pair density γ → exact e-e repulsion: Z Z 1 γ(r1 ; r2 ) 3 d r1 d3 r2 . Eee = hψ|V̂ee |ψi = 2 R3 R3 r12 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 29 / 101 Fermi and Coulomb Holes Definitions Electron-electron repulsion The source of all troubles and misfortunes (but also a nice grant-generator) in quantum chemistry: electron-electron repulsion operator, V̂ee = N −1 X N X 1 . rij i=1 j=i+1 Exact wavefunction ψ → exact pair density γ → exact e-e repulsion: Z Z 1 γ(r1 ; r2 ) 3 d r1 d3 r2 . Eee = hψ|V̂ee |ψi = 2 R3 R3 r12 In HF the e-e repulsion is Z Z 1 ρ(r1 )ρ(r2 ) 1 ρ(r1 ; r2 )ρ(r2 ; r1 ) J[ρ] + Ex [ρ] = − d3 r1 d3 r2 . 2 R3 R3 r12 2 r12 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 29 / 101 Fermi and Coulomb Holes Definitions Correlation factor So, in HF we reduce the Devil (e-e repulsion) as: 1 γHF (r1 ; r2 ) = ρ(r1 )ρ(r2 ) − ρ(r1 ; r2 )ρ(r2 ; r1 ). 2 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 30 / 101 Fermi and Coulomb Holes Definitions Correlation factor So, in HF we reduce the Devil (e-e repulsion) as: 1 γHF (r1 ; r2 ) = ρ(r1 )ρ(r2 ) − ρ(r1 ; r2 )ρ(r2 ; r1 ). 2 Clearly, in HF there’s some correlation included, otherwise γ(r1 ; r2 ) would simply decompose to ρ(r1 )ρ(r2 ). We already know HF accounts for the Fermi correlation. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 30 / 101 Fermi and Coulomb Holes Definitions Correlation factor So, in HF we reduce the Devil (e-e repulsion) as: 1 γHF (r1 ; r2 ) = ρ(r1 )ρ(r2 ) − ρ(r1 ; r2 )ρ(r2 ; r1 ). 2 Clearly, in HF there’s some correlation included, otherwise γ(r1 ; r2 ) would simply decompose to ρ(r1 )ρ(r2 ). We already know HF accounts for the Fermi correlation. Let’s generalize and write γ(r1 ; r2 ) = ρ(r1 )ρ(r2 ) 1 + f (r1 ; r2 ) , thus f (r1 ; r2 ) = 0 refers to uncorrelated case. f (r1 ; r2 ) — correlation factor. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 30 / 101 Fermi and Coulomb Holes Definitions Correlation factor So, in HF we reduce the Devil (e-e repulsion) as: 1 γHF (r1 ; r2 ) = ρ(r1 )ρ(r2 ) − ρ(r1 ; r2 )ρ(r2 ; r1 ). 2 Clearly, in HF there’s some correlation included, otherwise γ(r1 ; r2 ) would simply decompose to ρ(r1 )ρ(r2 ). We already know HF accounts for the Fermi correlation. Let’s generalize and write γ(r1 ; r2 ) = ρ(r1 )ρ(r2 ) 1 + f (r1 ; r2 ) , thus f (r1 ; r2 ) = 0 refers to uncorrelated case. f (r1 ; r2 ) — correlation factor. For HF we easily get f (r1 ; r2 ) = − Łukasz Rajchel (University of Warsaw) 1 ρ(r1 ; r2 )ρ(r2 ; r1 ) . 2 ρ(r1 )ρ(r2 ) DFT Warsaw, 2010 30 / 101 Fermi and Coulomb Holes Definitions Conditional probability The probability of A under the condition B: P (A|B) = P (A ∩ B) , P (B) P (A ∩ B) is the probability of both events together. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 31 / 101 Fermi and Coulomb Holes Definitions Conditional probability The probability of A under the condition B: P (A|B) = P (A ∩ B) , P (B) P (A ∩ B) is the probability of both events together. Eo ipso, γ(r1 ; r2 ) Ω(r2 |r1 ) = ρ(r1 ) is the probability density of finding any electron at r2 if there is one already known to be at r1 . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 31 / 101 Fermi and Coulomb Holes Definitions Conditional probability The probability of A under the condition B: P (A|B) = P (A ∩ B) , P (B) P (A ∩ B) is the probability of both events together. Eo ipso, γ(r1 ; r2 ) Ω(r2 |r1 ) = ρ(r1 ) is the probability density of finding any electron at r2 if there is one already known to be at r1 . If we integrate over all coordinates of electron 2, we get Z (N − 1)ρ(r1 ) Ω(r2 |r1 ) d3 r2 = = N − 1, ρ(r1 ) 3 R the number of all electrons of the systems but our reference one (which sits at r1 ). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 31 / 101 Fermi and Coulomb Holes Definitions Exchange-correlation hole Short recapitulation: exact pair density, γ(r1 ; r2 ), would include all previously mentioned effects: self-interaction, correlation, exchange; Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 32 / 101 Fermi and Coulomb Holes Definitions Exchange-correlation hole Short recapitulation: exact pair density, γ(r1 ; r2 ), would include all previously mentioned effects: self-interaction, correlation, exchange; but we don’t know it. For instance, HF pair density takes care of self-interaction and exchange, but misses any correlation. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 32 / 101 Fermi and Coulomb Holes Definitions Exchange-correlation hole Short recapitulation: exact pair density, γ(r1 ; r2 ), would include all previously mentioned effects: self-interaction, correlation, exchange; but we don’t know it. For instance, HF pair density takes care of self-interaction and exchange, but misses any correlation. Let’s now introduce the quantity: hxc (r1 ; r2 ) = Ω(r2 |r1 ) − ρ(r2 ) = ρ(r2 )f (r1 ; r2 ). It’s obviously the difference between conditional probability density of finding any electron at r2 if there is one already known to be at r1 and the uncorrelated probabily density of finding any electron at r2 . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 32 / 101 Fermi and Coulomb Holes Definitions Exchange-correlation hole Short recapitulation: exact pair density, γ(r1 ; r2 ), would include all previously mentioned effects: self-interaction, correlation, exchange; but we don’t know it. For instance, HF pair density takes care of self-interaction and exchange, but misses any correlation. Let’s now introduce the quantity: hxc (r1 ; r2 ) = Ω(r2 |r1 ) − ρ(r2 ) = ρ(r2 )f (r1 ; r2 ). It’s obviously the difference between conditional probability density of finding any electron at r2 if there is one already known to be at r1 and the uncorrelated probabily density of finding any electron at r2 . The conditional probability density is likely lower than the independent one, so hxc (r1 ; r2 ) is called the exchange-correlation hole (xc hole). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 32 / 101 Fermi and Coulomb Holes Definitions Exchange-correlation hole Short recapitulation: exact pair density, γ(r1 ; r2 ), would include all previously mentioned effects: self-interaction, correlation, exchange; but we don’t know it. For instance, HF pair density takes care of self-interaction and exchange, but misses any correlation. Let’s now introduce the quantity: hxc (r1 ; r2 ) = Ω(r2 |r1 ) − ρ(r2 ) = ρ(r2 )f (r1 ; r2 ). It’s obviously the difference between conditional probability density of finding any electron at r2 if there is one already known to be at r1 and the uncorrelated probabily density of finding any electron at r2 . Z Because we love integrals, let’s integrate: hxc (r1 ; r2 ) d3 r2 = −1: xc R3 hole contains exactly the charge of one electron. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 32 / 101 Fermi and Coulomb Holes Definitions Fermi and Coulomb holes The xc hole can be split into the Fermi and Coulomb holes: hxc (r1 ; r2 ) = hx (r1 ; r2 ) + hc (r1 ; r2 ). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 33 / 101 Fermi and Coulomb Holes Definitions Fermi and Coulomb holes The xc hole can be split into the Fermi and Coulomb holes: hxc (r1 ; r2 ) = hx (r1 ; r2 ) + hc (r1 ; r2 ). Fermi hole: applies to electrons with the same spin. integrates to −1. takes care of the self-interaction problem. ensures the Pauli principle is fulfilled (no two electron with the same spin in the same point of space). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 33 / 101 Fermi and Coulomb Holes Definitions Fermi and Coulomb holes The xc hole can be split into the Fermi and Coulomb holes: hxc (r1 ; r2 ) = hx (r1 ; r2 ) + hc (r1 ; r2 ). Fermi hole: applies to electrons with the same spin. Coulomb hole: applies to all electrons. integrates to 0. integrates to −1. ensures the cusp condition is fulfilled. takes care of the self-interaction problem. is dominated by the Fermi hole. ensures the Pauli principle is fulfilled (no two electron with the same spin in the same point of space). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 33 / 101 Fermi and Coulomb Holes Definitions xc hole for H2 Pictorially, we can imagine that electron digs a hole around itself so that the probability of finding another electron around it is diminished. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 34 / 101 Fermi and Coulomb Holes Definitions xc hole for H2 Pictorially, we can imagine that electron digs a hole around itself so that the probability of finding another electron around it is diminished. The reference electron is 0.3 bohr to the left from the right proton. Only the total xc hole has a physical sense. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 34 / 101 Fermi and Coulomb Holes Definitions Back to electron-electron repulsion again Using the xc hole we’ve just made friends with, we can write γ(r1 ; r2 ) = ρ(r1 )ρ(r2 ) + ρ(r1 )hxc (r1 ; r2 ), Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 35 / 101 Fermi and Coulomb Holes Definitions Back to electron-electron repulsion again Using the xc hole we’ve just made friends with, we can write γ(r1 ; r2 ) = ρ(r1 )ρ(r2 ) + ρ(r1 )hxc (r1 ; r2 ), so the e-e repulsion becomes Z Z Z Z γ(r1 ; r2 ) 3 1 ρ(r1 )ρ(r2 ) 3 1 3 d r1 d r2 = d r1 d3 r2 + Eee = 2 R3 R3 r12 2 R3 R3 r12 Z Z 1 ρ(r1 )hxc (r1 ; r2 ) 3 + d r1 d3 r2 = J[ρ] + Encl [ρ] : 2 R3 R3 r12 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 35 / 101 Fermi and Coulomb Holes Definitions Back to electron-electron repulsion again Using the xc hole we’ve just made friends with, we can write γ(r1 ; r2 ) = ρ(r1 )ρ(r2 ) + ρ(r1 )hxc (r1 ; r2 ), so the e-e repulsion becomes Z Z Z Z γ(r1 ; r2 ) 3 1 ρ(r1 )ρ(r2 ) 3 1 3 d r1 d r2 = d r1 d3 r2 + Eee = 2 R3 R3 r12 2 R3 R3 r12 Z Z 1 ρ(r1 )hxc (r1 ; r2 ) 3 + d r1 d3 r2 = J[ρ] + Encl [ρ] : 2 R3 R3 r12 J[ρ]: classical electrostatic energy of a charge distribution with itself, it contains unphysical self-interaction (remember — H atom). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 35 / 101 Fermi and Coulomb Holes Definitions Back to electron-electron repulsion again Using the xc hole we’ve just made friends with, we can write γ(r1 ; r2 ) = ρ(r1 )ρ(r2 ) + ρ(r1 )hxc (r1 ; r2 ), so the e-e repulsion becomes Z Z Z Z γ(r1 ; r2 ) 3 1 ρ(r1 )ρ(r2 ) 3 1 3 d r1 d r2 = d r1 d3 r2 + Eee = 2 R3 R3 r12 2 R3 R3 r12 Z Z 1 ρ(r1 )hxc (r1 ; r2 ) 3 + d r1 d3 r2 = J[ρ] + Encl [ρ] : 2 R3 R3 r12 J[ρ]: classical electrostatic energy of a charge distribution with itself, it contains unphysical self-interaction (remember — H atom). Encl [ρ]: interaction between the charge density and the charge distribution of the xc hole. It includes the correction for the self-interaction and all contributions of quantum-mechanical (non-classical) correlation effects. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 35 / 101 Summary Points to remember: The density gives all the information on the molecule. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 36 / 101 Summary Points to remember: The density gives all the information on the molecule. Hartree-Fock method treats electron-electron repulsion in a very simplified manner: it properly accounts for the exchange, but lacks any Coulomb correlation. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 36 / 101 Summary Points to remember: The density gives all the information on the molecule. Hartree-Fock method treats electron-electron repulsion in a very simplified manner: it properly accounts for the exchange, but lacks any Coulomb correlation. But Hartree-Fock properly deals with the self-interaction problem. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 36 / 101 Summary Points to remember: The density gives all the information on the molecule. Hartree-Fock method treats electron-electron repulsion in a very simplified manner: it properly accounts for the exchange, but lacks any Coulomb correlation. But Hartree-Fock properly deals with the self-interaction problem. xc hole is a nice concept allowing for the separation of electron-electron repulsion into the classical and non-classical parts. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 36 / 101 Summary Points to remember: The density gives all the information on the molecule. Hartree-Fock method treats electron-electron repulsion in a very simplified manner: it properly accounts for the exchange, but lacks any Coulomb correlation. But Hartree-Fock properly deals with the self-interaction problem. xc hole is a nice concept allowing for the separation of electron-electron repulsion into the classical and non-classical parts. But the non-classical part takes a lot of responsibility: it has to account for Coulomb and Fermi types of correlation and to remove the unphysical self-interaction included in the classical part. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 36 / 101 Summary Points to remember: The density gives all the information on the molecule. Hartree-Fock method treats electron-electron repulsion in a very simplified manner: it properly accounts for the exchange, but lacks any Coulomb correlation. But Hartree-Fock properly deals with the self-interaction problem. xc hole is a nice concept allowing for the separation of electron-electron repulsion into the classical and non-classical parts. But the non-classical part takes a lot of responsibility: it has to account for Coulomb and Fermi types of correlation and to remove the unphysical self-interaction included in the classical part. The End (for today) Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 36 / 101 Part II DFT: How It’s Made Outline of the Talk 5 Density and Energy Remarks and Problems Historical Models Results 6 Hohenberg-Kohn Theorems 7 Kohn-Sham Approach 8 xc Functionals Density and Energy Remarks and Problems Energy of a molecule How to get the energy of a molecule — a kosher recipe: specify molecule’s geometry: positions of nuclei {Rα }M α=1 , Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 39 / 101 Density and Energy Remarks and Problems Energy of a molecule How to get the energy of a molecule — a kosher recipe: specify molecule’s geometry: positions of nuclei {Rα }M α=1 , specify molecular P charge: N — number of electrons, the total charge Q = M α=1 Zα − N , Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 39 / 101 Density and Energy Remarks and Problems Energy of a molecule How to get the energy of a molecule — a kosher recipe: specify molecule’s geometry: positions of nuclei {Rα }M α=1 , specify molecular P charge: N — number of electrons, the total charge Q = M α=1 Zα − N , write the total hamiltonian: Ĥ = T̂ + V̂ne + V̂ee + V̂nn , Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 39 / 101 Density and Energy Remarks and Problems Energy of a molecule How to get the energy of a molecule — a kosher recipe: specify molecule’s geometry: positions of nuclei {Rα }M α=1 , specify molecular P charge: N — number of electrons, the total charge Q = M α=1 Zα − N , write the total hamiltonian: Ĥ = T̂ + V̂ne + V̂ee + V̂nn , solve the Schrödinger equation, Ĥψ0 = E0 ψ0 : we’ve got the energy. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 39 / 101 Density and Energy Remarks and Problems Energy of a molecule How to get the energy of a molecule — a kosher recipe: specify molecule’s geometry: positions of nuclei {Rα }M α=1 , specify molecular P charge: N — number of electrons, the total charge Q = M α=1 Zα − N , write the total hamiltonian: Ĥ = T̂ + V̂ne + V̂ee + V̂nn , solve the Schrödinger equation, Ĥψ0 = E0 ψ0 : we’ve got the energy. But we don’t need ψ — one-matrix ρ and pair density γ suffice: E = hψ|Ĥ|ψi = T [ρ] + Vne [ρ] + Eee [γ] + Vnn = Z Z 1 0 3 ∆r ρ(r; r ) r0 =r d r + vne (r)ρ(r) d3 r + = − 2 R3 3 R Z Z M −1 X M X Zα Zβ 1 γ(r1 ; r2 ) 3 + d r1 d3 r2 + . 2 R3 R3 r12 Rαβ α=1 β=α+1 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 39 / 101 Density and Energy Remarks and Problems Energy of a molecule How to get the energy of a molecule — a kosher recipe: specify molecule’s geometry: positions of nuclei {Rα }M α=1 , specify molecular P charge: N — number of electrons, the total charge Q = M α=1 Zα − N , write the total hamiltonian: Ĥ = T̂ + V̂ne + V̂ee + V̂nn , solve the Schrödinger equation, Ĥψ0 = E0 ψ0 : we’ve got the energy. e-e repulsion can be separated into classical interaction of charge density with itself (with unphysical self-interaction) and the interaction of charge density with the xc hole, containing all non-classical effects (correlation, exchange, correction for self-interaction): Eee = J[ρ] + Encl [ρ] = Z Z Z Z 1 ρ(r1 )ρ(r2 ) 3 1 ρ(r1 )hxc (r1 ; r2 ) 3 d r1 d3 r2 + d r1 d3 r2 . 2 R3 R3 r12 2 R3 R3 r12 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 39 / 101 Density and Energy Remarks and Problems Wavefunction and density Question : can we replace ψ(q1 ; . . . ; qN ), depending on 4N variables, with ρ(r), depending on just 3 variables? Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 40 / 101 Density and Energy Remarks and Problems Wavefunction and density Question : can we replace ψ(q1 ; . . . ; qN ), depending on 4N variables, with ρ(r), depending on just 3 variables? Ground-state density: Hamiltonian uniquely defined by: Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 40 / 101 Density and Energy Remarks and Problems Wavefunction and density Question : can we replace ψ(q1 ; . . . ; qN ), depending on 4N variables, with ρ(r), depending on just 3 variables? Ground-state density: Hamiltonian uniquely defined by: the number of electrons, Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 40 / 101 Density and Energy Remarks and Problems Wavefunction and density Question : can we replace ψ(q1 ; . . . ; qN ), depending on 4N variables, with ρ(r), depending on just 3 variables? Ground-state density: integrates to the number of electrons. Hamiltonian uniquely defined by: the number of electrons, Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 40 / 101 Density and Energy Remarks and Problems Wavefunction and density Question : can we replace ψ(q1 ; . . . ; qN ), depending on 4N variables, with ρ(r), depending on just 3 variables? Ground-state density: integrates to the number of electrons. Hamiltonian uniquely defined by: the number of electrons, the position of the nuclei, and Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 40 / 101 Density and Energy Remarks and Problems Wavefunction and density Question : can we replace ψ(q1 ; . . . ; qN ), depending on 4N variables, with ρ(r), depending on just 3 variables? Ground-state density: integrates to the number of electrons. Hamiltonian uniquely defined by: the number of electrons, the position of the nuclei, and Łukasz Rajchel (University of Warsaw) has cusps at the position of the nuclei. DFT Warsaw, 2010 40 / 101 Density and Energy Remarks and Problems Wavefunction and density Question : can we replace ψ(q1 ; . . . ; qN ), depending on 4N variables, with ρ(r), depending on just 3 variables? Ground-state density: integrates to the number of electrons. Hamiltonian uniquely defined by: the number of electrons, the position of the nuclei, and has cusps at the position of the nuclei. the charges of the nuclei. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 40 / 101 Density and Energy Remarks and Problems Wavefunction and density Question : can we replace ψ(q1 ; . . . ; qN ), depending on 4N variables, with ρ(r), depending on just 3 variables? Ground-state density: integrates to the number of electrons. Hamiltonian uniquely defined by: the number of electrons, the position of the nuclei, and has cusps at the position of the nuclei. the charges of the nuclei. the cusp steepness is intimately related to the charge of the nucleus. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 40 / 101 Density and Energy Remarks and Problems Wavefunction and density Question : can we replace ψ(q1 ; . . . ; qN ), depending on 4N variables, with ρ(r), depending on just 3 variables? Ground-state density: integrates to the number of electrons. Hamiltonian uniquely defined by: the number of electrons, the position of the nuclei, and has cusps at the position of the nuclei. the charges of the nuclei. the cusp steepness is intimately related to the charge of the nucleus. Answer : yes! The ground-state density provides us with all the information we need to solve the Schrödinger equation. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 40 / 101 Density and Energy Remarks and Problems Problems with energy Problems with calculation of the energy of the system, E = T [ρ] + Vne [ρ] + Eee [γ] + Vne : Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 41 / 101 Density and Energy Remarks and Problems Problems with energy Problems with calculation of the energy of the system, E = T [ρ] + Vne [ρ] + Eee [γ] + Vne : the rigorous expression for the kinetic Z energy employs the one-matrix 1 instead of the density: T [ρ] = − ∆r ρ(r; r0 ) r0 =r d3 r. 2 R3 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 41 / 101 Density and Energy Remarks and Problems Problems with energy Problems with calculation of the energy of the system, E = T [ρ] + Vne [ρ] + Eee [γ] + Vne : the rigorous expression for the kinetic Z energy employs the one-matrix 1 instead of the density: T [ρ] = − ∆r ρ(r; r0 ) r0 =r d3 r. 2 R3 the notorious e-e repulsion term which on the pair density instead of Z Z 1 γ(r1 ; r2 ) 3 the density alone: Eee [γ] = d r1 d3 r2 . 2 R3 R3 r12 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 41 / 101 Density and Energy Remarks and Problems Problems with energy Problems with calculation of the energy of the system, E = T [ρ] + Vne [ρ] + Eee [γ] + Vne : the rigorous expression for the kinetic Z energy employs the one-matrix 1 instead of the density: T [ρ] = − ∆r ρ(r; r0 ) r0 =r d3 r. 2 R3 the notorious e-e repulsion term which on the pair density instead of Z Z 1 γ(r1 ; r2 ) 3 the density alone: Eee [γ] = d r1 d3 r2 . 2 R3 R3 r12 So, formally density is not enough — we need the one-matrix and pair-density to calculate E. . . On the other hand, we’ve already learnt that the density yields all the information on the system. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 41 / 101 Density and Energy Historical Models Thomas-Fermi model Thomas and Fermi (1920s) were the first to give an approximate expression of the energy in therms of the density only. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 42 / 101 Density and Energy Historical Models Thomas-Fermi model Thomas and Fermi (1920s) were the first to give an approximate expression of the energy in therms of the density only. Assumptions of the Thomas-Fermi model: Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 42 / 101 Density and Energy Historical Models Thomas-Fermi model Thomas and Fermi (1920s) were the first to give an approximate expression of the energy in therms of the density only. Assumptions of the Thomas-Fermi model: the kinetic energy functional is taken from the theory of uniform noninteracting electron gas. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 42 / 101 Density and Energy Historical Models Thomas-Fermi model Thomas and Fermi (1920s) were the first to give an approximate expression of the energy in therms of the density only. Assumptions of the Thomas-Fermi model: the kinetic energy functional is taken from the theory of uniform noninteracting electron gas. the electronic exchange and correlations effects are completely neglected, the electron-electron repulsion and electron-nucleus attraction are treated in a classical way only. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 42 / 101 Density and Energy Historical Models Thomas-Fermi model Thomas and Fermi (1920s) were the first to give an approximate expression of the energy in therms of the density only. Assumptions of the Thomas-Fermi model: the kinetic energy functional is taken from the theory of uniform noninteracting electron gas. the electronic exchange and correlations effects are completely neglected, the electron-electron repulsion and electron-nucleus attraction are treated in a classical way only. The energy functional in TF model reads E[ρ] = TTF [ρ] + Vne [ρ] + J[ρ] + Vnn , Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 42 / 101 Density and Energy Historical Models Thomas-Fermi model Thomas and Fermi (1920s) were the first to give an approximate expression of the energy in therms of the density only. Assumptions of the Thomas-Fermi model: the kinetic energy functional is taken from the theory of uniform noninteracting electron gas. the electronic exchange and correlations effects are completely neglected, the electron-electron repulsion and electron-nucleus attraction are treated in a classical way only. The energy functional in TF model reads E[ρ] = TTF [ρ] + Vne [ρ] + J[ρ] + Vnn , Z TTF [ρ] = CF ρ5/3 (r) d3 r. R3 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 42 / 101 Density and Energy Historical Models Thomas-Fermi-Dirac model In the Thomas-Fermi-Dirac model the Thomas-Fermi energy functional E[ρ] = TTF [ρ] + Vne [ρ] + J[ρ] + Łukasz Rajchel (University of Warsaw) DFT +Vnn . Warsaw, 2010 43 / 101 Density and Energy Historical Models Thomas-Fermi-Dirac model In the Thomas-Fermi-Dirac model the Thomas-Fermi energy functional is supplemented with the exchange energy taken from the theory of uniform noninteracting electron gas, as was the case for the kinetic energy: E[ρ] = TTF [ρ] + Vne [ρ] + J[ρ] + Ex [ρ]+Vnn . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 43 / 101 Density and Energy Historical Models Thomas-Fermi-Dirac model In the Thomas-Fermi-Dirac model the Thomas-Fermi energy functional is supplemented with the exchange energy taken from the theory of uniform noninteracting electron gas, as was the case for the kinetic energy: E[ρ] = TTF [ρ] + Vne [ρ] + J[ρ] + Ex [ρ]+Vnn . Z Ex [ρ] = −Cx ρ4/3 (r) d3 r. R3 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 43 / 101 Density and Energy Historical Models Thomas-Fermi-Dirac model In the Thomas-Fermi-Dirac model the Thomas-Fermi energy functional is supplemented with the exchange energy taken from the theory of uniform noninteracting electron gas, as was the case for the kinetic energy: E[ρ] = TTF [ρ] + Vne [ρ] + J[ρ] + Ex [ρ]+Vnn . Z Ex [ρ] = −Cx ρ4/3 (r) d3 r. R3 The same formula for the exchange was derived by Slater in 1950s based on the assumption of the Fermi hole being spherically symmetric around the reference electron. That expression, depending only on local values of electron density, replaced the original non-local Hartree-Fock formula: Z Z ρ(r1 )hx (r1 ; r2 ) 3 1 ExHF [ρ] = d r1 d3 r2 . 2 R3 R3 r12 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 43 / 101 Density and Energy Historical Models Thomas-Fermi-Dirac model In the Thomas-Fermi-Dirac model the Thomas-Fermi energy functional is supplemented with the exchange energy taken from the theory of uniform noninteracting electron gas, as was the case for the kinetic energy: E[ρ] = TTF [ρ] + Vne [ρ] + J[ρ] + Ex [ρ]+Vnn . Z Ex [ρ] = −Cx ρ4/3 (r) d3 r. R3 The same formula for the exchange was derived by Slater in 1950s based on the assumption of the Fermi hole being spherically symmetric around the reference electron. That expression, depending only on local values of electron density, replaced the original non-local Hartree-Fock formula: Z Z ρ(r1 ; r1 )ρ(r2 ; r1 ) 3 1 ExHF [ρ] = − d r1 d3 r2 . 4 R3 R3 r12 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 43 / 101 Density and Energy Results Some results . . . Both TF and TFD models are based on the theory of uniform noninteracting electron gas. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 44 / 101 Density and Energy Results Some results . . . Both TF and TFD models are based on the theory of uniform noninteracting electron gas. But the electronic density in an atom or molecule is obviously not uniform at all. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 44 / 101 Density and Energy Results Some results . . . Both TF and TFD models are based on the theory of uniform noninteracting electron gas. But the electronic density in an atom or molecule is obviously not uniform at all. That has drastic consequences — for instance, TF and TFD models do not allow for any chemical bonding! Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 44 / 101 Density and Energy Results Some results . . . Both TF and TFD models are based on the theory of uniform noninteracting electron gas. But the electronic density in an atom or molecule is obviously not uniform at all. That has drastic consequences — for instance, TF and TFD models do not allow for any chemical bonding! For atoms the TFD model results are not too bad: Atom He Ne Ar Kr Xe Rn −EHF 2.8615 128.5551 526.7942 2752.0164 7232.4982 21866.2779 −ETFD 2.2159 124.1601 518.8124 2755.4398 7273.2788 22019.7140 Source: [Parr and Yang(1989)] Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 44 / 101 Outline of the Talk 5 Density and Energy 6 Hohenberg-Kohn Theorems Definitions The Theorems Representability of the Density 7 Kohn-Sham Approach 8 xc Functionals Hohenberg-Kohn Theorems Definitions External potential The external potential — potential vext acting on electrons the source of which are not electrons themselves. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 46 / 101 Hohenberg-Kohn Theorems Definitions External potential The external potential — potential vext acting on electrons the source of which are not electrons themselves. Ĥ = T̂ + V̂ext + V̂ee + V̂nn , Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 46 / 101 Hohenberg-Kohn Theorems Definitions External potential The external potential — potential vext acting on electrons the source of which are not electrons themselves. Ĥ = T̂ + V̂ext + V̂ee + V̂nn , V̂ext = N X vext (ri ). i=1 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 46 / 101 Hohenberg-Kohn Theorems Definitions External potential The external potential — potential vext acting on electrons the source of which are not electrons themselves. Ĥ = T̂ + V̂ext + V̂ee + V̂nn , V̂ext = N X vext (ri ). i=1 Without any external (electric, magnetic) fields it’s just the nuclear potential of the system: vext (r) = vne (r) = − M X α=1 Łukasz Rajchel (University of Warsaw) DFT Zα . |r − Rα | Warsaw, 2010 46 / 101 Hohenberg-Kohn Theorems Definitions Hohenberg-Kohn functional We assume that the kinetic energy and e-e repulsion may be represented as a functional of the density only: E[ρ] = T [ρ] + Vext [ρ] + Vee [ρ] + Vnn . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 47 / 101 Hohenberg-Kohn Theorems Definitions Hohenberg-Kohn functional We assume that the kinetic energy and e-e repulsion may be represented as a functional of the density only: E[ρ] = T [ρ] + Vext [ρ] + Vee [ρ] + Vnn . Let’s now regroup the energy functional a little bit: E[ρ] = Vext [ρ] + FHK [ρ], Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 47 / 101 Hohenberg-Kohn Theorems Definitions Hohenberg-Kohn functional We assume that the kinetic energy and e-e repulsion may be represented as a functional of the density only: E[ρ] = T [ρ] + Vext [ρ] + Vee [ρ] + Vnn . Let’s now regroup the energy functional a little bit: E[ρ] = Vext [ρ] + FHK [ρ], Z Vext [ρ] = vext (r)ρ(r) d3 r — system-dependent part (vext changes R3 with the system). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 47 / 101 Hohenberg-Kohn Theorems Definitions Hohenberg-Kohn functional We assume that the kinetic energy and e-e repulsion may be represented as a functional of the density only: E[ρ] = T [ρ] + Vext [ρ] + Vee [ρ] + Vnn . Let’s now regroup the energy functional a little bit: E[ρ] = Vext [ρ] + FHK [ρ], Z Vext [ρ] = vext (r)ρ(r) d3 r — system-dependent part (vext changes R3 with the system). FHK [ρ] = T [ρ] + J[ρ] + Encl [ρ] — Hohenberg-Kohn functional: universal for all systems. But we don’t know T [ρ] nor Encl [ρ]. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 47 / 101 Hohenberg-Kohn Theorems The Theorems Hohenberg-Kohn Theorems Z E[ρ] = vext (r)ρ(r) d3 r + FHK [ρ] R3 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 48 / 101 Hohenberg-Kohn Theorems The Theorems Hohenberg-Kohn Theorems Z E[ρ] = vext (r)ρ(r) d3 r + FHK [ρ] R3 Theorem (One, HK1) The external potential vext (r) and hence the total energy, is a unique functional of the electron density ρ(r). So, there is one-to-one mapping vext ↔ ρ. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 48 / 101 Hohenberg-Kohn Theorems The Theorems Hohenberg-Kohn Theorems Z E[ρ] = vext (r)ρ(r) d3 r + FHK [ρ] R3 Theorem (One, HK1) The external potential vext (r) and hence the total energy, is a unique functional of the electron density ρ(r). So, there is one-to-one mapping vext ↔ ρ. Theorem (Two, HK2) The density ρ0 minimizing the total energy is the exact ground-state density. So, given a trial density ρ̃ (non-negative and integrating to N ) we get E[ρ̃] ≥ E[ρ0 ] = E0 . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 48 / 101 Hohenberg-Kohn Theorems The Theorems A few remaks on Hohenberg-Kohn theorems: if we knew FHK [ρ], we would get gorund-state density and energy. FHK [ρ] is the Holy Grail of DFT! Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 49 / 101 Hohenberg-Kohn Theorems The Theorems A few remaks on Hohenberg-Kohn theorems: if we knew FHK [ρ], we would get gorund-state density and energy. FHK [ρ] is the Holy Grail of DFT! HK theorems prove that there is indeed one-to-one mapping between ground-state density and energy: ρ ↔ E, but give us no clue how to construct the functional yielding the ground-state density. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 49 / 101 Hohenberg-Kohn Theorems The Theorems A few remaks on Hohenberg-Kohn theorems: if we knew FHK [ρ], we would get gorund-state density and energy. FHK [ρ] is the Holy Grail of DFT! HK theorems prove that there is indeed one-to-one mapping between ground-state density and energy: ρ ↔ E, but give us no clue how to construct the functional yielding the ground-state density. the variational principle introduced by HK2 applies to the exact functional only! And we don’t know it — we use only some approximations. That means variational principle doesn’t work in practice — we can get energies lower than the true ones. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 49 / 101 Hohenberg-Kohn Theorems The Theorems A few remaks on Hohenberg-Kohn theorems: if we knew FHK [ρ], we would get gorund-state density and energy. FHK [ρ] is the Holy Grail of DFT! HK theorems prove that there is indeed one-to-one mapping between ground-state density and energy: ρ ↔ E, but give us no clue how to construct the functional yielding the ground-state density. the variational principle introduced by HK2 applies to the exact functional only! And we don’t know it — we use only some approximations. That means variational principle doesn’t work in practice — we can get energies lower than the true ones. example: using BPW91 functional in cc-pV5Z basis set, for H atom we get E = −0.5042, the true energy being E = −0.5: −0.5042 < −0.5. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 49 / 101 Hohenberg-Kohn Theorems Representability of the Density v-representability and N -representability The density ρ is: v-representable if it is associated with ground-state antisymmetric ψ0 satisfying Ĥψ0 = E0 ψ0 , where Ĥ contains the external potential vext : T̂ + N X vext (ri ) + V̂ee + V̂nn → Ĥψ = Eψ → ψ → ρ . i=1 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 50 / 101 Hohenberg-Kohn Theorems Representability of the Density v-representability and N -representability The density ρ is: v-representable if it is associated with ground-state antisymmetric ψ0 satisfying Ĥψ0 = E0 ψ0 , where Ĥ contains the external potential vext : T̂ + N X vext (ri ) + V̂ee + V̂nn → Ĥψ = Eψ → ψ → ρ . i=1 N -representable if it can be obtained from some antisymmetric ψ: ψ→ρ. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 50 / 101 Hohenberg-Kohn Theorems Representability of the Density v-representability and N -representability The density ρ is: v-representable if it is associated with ground-state antisymmetric ψ0 satisfying Ĥψ0 = E0 ψ0 , where Ĥ contains the external potential vext : T̂ + N X vext (ri ) + V̂ee + V̂nn → Ĥψ = Eψ → ψ → ρ . i=1 N -representable if it can be obtained from some antisymmetric ψ: ψ→ρ. All v-representable ρ’s are N -representable. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 50 / 101 Hohenberg-Kohn Theorems Representability of the Density v-representability and N -representability The density ρ is: v-representable if it is associated with ground-state antisymmetric ψ0 satisfying Ĥψ0 = E0 ψ0 , where Ĥ contains the external potential vext : T̂ + N X vext (ri ) + V̂ee + V̂nn → Ĥψ = Eψ → ψ → ρ . i=1 N -representable if it can be obtained from some antisymmetric ψ: ψ→ρ. All v-representable ρ’s are N -representable. HK2 originally deals only with v-representable ρ’s. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 50 / 101 Hohenberg-Kohn Theorems Representability of the Density v-representability and N -representability The density ρ is: v-representable if it is associated with ground-state antisymmetric ψ0 satisfying Ĥψ0 = E0 ψ0 , where Ĥ contains the external potential vext : T̂ + N X vext (ri ) + V̂ee + V̂nn → Ĥψ = Eψ → ψ → ρ . i=1 N -representable if it can be obtained from some antisymmetric ψ: ψ→ρ. All v-representable ρ’s are N -representable. HK2 originally deals only with v-representable ρ’s. But the conditions of ρ’s v-representability are yet unknown. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 50 / 101 Hohenberg-Kohn Theorems Representability of the Density v-representability and N -representability The density ρ is: v-representable if it is associated with ground-state antisymmetric ψ0 satisfying Ĥψ0 = E0 ψ0 , where Ĥ contains the external potential vext : T̂ + N X vext (ri ) + V̂ee + V̂nn → Ĥψ = Eψ → ψ → ρ . i=1 N -representable if it can be obtained from some antisymmetric ψ: ψ→ρ. All v-representable ρ’s are N -representable. HK2 originally deals only with v-representable ρ’s. But the conditions of ρ’s v-representability are yet unknown. Fortunately, it turns out we can lift that condition and extend our variational search on all N -representable ρ’s, without the explicit connection to an external potential. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 50 / 101 Hohenberg-Kohn Theorems Representability of the Density Non-interacting v-representability Suppose we have a system described with hamiltonian containing only one-body electron operators (no e-e interaction): ĤS = T̂ + N X v(ri ) + V̂nn . i=1 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 51 / 101 Hohenberg-Kohn Theorems Representability of the Density Non-interacting v-representability Suppose we have a system described with hamiltonian containing only one-body electron operators (no e-e interaction): ĤS = T̂ + N X v(ri ) + V̂nn . i=1 We solve the Schrödinger equation, ĤS ψS = EψS and obtain the density (through the integration): ψS → ρ. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 51 / 101 Hohenberg-Kohn Theorems Representability of the Density Non-interacting v-representability Suppose we have a system described with hamiltonian containing only one-body electron operators (no e-e interaction): ĤS = T̂ + N X v(ri ) + V̂nn . i=1 We solve the Schrödinger equation, ĤS ψS = EψS and obtain the density (through the integration): ψS → ρ. Such a density is said to be non-interacting v-representable, because it refers to the system of non-interacting electrons. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 51 / 101 Hohenberg-Kohn Theorems Representability of the Density Constrained-search approach Variational principle in the quest for ground-state energy: minimize energy expectation value over all antisymmetric N -electron ψ’s: E D E0 = min ψ T̂ + V̂ext + V̂ee + V̂nn ψ . ψ→N Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 52 / 101 Hohenberg-Kohn Theorems Representability of the Density Constrained-search approach Variational principle in the quest for ground-state energy: minimize energy expectation value over all antisymmetric N -electron ψ’s: E D E0 = min ψ T̂ + V̂ext + V̂ee + V̂nn ψ . ψ→N Constrained-search approach is performed in the two steps: Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 52 / 101 Hohenberg-Kohn Theorems Representability of the Density Constrained-search approach Variational principle in the quest for ground-state energy: minimize energy expectation value over all antisymmetric N -electron ψ’s: E D E0 = min ψ T̂ + V̂ext + V̂ee + V̂nn ψ . ψ→N Constrained-search approach is performed in the two steps: given a particular ρi integrating to N , find the ψi yielding ρi that gives the minimal energy: in result we get ψimin . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 52 / 101 Hohenberg-Kohn Theorems Representability of the Density Constrained-search approach Variational principle in the quest for ground-state energy: minimize energy expectation value over all antisymmetric N -electron ψ’s: E D E0 = min ψ T̂ + V̂ext + V̂ee + V̂nn ψ . ψ→N Constrained-search approach is performed in the two steps: given a particular ρi integrating to N , find the ψi yielding ρi that gives the minimal energy: in result we get ψimin . from the set of the densities {ρi }M i=1 and corresponding wavefunctions min M {ψi }i=1 choose the one which yields the smallest energy. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 52 / 101 Hohenberg-Kohn Theorems Representability of the Density Constrained-search approach Variational principle in the quest for ground-state energy: minimize energy expectation value over all antisymmetric N -electron ψ’s: E D E0 = min ψ T̂ + V̂ext + V̂ee + V̂nn ψ . ψ→N Constrained-search approach is performed in the two steps: given a particular ρi integrating to N , find the ψi yielding ρi that gives the minimal energy: in result we get ψimin . from the set of the densities {ρi }M i=1 and corresponding wavefunctions min M {ψi }i=1 choose the one which yields the smallest energy. Schematically, E D E0 = min min ψ T̂ + V̂ext + V̂ee + V̂nn ψ . ρ Łukasz Rajchel (University of Warsaw) ψ→ρ DFT Warsaw, 2010 52 / 101 Hohenberg-Kohn Theorems Representability of the Density Constrained-search approach To identify the tallest child in aschool, we don’t need to line all the children up in the schoolyard. Simply choose the tallest child in each classroom and ask those to come to the schoolyard, where the final search is performed. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 52 / 101 Hohenberg-Kohn Theorems Representability of the Density Constrained-search approach Each striped area represents ψ’s giving the particular ρi . min : we constrain our search to the particular striped area and find ψimin ψ→ρ yielding the smallest energy, represented by the point •. min: we minimize over all points (•) to find E0 . ρ Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 52 / 101 Hohenberg-Kohn Theorems Representability of the Density Constrained-search approach E D E0 = min min ψ T̂ + V̂ext + V̂ee + V̂nn ψ ρ ψ→ρ Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 52 / 101 Hohenberg-Kohn Theorems Representability of the Density Constrained-search approach E D E0 = min min ψ T̂ + V̂ext + V̂ee + V̂nn ψ = ρ ψ→ρ E Z D 3 = min min ψ T̂ + V̂ee ψ + vext (r)ρ(r) d r + Vnn ρ ψ→ρ Łukasz Rajchel (University of Warsaw) R3 DFT Warsaw, 2010 52 / 101 Hohenberg-Kohn Theorems Representability of the Density Constrained-search approach E D E0 = min min ψ T̂ + V̂ext + V̂ee + V̂nn ψ = ρ ψ→ρ E Z D 3 = min min ψ T̂ + V̂ee ψ + vext (r)ρ(r) d r + Vnn = ρ ψ→ρ R3 Z = min F [ρ] + vext (r)ρ(r) d3 r + Vnn , ρ Łukasz Rajchel (University of Warsaw) R3 DFT Warsaw, 2010 52 / 101 Hohenberg-Kohn Theorems Representability of the Density Constrained-search approach E D E0 = min min ψ T̂ + V̂ext + V̂ee + V̂nn ψ = ρ ψ→ρ E Z D 3 = min min ψ T̂ + V̂ee ψ + vext (r)ρ(r) d r + Vnn = ρ ψ→ρ R3 Z = min F [ρ] + vext (r)ρ(r) d3 r + Vnn , ρ R3 E D F [ρ] = min ψ T̂ + V̂ee ψ . ψ→ρ Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 52 / 101 Hohenberg-Kohn Theorems Representability of the Density Constrained-search approach E D E0 = min min ψ T̂ + V̂ext + V̂ee + V̂nn ψ = ρ ψ→ρ E Z D 3 = min min ψ T̂ + V̂ee ψ + vext (r)ρ(r) d r + Vnn = ρ ψ→ρ R3 Z = min F [ρ] + vext (r)ρ(r) d3 r + Vnn , ρ R3 E D F [ρ] = min ψ T̂ + V̂ee ψ . ψ→ρ We’ve already introduced HK functional, FHK [ρ] = T [ρ] + Vee [ρ]. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 52 / 101 Hohenberg-Kohn Theorems Representability of the Density Constrained-search approach E D E0 = min min ψ T̂ + V̂ext + V̂ee + V̂nn ψ = ρ ψ→ρ E Z D 3 = min min ψ T̂ + V̂ee ψ + vext (r)ρ(r) d r + Vnn = ρ ψ→ρ R3 Z = min F [ρ] + vext (r)ρ(r) d3 r + Vnn , ρ R3 E D F [ρ] = min ψ T̂ + V̂ee ψ . ψ→ρ We’ve already introduced HK functional, FHK [ρ] = T [ρ] + Vee [ρ]. Clearly, for the ground-state density we have F [ρ0 ] = FHK [ρ0 ]. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 52 / 101 Outline of the Talk 5 Density and Energy 6 Hohenberg-Kohn Theorems 7 Kohn-Sham Approach Introductory Remarks KS Determinant and KS Energy 8 xc Functionals Kohn-Sham Approach Introductory Remarks A few remaks on the Hartree-Fock model It lacks any correlation, but it properly describes the exchange. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 54 / 101 Kohn-Sham Approach Introductory Remarks A few remaks on the Hartree-Fock model It lacks any correlation, but it properly describes the exchange. So, the electrons described by HF function may be viewed as uncharged fermions: particles obeying the Pauli principle and neglecting the Coulomb repulsion. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 54 / 101 Kohn-Sham Approach Introductory Remarks A few remaks on the Hartree-Fock model It lacks any correlation, but it properly describes the exchange. So, the electrons described by HF function may be viewed as uncharged fermions: particles obeying the Pauli principle and neglecting the Coulomb repulsion. In this sense the HF function, ψHF , can be considered as the exact wavefunction of a fictitious system of N non-interacting electrons. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 54 / 101 Kohn-Sham Approach Introductory Remarks A few remaks on the Hartree-Fock model It lacks any correlation, but it properly describes the exchange. So, the electrons described by HF function may be viewed as uncharged fermions: particles obeying the Pauli principle and neglecting the Coulomb repulsion. In this sense the HF function, ψHF , can be considered as the exact wavefunction of a fictitious system of N non-interacting electrons. Each electron is described by the orbital ϕi , which is the solution of the HF equation, fˆϕi = i ϕi , with the Fock operator 1 fˆ(r) = − ∆r + vne (r) + vHF (r), 2 so each electron moves in the effective potential veff = vne + vHF . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 54 / 101 Kohn-Sham Approach Introductory Remarks A few remaks on the Hartree-Fock model It lacks any correlation, but it properly describes the exchange. So, the electrons described by HF function may be viewed as uncharged fermions: particles obeying the Pauli principle and neglecting the Coulomb repulsion. In this sense the HF function, ψHF , can be considered as the exact wavefunction of a fictitious system of N non-interacting electrons. Each electron is described by the orbital ϕi , which is the solution of the HF equation, fˆϕi = i ϕi , with the Fock operator 1 fˆ(r) = − ∆r + vne (r) + vHF (r), 2 so each electron moves in the effective potential veff = vne + vHF . The electronic kinetic energy reads Z N/2 X 1 0 3 T [ρ] = − ∆r ρ(r; r ) r0 =r d r = − hϕi |∆r |ϕi i . 2 R3 i=1 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 54 / 101 Kohn-Sham Approach KS Determinant and KS Energy Non-interacting reference system We now set up a system described by the Hamiltonian ĤS = T̂ + V̂S + V̂nn with V̂S = N X vS (ri ). i=1 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 55 / 101 Kohn-Sham Approach KS Determinant and KS Energy Non-interacting reference system We now set up a system described by the Hamiltonian ĤS = T̂ + V̂S + V̂nn with V̂S = N X vS (ri ). i=1 ĤS contains no e-e interaction, so it obviously describes the non-interacting system! Its wavefunction is then a single Slater determinant: ψS = |ϕ1 αϕ1 β . . . ϕN/2 αϕN/2 βi . ψS → ρS : non-interacting v-representable (no e-e interaction in ĤS ). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 55 / 101 Kohn-Sham Approach KS Determinant and KS Energy Non-interacting reference system We now set up a system described by the Hamiltonian ĤS = T̂ + V̂S + V̂nn with V̂S = N X vS (ri ). i=1 ĤS contains no e-e interaction, so it obviously describes the non-interacting system! Its wavefunction is then a single Slater determinant: ψS = |ϕ1 αϕ1 β . . . ϕN/2 αϕN/2 βi . ψS → ρS : non-interacting v-representable (no e-e interaction in ĤS ). Each electron of this system moves in the effective potential veff = vS , so the orbitals are obtained from HF-like equations: fˆKS ϕi = i ϕi with the operator (called Kohn-Sham operator) being 1 fˆKS = − ∆r + vS (r). 2 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 55 / 101 Kohn-Sham Approach KS Determinant and KS Energy Kohn-Sham model We’ve already set up the non-interaction reference system and come up with the orbital equations. But what is vS and how do we get it? Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 56 / 101 Kohn-Sham Approach KS Determinant and KS Energy Kohn-Sham model We’ve already set up the non-interaction reference system and come up with the orbital equations. But what is vS and how do we get it? The recipe = Kohn-Sham model: Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 56 / 101 Kohn-Sham Approach KS Determinant and KS Energy Kohn-Sham model We’ve already set up the non-interaction reference system and come up with the orbital equations. But what is vS and how do we get it? The recipe = Kohn-Sham model: we require that the density resulting from KS determinant ψKS : ρS (r) = 2 N/2 X |ϕi (r)|2 i=1 is the same as the density of the real target system of interacting electrons: ρS = ρ. So, ρ is also non-interacting v-representable. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 56 / 101 Kohn-Sham Approach KS Determinant and KS Energy Kohn-Sham model We’ve already set up the non-interaction reference system and come up with the orbital equations. But what is vS and how do we get it? The recipe = Kohn-Sham model: we require that the density resulting from KS determinant ψKS : ρS (r) = 2 N/2 X |ϕi (r)|2 i=1 is the same as the density of the real target system of interacting electrons: ρS = ρ. So, ρ is also non-interacting v-representable. since we don’t know the explicit T [ρ] functional, the kinetic energy is computed using HF-like expression: TS [ρ] = − N/2 X hϕi |∆r |ϕi i , i=1 and the remainder is shifted to the exchange-correlation energy. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 56 / 101 Kohn-Sham Approach KS Determinant and KS Energy Kohn-Sham energy functional The total energy of a real system in Kohn-Sham model: E[ρ] = T [ρ] + Vne [ρ] + Eee [ρ] + Vnn . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 57 / 101 Kohn-Sham Approach KS Determinant and KS Energy Kohn-Sham energy functional The total energy of a real system in Kohn-Sham model: E[ρ] = T [ρ] + Vne [ρ] + Eee [ρ] + Vnn = = T [ρ] + Vne [ρ] + J[ρ] + Encl [ρ] + Vnn . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 57 / 101 Kohn-Sham Approach KS Determinant and KS Energy Kohn-Sham energy functional The total energy of a real system in Kohn-Sham model: E[ρ] = T [ρ] + Vne [ρ] + Eee [ρ] + Vnn = = T [ρ] + Vne [ρ] + J[ρ] + Encl [ρ] + Vnn = = TS [ρ] + T [ρ] − TS [ρ] + Vne [ρ] + J[ρ] + Encl [ρ] + Vnn . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 57 / 101 Kohn-Sham Approach KS Determinant and KS Energy Kohn-Sham energy functional The total energy of a real system in Kohn-Sham model: E[ρ] = T [ρ] + Vne [ρ] + Eee [ρ] + Vnn = = T [ρ] + Vne [ρ] + J[ρ] + Encl [ρ] + Vnn = = TS [ρ] + T [ρ] − TS [ρ] + Vne [ρ] + J[ρ] + Encl [ρ] + Vnn = = TS [ρ] + Vne [ρ] + J[ρ] + T [ρ] − TS [ρ] + Encl [ρ] | {z } +Vnn . Exc [ρ]: exchange-correlation energy Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 57 / 101 Kohn-Sham Approach KS Determinant and KS Energy Kohn-Sham energy functional The total energy of a real system in Kohn-Sham model: E[ρ] = T [ρ] + Vne [ρ] + Eee [ρ] + Vnn = = T [ρ] + Vne [ρ] + J[ρ] + Encl [ρ] + Vnn = = TS [ρ] + T [ρ] − TS [ρ] + Vne [ρ] + J[ρ] + Encl [ρ] + Vnn = = TS [ρ] + Vne [ρ] + J[ρ] + T [ρ] − TS [ρ] + Encl [ρ] | {z } +Vnn . Exc [ρ]: exchange-correlation energy Finally, the famous exchange-correlation (xc) energy functional is Exc [ρ] = T [ρ] − TS [ρ] + Encl [ρ]. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 57 / 101 Kohn-Sham Approach KS Determinant and KS Energy Kohn-Sham energy functional The total energy of a real system in Kohn-Sham model: E[ρ] = T [ρ] + Vne [ρ] + Eee [ρ] + Vnn = = T [ρ] + Vne [ρ] + J[ρ] + Encl [ρ] + Vnn = = TS [ρ] + T [ρ] − TS [ρ] + Vne [ρ] + J[ρ] + Encl [ρ] + Vnn = = TS [ρ] + Vne [ρ] + J[ρ] + T [ρ] − TS [ρ] + Encl [ρ] | {z } +Vnn . Exc [ρ]: exchange-correlation energy Finally, the famous exchange-correlation (xc) energy functional is Exc [ρ] = T [ρ] − TS [ρ] + Encl [ρ]. Apparently, the Exc [ρ]’s responsibility is enormous: it contains non-classical effects of self-interaction correction, exchange and correlation, Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 57 / 101 Kohn-Sham Approach KS Determinant and KS Energy Kohn-Sham energy functional The total energy of a real system in Kohn-Sham model: E[ρ] = T [ρ] + Vne [ρ] + Eee [ρ] + Vnn = = T [ρ] + Vne [ρ] + J[ρ] + Encl [ρ] + Vnn = = TS [ρ] + T [ρ] − TS [ρ] + Vne [ρ] + J[ρ] + Encl [ρ] + Vnn = = TS [ρ] + Vne [ρ] + J[ρ] + T [ρ] − TS [ρ] + Encl [ρ] | {z } +Vnn . Exc [ρ]: exchange-correlation energy Finally, the famous exchange-correlation (xc) energy functional is Exc [ρ] = T [ρ] − TS [ρ] + Encl [ρ]. Apparently, the Exc [ρ]’s responsibility is enormous: it contains non-classical effects of self-interaction correction, exchange and correlation, plus portion of kinetic energy not present in the non-interacting reference system! Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 57 / 101 Kohn-Sham Approach KS Determinant and KS Energy Kohn-Sham equations Thus, we have established the total energy of a systems as a functional of density: E[ρ] = TS [ρ] + Vne [ρ] + J[ρ] + Exc [ρ] + Vnn . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 58 / 101 Kohn-Sham Approach KS Determinant and KS Energy Kohn-Sham equations Thus, we have established the total energy of a systems as a functional of density: E[ρ] = TS [ρ] + Vne [ρ] + J[ρ] + Exc [ρ] + Vnn . Task : minimze E[ρ] with the constraint on the density integration: Z ρ(r) d3 r = N, R3 and the calculus of variations comes with the proper tools to do it! Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 58 / 101 Kohn-Sham Approach KS Determinant and KS Energy Kohn-Sham equations Thus, we have established the total energy of a systems as a functional of density: E[ρ] = TS [ρ] + Vne [ρ] + J[ρ] + Exc [ρ] + Vnn . Task : minimze E[ρ] with the constraint on the density integration: Z ρ(r) d3 r = N, R3 and the calculus of variations comes with the proper tools to do it! Result : Kohn-Sham equations for optimal orbitals: fˆKS ϕi = i ϕi . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 58 / 101 Kohn-Sham Approach KS Determinant and KS Energy Kohn-Sham operator Based on the considerations about the non-interacting reference system we’ve arrived at 1 fˆKS (r) = − ∆r + vS (r). 2 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 59 / 101 Kohn-Sham Approach KS Determinant and KS Energy Kohn-Sham operator Based on the considerations about the non-interacting reference system we’ve arrived at 1 fˆKS (r) = − ∆r + vS (r). 2 And now, based on the total Kohn-Sham energy functional minimization we find the effective potential we’ve been looking for: vS (r) = vne (r) + ̂(r) + vxc (r), Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 59 / 101 Kohn-Sham Approach KS Determinant and KS Energy Kohn-Sham operator Based on the considerations about the non-interacting reference system we’ve arrived at 1 fˆKS (r) = − ∆r + vS (r). 2 And now, based on the total Kohn-Sham energy functional minimization we find the effective potential we’ve been looking for: vS (r) = vne (r) + ̂(r) + vxc (r), but since we don’t know the explicit form of xc energy, we don’t know how xc potential looks either, so we can only put it as the functional derivative of the xc energy with respect to the density: vxc (r) = Łukasz Rajchel (University of Warsaw) δExc [ρ] . δρ(r) DFT Warsaw, 2010 59 / 101 Kohn-Sham Approach KS Determinant and KS Energy Hartree-Fock and Kohn-Sham models Hartree-Fock operator: fˆ(r) = − 12 ∆r + vne (r) + ̂(r) − k̂(r), Kohn-Sham operator: fˆKS (r) = − 12 ∆r + vne (r) + ̂(r) + vxc (r). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 60 / 101 Kohn-Sham Approach KS Determinant and KS Energy Hartree-Fock and Kohn-Sham models Hartree-Fock operator: fˆ(r) = − 12 ∆r + vne (r) + ̂(r) − k̂(r), Kohn-Sham operator: fˆKS (r) = − 12 ∆r + vne (r) + ̂(r) + vxc (r). Hartree-Fock: contains non-local exchange operator. takes no parameters, the energy is well-defined. is purely variational, the energy is always higher than its true value. yields the best energy within one-electron approximation. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 60 / 101 Kohn-Sham Approach KS Determinant and KS Energy Hartree-Fock and Kohn-Sham models Hartree-Fock operator: fˆ(r) = − 12 ∆r + vne (r) + ̂(r) − k̂(r), Kohn-Sham operator: fˆKS (r) = − 12 ∆r + vne (r) + ̂(r) + vxc (r). Hartree-Fock: contains non-local exchange operator. Kohn-Sham: all operators are local. the energy depends on the approximation to xc energy. takes no parameters, the energy is well-defined. variational method works only for exact xc functional, in practice it does not apply. is purely variational, the energy is always higher than its true value. is potentially exact — once we knew exact xc functional, we would get the exact energy. yields the best energy within one-electron approximation. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 60 / 101 Outline of the Talk 5 Density and Energy 6 Hohenberg-Kohn Theorems 7 Kohn-Sham Approach 8 xc Functionals Is There a Road Map? Adiabatic Connection Kohn-Sham Machinery xc Functionals Is There a Road Map? Some remarks on xc functionals Exc [ρ] is the central object in DFT and KS. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 62 / 101 xc Functionals Is There a Road Map? Some remarks on xc functionals Exc [ρ] is the central object in DFT and KS. Exact Exc [ρ] gives exact energy, i.e. energy strictly satisfying Schrödinger equation. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 62 / 101 xc Functionals Is There a Road Map? Some remarks on xc functionals Exc [ρ] is the central object in DFT and KS. Exact Exc [ρ] gives exact energy, i.e. energy strictly satisfying Schrödinger equation. But no one knows the exact Exc [ρ]! Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 62 / 101 xc Functionals Is There a Road Map? Some remarks on xc functionals Exc [ρ] is the central object in DFT and KS. Exact Exc [ρ] gives exact energy, i.e. energy strictly satisfying Schrödinger equation. But no one knows the exact Exc [ρ]! So, we must make explicit approximations to this functional, otherwise KS model makes no sense and is practically useless! Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 62 / 101 xc Functionals Is There a Road Map? Some remarks on xc functionals Exc [ρ] is the central object in DFT and KS. Exact Exc [ρ] gives exact energy, i.e. energy strictly satisfying Schrödinger equation. But no one knows the exact Exc [ρ]! So, we must make explicit approximations to this functional, otherwise KS model makes no sense and is practically useless! DFT mission: the never-ending quest for better and better xc functionals . . . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 62 / 101 xc Functionals Is There a Road Map? The Holy Grail of DFT: exact xc functional There is no systematic strategy how to get closer to the exact xc functional, as is the case in wavefunction-based approaches, where the the variational method is the cornerstone. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 63 / 101 xc Functionals Is There a Road Map? The Holy Grail of DFT: exact xc functional There is no systematic strategy how to get closer to the exact xc functional, as is the case in wavefunction-based approaches, where the the variational method is the cornerstone. The explicit form of the exact functional remains a total mystery to us. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 63 / 101 xc Functionals Is There a Road Map? The Holy Grail of DFT: exact xc functional There is no systematic strategy how to get closer to the exact xc functional, as is the case in wavefunction-based approaches, where the the variational method is the cornerstone. The explicit form of the exact functional remains a total mystery to us. The attempts to find better functionals rely to a large extent on physical and mathematical intuition, and have strong trial and error component. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 63 / 101 xc Functionals Is There a Road Map? The Holy Grail of DFT: exact xc functional There is no systematic strategy how to get closer to the exact xc functional, as is the case in wavefunction-based approaches, where the the variational method is the cornerstone. The explicit form of the exact functional remains a total mystery to us. The attempts to find better functionals rely to a large extent on physical and mathematical intuition, and have strong trial and error component. There are some physical constraints that the reasonable functionals should obey: sum rules for the xc holes, cusp condition, asymptotic properties of the resulting xc potentials, etc. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 63 / 101 xc Functionals Is There a Road Map? The Holy Grail of DFT: exact xc functional There is no systematic strategy how to get closer to the exact xc functional, as is the case in wavefunction-based approaches, where the the variational method is the cornerstone. The explicit form of the exact functional remains a total mystery to us. The attempts to find better functionals rely to a large extent on physical and mathematical intuition, and have strong trial and error component. There are some physical constraints that the reasonable functionals should obey: sum rules for the xc holes, cusp condition, asymptotic properties of the resulting xc potentials, etc. Nevertheless, it turns out that some successful functionals obey several of these conditions and are still better than some kosher ones . . . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 63 / 101 xc Functionals Adiabatic Connection xc holes and xc functionals We remember that the non-classical e-e repulsion in terms of xc hole is Z Z 1 ρ(r1 )hxc (r1 ; r2 ) 3 Encl [ρ] = d r1 d3 r2 . 2 R3 R3 r12 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 64 / 101 xc Functionals Adiabatic Connection xc holes and xc functionals We remember that the non-classical e-e repulsion in terms of xc hole is Z Z 1 ρ(r1 )hxc (r1 ; r2 ) 3 Encl [ρ] = d r1 d3 r2 . 2 R3 R3 r12 But the xc functional of KS scheme includes also the kinetic energy correlation contribution: Exc [ρ] = T [ρ] − TS [ρ] + Encl [ρ] = Tcor [ρ] + Encl [ρ]. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 64 / 101 xc Functionals Adiabatic Connection xc holes and xc functionals We remember that the non-classical e-e repulsion in terms of xc hole is Z Z 1 ρ(r1 )hxc (r1 ; r2 ) 3 Encl [ρ] = d r1 d3 r2 . 2 R3 R3 r12 But the xc functional of KS scheme includes also the kinetic energy correlation contribution: Exc [ρ] = T [ρ] − TS [ρ] + Encl [ρ] = Tcor [ρ] + Encl [ρ]. We also know that KS model uses two crucial objects: Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 64 / 101 xc Functionals Adiabatic Connection xc holes and xc functionals We remember that the non-classical e-e repulsion in terms of xc hole is Z Z 1 ρ(r1 )hxc (r1 ; r2 ) 3 Encl [ρ] = d r1 d3 r2 . 2 R3 R3 r12 But the xc functional of KS scheme includes also the kinetic energy correlation contribution: Exc [ρ] = T [ρ] − TS [ρ] + Encl [ρ] = Tcor [ρ] + Encl [ρ]. We also know that KS model uses two crucial objects: non-interacting reference system (density: ρS ), Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 64 / 101 xc Functionals Adiabatic Connection xc holes and xc functionals We remember that the non-classical e-e repulsion in terms of xc hole is Z Z 1 ρ(r1 )hxc (r1 ; r2 ) 3 Encl [ρ] = d r1 d3 r2 . 2 R3 R3 r12 But the xc functional of KS scheme includes also the kinetic energy correlation contribution: Exc [ρ] = T [ρ] − TS [ρ] + Encl [ρ] = Tcor [ρ] + Encl [ρ]. We also know that KS model uses two crucial objects: non-interacting reference system (density: ρS ), the real system with fully interacting electrons (density: ρ). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 64 / 101 xc Functionals Adiabatic Connection xc holes and xc functionals We remember that the non-classical e-e repulsion in terms of xc hole is Z Z 1 ρ(r1 )hxc (r1 ; r2 ) 3 Encl [ρ] = d r1 d3 r2 . 2 R3 R3 r12 But the xc functional of KS scheme includes also the kinetic energy correlation contribution: Exc [ρ] = T [ρ] − TS [ρ] + Encl [ρ] = Tcor [ρ] + Encl [ρ]. We also know that KS model uses two crucial objects: non-interacting reference system (density: ρS ), the real system with fully interacting electrons (density: ρ). The two systems share the same density: ρS = ρ. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 64 / 101 xc Functionals Adiabatic Connection Coupling the two systems of KS model We introduce the coupling parameter λ ∈ h0; 1i: Ĥ(λ) = T̂ + V̂ext (λ) + λV̂ee + V̂nn . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 65 / 101 xc Functionals Adiabatic Connection Coupling the two systems of KS model We introduce the coupling parameter λ ∈ h0; 1i: Ĥ(λ) = T̂ + V̂ext (λ) + λV̂ee + V̂nn . V̂ext (λ) changes with λ so that the density of the system described with Ĥ(λ) equals the density of the real system. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 65 / 101 xc Functionals Adiabatic Connection Coupling the two systems of KS model We introduce the coupling parameter λ ∈ h0; 1i: Ĥ(λ) = T̂ + V̂ext (λ) + λV̂ee + V̂nn . V̂ext (λ) changes with λ so that the density of the system described with Ĥ(λ) equals the density of the real system. Boundary cases: Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 65 / 101 xc Functionals Adiabatic Connection Coupling the two systems of KS model We introduce the coupling parameter λ ∈ h0; 1i: Ĥ(λ) = T̂ + V̂ext (λ) + λV̂ee + V̂nn . V̂ext (λ) changes with λ so that the density of the system described with Ĥ(λ) equals the density of the real system. Boundary cases: λ = 0: non-interacting system Hamiltonian, Vext (0) = V̂S . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 65 / 101 xc Functionals Adiabatic Connection Coupling the two systems of KS model We introduce the coupling parameter λ ∈ h0; 1i: Ĥ(λ) = T̂ + V̂ext (λ) + λV̂ee + V̂nn . V̂ext (λ) changes with λ so that the density of the system described with Ĥ(λ) equals the density of the real system. Boundary cases: λ = 0: non-interacting system Hamiltonian, Vext (0) = V̂S . λ = 1: real system Hamiltonian, V̂ext (1) = V̂ne for isolated molecule. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 65 / 101 xc Functionals Adiabatic Connection Adiabatic connection formula Through the artificial and smooth coupling of the two systems the following energy expression is derived: Z Z 1 ρ(r1 )h̄xc (r1 ; r2 ) 3 E[ρ] = TS [ρ] + Vne [ρ] + Jne [ρ] + d r1 d3 r2 + Vnn , 2 R3 R3 r12 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 66 / 101 xc Functionals Adiabatic Connection Adiabatic connection formula Through the artificial and smooth coupling of the two systems the following energy expression is derived: Z Z 1 ρ(r1 )h̄xc (r1 ; r2 ) 3 E[ρ] = TS [ρ] + Vne [ρ] + Jne [ρ] + d r1 d3 r2 + Vnn , 2 R3 R3 r12 whereas the equivalent expression, which we already know very well, reads Z Z 1 ρ(r1 )hxc (r1 ; r2 ) 3 E[ρ] = T [ρ] + Vne [ρ] + Jne [ρ] + d r1 d3 r2 +, Vnn . 2 R3 R3 r12 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 66 / 101 xc Functionals Adiabatic Connection Adiabatic connection formula Through the artificial and smooth coupling of the two systems the following energy expression is derived: Z Z 1 ρ(r1 )h̄xc (r1 ; r2 ) 3 E[ρ] = TS [ρ] + Vne [ρ] + Jne [ρ] + d r1 d3 r2 + Vnn , 2 R3 R3 r12 whereas the equivalent expression, which we already know very well, reads Z Z 1 ρ(r1 )hxc (r1 ; r2 ) 3 E[ρ] = T [ρ] + Vne [ρ] + Jne [ρ] + d r1 d3 r2 +, Vnn . 2 R3 R3 r12 Z h̄xc (r1 ; r2 ) = 1 hxc (r1 ; r2 ; λ) dλ : 0 coupling-strength integrated xc hole: it has the same formal properties as the standard xc hole (sum rules, cusp conditions). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 66 / 101 xc Functionals Adiabatic Connection Adiabatic connection formula Through the artificial and smooth coupling of the two systems the following energy expression is derived: Z Z 1 ρ(r1 )h̄xc (r1 ; r2 ) 3 E[ρ] = TS [ρ] + Vne [ρ] + Jne [ρ] + d r1 d3 r2 + Vnn , 2 R3 R3 r12 whereas the equivalent expression, which we already know very well, reads Z Z 1 ρ(r1 )hxc (r1 ; r2 ) 3 E[ρ] = T [ρ] + Vne [ρ] + Jne [ρ] + d r1 d3 r2 +, Vnn . 2 R3 R3 r12 Finally, the xc energy in the adiabatic connection approach reads Exc [ρ] = Łukasz Rajchel (University of Warsaw) ρ(r1 )h̄xc (r1 ; r2 ) 3 d r1 d3 r2 . r12 DFT Warsaw, 2010 66 / 101 xc Functionals Kohn-Sham Machinery How KS method works KS orbitals satisfy KS equations: 1 ˆ fKS (r)ϕi (r) = − ∆r + vne (r) + j(r) + vxc (r) ϕi (r) = i ϕi (r). 2 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 67 / 101 xc Functionals Kohn-Sham Machinery How KS method works KS orbitals satisfy KS equations: 1 ˆ fKS (r)ϕi (r) = − ∆r + vne (r) + j(r) + vxc (r) ϕi (r) = i ϕi (r). 2 We expand the molecular orbitals (MOs) in the atomic orbitals (AOs):, ϕi (r) = M X cji χj (r). j=1 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 67 / 101 xc Functionals Kohn-Sham Machinery How KS method works KS orbitals satisfy KS equations: 1 ˆ fKS (r)ϕi (r) = − ∆r + vne (r) + j(r) + vxc (r) ϕi (r) = i ϕi (r). 2 We expand the molecular orbitals (MOs) in the atomic orbitals (AOs):, ϕi (r) = M X cji χj (r). j=1 Now the KS equations can be cast into a nice M × M matrix form: FKS C = SC, Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 67 / 101 xc Functionals Kohn-Sham Machinery How KS method works KS orbitals satisfy KS equations: 1 ˆ fKS (r)ϕi (r) = − ∆r + vne (r) + j(r) + vxc (r) ϕi (r) = i ϕi (r). 2 We expand the molecular orbitals (MOs) in the atomic orbitals (AOs):, ϕi (r) = M X cji χj (r). j=1 Now the KS equations can be cast into a nice M × M matrix form: FKS C = SC, (FKS )ij = hχi |fˆKS |χj i, (C)ij = cij , (S)ij = hχi |χj i, = M X i IM . i=1 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 67 / 101 Summary Points to remember: HK theorems state that there is indeed one-to-one mapping between the external potential and the ground-state density. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 68 / 101 Summary Points to remember: HK theorems state that there is indeed one-to-one mapping between the external potential and the ground-state density. Although HF theorems state that there is variational principle, in practice we can’t make any use of it since we don’t know the exact xc functional. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 68 / 101 Summary Points to remember: HK theorems state that there is indeed one-to-one mapping between the external potential and the ground-state density. Although HF theorems state that there is variational principle, in practice we can’t make any use of it since we don’t know the exact xc functional. KS method is the central to DFT, like HF to wavefunction theory. It is potentially exact and all the operators it uses are local. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 68 / 101 Summary Points to remember: HK theorems state that there is indeed one-to-one mapping between the external potential and the ground-state density. Although HF theorems state that there is variational principle, in practice we can’t make any use of it since we don’t know the exact xc functional. KS method is the central to DFT, like HF to wavefunction theory. It is potentially exact and all the operators it uses are local. KS introduces the xc energy which contains the correlation kinetic energy, self-interaction correction, correlation and exchange. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 68 / 101 Summary Points to remember: HK theorems state that there is indeed one-to-one mapping between the external potential and the ground-state density. Although HF theorems state that there is variational principle, in practice we can’t make any use of it since we don’t know the exact xc functional. KS method is the central to DFT, like HF to wavefunction theory. It is potentially exact and all the operators it uses are local. KS introduces the xc energy which contains the correlation kinetic energy, self-interaction correction, correlation and exchange. But we don’t know how the exact xc functional looks like, it remains a complete mystery to us. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 68 / 101 Summary Points to remember: HK theorems state that there is indeed one-to-one mapping between the external potential and the ground-state density. Although HF theorems state that there is variational principle, in practice we can’t make any use of it since we don’t know the exact xc functional. KS method is the central to DFT, like HF to wavefunction theory. It is potentially exact and all the operators it uses are local. KS introduces the xc energy which contains the correlation kinetic energy, self-interaction correction, correlation and exchange. But we don’t know how the exact xc functional looks like, it remains a complete mystery to us. The End (for today) Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 68 / 101 Part III DFT in Real Life: Defective Functional Theory Outline of the Talk 9 Approximate xc Functionals Introduction LDA and LSD GGA Hybrid Functionals Beyond GGA Problems of Approximate Functionals Approximate xc Functionals Introduction The desired features of an approximate xc energy functional: a non-empirical derivation, since the principles of quantum mechanics are well-known and sufficient. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 71 / 101 Approximate xc Functionals Introduction The desired features of an approximate xc energy functional: a non-empirical derivation, since the principles of quantum mechanics are well-known and sufficient. universality, since in principle one functional should work for diverse systems (atoms, molecules, solids) with different bonding characters (covalent, ionic, metallic, hydrogen, and van der Waals). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 71 / 101 Approximate xc Functionals Introduction The desired features of an approximate xc energy functional: a non-empirical derivation, since the principles of quantum mechanics are well-known and sufficient. universality, since in principle one functional should work for diverse systems (atoms, molecules, solids) with different bonding characters (covalent, ionic, metallic, hydrogen, and van der Waals). simplicity, since this is our only hope for intuitive understanding and our best hope for practical calculation. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 71 / 101 Approximate xc Functionals Introduction The desired features of an approximate xc energy functional: a non-empirical derivation, since the principles of quantum mechanics are well-known and sufficient. universality, since in principle one functional should work for diverse systems (atoms, molecules, solids) with different bonding characters (covalent, ionic, metallic, hydrogen, and van der Waals). simplicity, since this is our only hope for intuitive understanding and our best hope for practical calculation. accuracy enough to be useful in calculations for real systems. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 71 / 101 Approximate xc Functionals Introduction The desired features of an approximate xc energy functional: a non-empirical derivation, since the principles of quantum mechanics are well-known and sufficient. universality, since in principle one functional should work for diverse systems (atoms, molecules, solids) with different bonding characters (covalent, ionic, metallic, hydrogen, and van der Waals). simplicity, since this is our only hope for intuitive understanding and our best hope for practical calculation. accuracy enough to be useful in calculations for real systems. Source: [Perdew and Kurt(2003)] Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 71 / 101 Approximate xc Functionals LDA and LSD Uniform electron gas model Uniform electron gas (jellium) is the central model on which almost all approximate xc functionals are based. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 72 / 101 Approximate xc Functionals LDA and LSD Uniform electron gas model Uniform electron gas (jellium) is the central model on which almost all approximate xc functionals are based. Electrons move in the external potential from uniformly distributed background positive charge (positive jelly background). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 72 / 101 Approximate xc Functionals LDA and LSD Uniform electron gas model Uniform electron gas (jellium) is the central model on which almost all approximate xc functionals are based. Electrons move in the external potential from uniformly distributed background positive charge (positive jelly background). Number of electrons N and the volume of electron gas V are infinite, but ∀r : ρ(r) = N V = const. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 72 / 101 Approximate xc Functionals LDA and LSD Uniform electron gas model Uniform electron gas (jellium) is the central model on which almost all approximate xc functionals are based. Electrons move in the external potential from uniformly distributed background positive charge (positive jelly background). Number of electrons N and the volume of electron gas V are infinite, but ∀r : ρ(r) = N V = const. It is quite a good model of metals with positive cores smeared out to obtain the uniform background positive charge. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 72 / 101 Approximate xc Functionals LDA and LSD Uniform electron gas model Uniform electron gas (jellium) is the central model on which almost all approximate xc functionals are based. Electrons move in the external potential from uniformly distributed background positive charge (positive jelly background). Number of electrons N and the volume of electron gas V are infinite, but ∀r : ρ(r) = N V = const. It is quite a good model of metals with positive cores smeared out to obtain the uniform background positive charge. Of course, the electron density in atoms and molecules can change drastically with r and is far from being homogeneous. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 72 / 101 Approximate xc Functionals LDA and LSD Uniform electron gas model Uniform electron gas (jellium) is the central model on which almost all approximate xc functionals are based. Electrons move in the external potential from uniformly distributed background positive charge (positive jelly background). Number of electrons N and the volume of electron gas V are infinite, but ∀r : ρ(r) = N V = const. It is quite a good model of metals with positive cores smeared out to obtain the uniform background positive charge. Of course, the electron density in atoms and molecules can change drastically with r and is far from being homogeneous. But it’s the only system for which we know the explicit functionals for kinetic energy, exchange ( Thomas-Fermi(-Dirac) models ) and, to very high accuracy, correlation. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 72 / 101 Approximate xc Functionals LDA and LSD Local density approximation (LDA) In the LDA the xc energy is assumed to be Z LDA Exc [ρ] = ρ(r)0xc ρ(r) d3 r R3 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 73 / 101 Approximate xc Functionals LDA and LSD Local density approximation (LDA) In the LDA the xc energy is assumed to be Z LDA Exc [ρ] = ρ(r)0xc ρ(r) d3 r R3 0xc ρ(r) — xc energy density in uniform electron gas model (depends only on the density). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 73 / 101 Approximate xc Functionals LDA and LSD Local density approximation (LDA) In the LDA the xc energy is assumed to be Z LDA Exc [ρ] = ρ(r)0xc ρ(r) d3 r R3 0xc ρ(r) — xc energy density in uniform electron gas model (depends only on the density). It splits into exchange and correlation parts 0xc ρ(r) = 0x ρ(r) + 0c ρ(r) . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 73 / 101 Approximate xc Functionals LDA and LSD Local density approximation (LDA) In the LDA the xc energy is assumed to be Z LDA Exc [ρ] = ρ(r)0xc ρ(r) d3 r R3 0xc ρ(r) — xc energy density in uniform electron gas model (depends only on the density). It splits into exchange and correlation parts 0xc ρ(r) = 0x ρ(r) + 0c ρ(r) . The 0x in uniform electron gas model was given by Dirac in late 1920s: 0x (ρ) = −Cx ρ1/3 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 73 / 101 Approximate xc Functionals LDA and LSD Local density approximation (LDA) In the LDA the xc energy is assumed to be Z LDA Exc [ρ] = ρ(r)0xc ρ(r) d3 r R3 0xc ρ(r) — xc energy density in uniform electron gas model (depends only on the density). It splits into exchange and correlation parts 0xc ρ(r) = 0x ρ(r) + 0c ρ(r) . The 0x in uniform electron gas model was given by Dirac in late 1920s: Z LDA 1/3 0 ρ4/3 (r) d3 r. x (ρ) = −Cx ρ ⇒ Ex [ρ] = −Cx R3 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 73 / 101 Approximate xc Functionals LDA and LSD Local density approximation (LDA) In the LDA the xc energy is assumed to be Z LDA Exc [ρ] = ρ(r)0xc ρ(r) d3 r R3 0xc ρ(r) — xc energy density in uniform electron gas model (depends only on the density). It splits into exchange and correlation parts 0xc ρ(r) = 0x ρ(r) + 0c ρ(r) . The 0x in uniform electron gas model was given by Dirac in late 1920s: Z LDA 1/3 0 ρ4/3 (r) d3 r. x (ρ) = −Cx ρ ⇒ Ex [ρ] = −Cx R3 But we don’t know the explicit form for c ρ(r) . However, sophisticated analytical fits to the numerical Monte Carlo simulations results are available and are termed as VWN (for Vosko, Wilk and Nusair who obtained the fits). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 73 / 101 Approximate xc Functionals LDA and LSD Local spin-density approximation (LSD) In the unrestricted version of KS model there two densities, for spin-up and spin-down electrons, respectively, summing to the total electron density: ρ(r) = ρα (r) + ρβ (r). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 74 / 101 Approximate xc Functionals LDA and LSD Local spin-density approximation (LSD) In the unrestricted version of KS model there two densities, for spin-up and spin-down electrons, respectively, summing to the total electron density: ρ(r) = ρα (r) + ρβ (r). In the LSD the xc energy depends on the two densities Z LSD Exc [ρα ; ρβ ] = ρ(r)xc ρ(r); ζ(r) d3 r. R3 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 74 / 101 Approximate xc Functionals LDA and LSD Local spin-density approximation (LSD) In the unrestricted version of KS model there two densities, for spin-up and spin-down electrons, respectively, summing to the total electron density: ρ(r) = ρα (r) + ρβ (r). In the LSD the xc energy depends on the two densities Z LSD Exc [ρα ; ρβ ] = ρ(r)xc ρ(r); ζ(r) d3 r. R3 Spin-polarization parameter: ( 0, spin-compenstated case (closed-shell). ρα (r) − ρβ (r) = ζ(r) = ρ(r) 1, completely spin-polarized ferromagnetic case. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 74 / 101 Approximate xc Functionals LDA and LSD Local spin-density approximation (LSD) In the unrestricted version of KS model there two densities, for spin-up and spin-down electrons, respectively, summing to the total electron density: ρ(r) = ρα (r) + ρβ (r). In the LSD the xc energy depends on the two densities Z LSD Exc [ρα ; ρβ ] = ρ(r)xc ρ(r); ζ(r) d3 r. R3 Spin-polarization parameter: ( 0, spin-compenstated case (closed-shell). ρα (r) − ρβ (r) = ζ(r) = ρ(r) 1, completely spin-polarized ferromagnetic case. Again, we only know the explicit expression for the exchange energy density: x (ρ; ζ = 0x (ρ) + Ax x (ρ; 1) − 0x (ρ) (1 + ζ)4/3 + (1 − ζ)4/3 − 2 . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 74 / 101 Approximate xc Functionals LDA and LSD Remarks on LDA and LSD The separation of the total density into spin-up and spin-down components is somewhat artificial as the exact xc functional will depend on the total density only. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 75 / 101 Approximate xc Functionals LDA and LSD Remarks on LDA and LSD The separation of the total density into spin-up and spin-down components is somewhat artificial as the exact xc functional will depend on the total density only. However, the division of ρ into ρα and ρβ : Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 75 / 101 Approximate xc Functionals LDA and LSD Remarks on LDA and LSD The separation of the total density into spin-up and spin-down components is somewhat artificial as the exact xc functional will depend on the total density only. However, the division of ρ into ρα and ρβ : I is necessary for spin-dependent external potential (e.g. magnetic field coupling to electronic spin). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 75 / 101 Approximate xc Functionals LDA and LSD Remarks on LDA and LSD The separation of the total density into spin-up and spin-down components is somewhat artificial as the exact xc functional will depend on the total density only. However, the division of ρ into ρα and ρβ : I I is necessary for spin-dependent external potential (e.g. magnetic field coupling to electronic spin). is needed if we are interested in the physical spin magnetization (e.g. in magnetic materials). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 75 / 101 Approximate xc Functionals LDA and LSD Remarks on LDA and LSD The separation of the total density into spin-up and spin-down components is somewhat artificial as the exact xc functional will depend on the total density only. However, the division of ρ into ρα and ρβ : I I I is necessary for spin-dependent external potential (e.g. magnetic field coupling to electronic spin). is needed if we are interested in the physical spin magnetization (e.g. in magnetic materials). allows for more flexibility in the approximate functionals which typically perform better when we use the two densities instead of just one. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 75 / 101 Approximate xc Functionals LDA and LSD Remarks on LDA and LSD The way we calculate the xc energy in LDA/LSD means we assume that the xc potentials depend only on the local values of density. But the density in real systems, atoms and molecules, often varies drastically with r. / Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 75 / 101 Approximate xc Functionals LDA and LSD Remarks on LDA and LSD The way we calculate the xc energy in LDA/LSD means we assume that the xc potentials depend only on the local values of density. But the density in real systems, atoms and molecules, often varies drastically with r. / It turns out that the xc hole in the uniform electron gas model, on wich LDA/LSD is based, satisfies the formal properties of the exact xc hole. , Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 75 / 101 Approximate xc Functionals LDA and LSD Remarks on LDA and LSD The way we calculate the xc energy in LDA/LSD means we assume that the xc potentials depend only on the local values of density. But the density in real systems, atoms and molecules, often varies drastically with r. / It turns out that the xc hole in the uniform electron gas model, on wich LDA/LSD is based, satisfies the formal properties of the exact xc hole. , Since LDA is a special case of LSD for spin-compensated cases, from now on we will refer to both methods as LSD. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 75 / 101 Approximate xc Functionals LDA and LSD Remarks on LDA and LSD The way we calculate the xc energy in LDA/LSD means we assume that the xc potentials depend only on the local values of density. But the density in real systems, atoms and molecules, often varies drastically with r. / It turns out that the xc hole in the uniform electron gas model, on wich LDA/LSD is based, satisfies the formal properties of the exact xc hole. , Since LDA is a special case of LSD for spin-compensated cases, from now on we will refer to both methods as LSD. LSD has been extensively popular in the solid state physics. But for the sparse matter which we have to do with in chemistry there was a need to go beyond the local approximation. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 75 / 101 Approximate xc Functionals GGA Beyond LSD The situation of people is totally different when they are on a steady, plain terrain with (almost) uniform density. . . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 76 / 101 Approximate xc Functionals GGA Beyond LSD . . . then when they are put in a region with very rapidly changing density! Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 76 / 101 Approximate xc Functionals GGA Beyond LSD In the LSD we used only the information on the density at the specific point to calculate the contribution to xc energy. . . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 76 / 101 Approximate xc Functionals GGA Beyond LSD In the LSD we used only the information on the density at the specific point to calculate the contribution to xc energy. . . so the obvious next step is to supplement that with the information on how density changes in that point. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 76 / 101 Approximate xc Functionals GGA Beyond LSD In the LSD we used only the information on the density at the specific point to calculate the contribution to xc energy. . . so the obvious next step is to supplement that with the information on how density changes in that point. That information is stored in the density gradient: ∂ρ 0 ρ vector pointing in the direction x ∂x ∂ρ 0 ∇ρ = ∂y = ρy ← of the greatest rate ∂ρ ρ0z of the increase of the density. ∂z Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 76 / 101 Approximate xc Functionals GGA Beyond LSD In the LSD we used only the information on the density at the specific point to calculate the contribution to xc energy. . . so the obvious next step is to supplement that with the information on how density changes in that point. That information is stored in the density gradient: ∂ρ 0 ρ vector pointing in the direction x ∂x ∂ρ 0 ∇ρ = ∂y = ρy ← of the greatest rate ∂ρ ρ0z of the increase of the density. ∂z The gradient magnitude (scalar!) gives the rate of the greatest change of the density: q p |∇ρ| = ∇ρ · ∇ρ = (ρ0x )2 + (ρ0y )2 + (ρ0z )2 . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 76 / 101 Approximate xc Functionals GGA Generalized gradient approximation (GGA) After insertion of gradients into the xc functional it turned out that the results are even worse than for LSD! Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 77 / 101 Approximate xc Functionals GGA Generalized gradient approximation (GGA) After insertion of gradients into the xc functional it turned out that the results are even worse than for LSD! This is because in such an approach the xc holes no longer satisfy formal properties as was the case for LSD. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 77 / 101 Approximate xc Functionals GGA Generalized gradient approximation (GGA) After insertion of gradients into the xc functional it turned out that the results are even worse than for LSD! This is because in such an approach the xc holes no longer satisfy formal properties as was the case for LSD. So, let’s be brutal: enforce the resulting holes to satisfy the formal properties by truncating them in the regions where they misbehave. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 77 / 101 Approximate xc Functionals GGA Generalized gradient approximation (GGA) After insertion of gradients into the xc functional it turned out that the results are even worse than for LSD! This is because in such an approach the xc holes no longer satisfy formal properties as was the case for LSD. So, let’s be brutal: enforce the resulting holes to satisfy the formal properties by truncating them in the regions where they misbehave. With the hope to correct the LSD we now introduce the gradient into the xc functional and correct the xc holes where necessary — this way we obtain the generalized gradient approximation to the xc energy: Z GGA GGA Exc [ρα ; ρβ ] = fxc ρα (r); ρβ (r); ∇ρα (r); ∇ρβ (r) d3 r. R3 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 77 / 101 Approximate xc Functionals GGA Generalized gradient approximation (GGA) After insertion of gradients into the xc functional it turned out that the results are even worse than for LSD! This is because in such an approach the xc holes no longer satisfy formal properties as was the case for LSD. So, let’s be brutal: enforce the resulting holes to satisfy the formal properties by truncating them in the regions where they misbehave. With the hope to correct the LSD we now introduce the gradient into the xc functional and correct the xc holes where necessary — this way we obtain the generalized gradient approximation to the xc energy: Z GGA GGA Exc [ρα ; ρβ ] = fxc ρα (r); ρβ (r); ∇ρα (r); ∇ρβ (r) d3 r. R3 GGA = E GGA + E GGA . As usual, we split the energy: Exc x c Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 77 / 101 Approximate xc Functionals GGA Exchange in GGA The exchange energy in GGA is usually assumed to be composed of LSD part plus some correction: XZ GGA LSD F sσ (r) ρ4/3 (r) d3 r, Ex [ρα ; ρβ ] = Ex [ρα ; ρβ ] − σ Łukasz Rajchel (University of Warsaw) DFT R3 Warsaw, 2010 78 / 101 Approximate xc Functionals GGA Exchange in GGA The exchange energy in GGA is usually assumed to be composed of LSD part plus some correction: XZ GGA LSD F sσ (r) ρ4/3 (r) d3 r, Ex [ρα ; ρβ ] = Ex [ρα ; ρβ ] − σ R3 where the reduced density gradient is a measure of local density inhomogeneity: sσ (r) = |∇ρ(r)| ρ4/3 (r) Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 78 / 101 Approximate xc Functionals GGA Exchange in GGA The exchange energy in GGA is usually assumed to be composed of LSD part plus some correction: XZ GGA LSD F sσ (r) ρ4/3 (r) d3 r, Ex [ρα ; ρβ ] = Ex [ρα ; ρβ ] − σ R3 where the reduced density gradient is a measure of local density inhomogeneity: it is large for large density gradients (regions of rapidly changing density) |∇ρ(r)| ← sσ (r) = 4/3 ρ (r) and for small densities (tails of density far from nuclei). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 78 / 101 Approximate xc Functionals GGA Exchange in GGA — examples: FB = βs2σ , β = 4.2 · 10−3 1 + 6βsσ sinh−1 sσ [Becke(1988)] β obtained by a least-squares fit to the exactly known exchange energies of the rare gas atoms He through Rn. The functional designed to recover the exchange energy density asymptotically far from a finite system. Sum rules for the exchange hole fulfilled. Empirical. Similar functionals: PW91, CAM(A), CAM(B), FT97. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 79 / 101 Approximate xc Functionals GGA Exchange in GGA — examples: 2 F PW91 1 + 0.19645sσ sinh−1 7.7956sσ + (0.2743 − 0.1508e−100sσ )s2σ = 1 + 0.19645sσ sinh−1 7.7956sσ + 0.004s4σ [Perdew et al.(1992)Perdew, Chevary, Vosko, Jackson, Pederson, Singh, and F The analytical fit to the second-order density-gradient expansion for the xc hole surrounding the electron in a system of slowly varying density. The spurious long-range parts of the xc hole cut off to satisfy sum rules on the exact hole. According to Perdew, overparametrized. Non-empirical. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 79 / 101 Approximate xc Functionals GGA Exchange in GGA — examples: F PBE κ =κ− , 1 + µκ s2σ ( κ = 0.804 µ = 0.21951 [Perdew et al.(1996)Perdew, Burke, and Ernzerhof] κ set to the maximum value allowed by the local Lieb-Oxford bound. µ set to recover the linear response of the uniform electron gas. Non-empirical. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 79 / 101 Approximate xc Functionals GGA Correlation in GGA EcGGA ’s have a very complicated analytical form and cannot be understood by simple physically motivated reasonings. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 80 / 101 Approximate xc Functionals GGA Correlation in GGA EcGGA ’s have a very complicated analytical form and cannot be understood by simple physically motivated reasonings. Some examples: Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 80 / 101 Approximate xc Functionals GGA Correlation in GGA EcGGA ’s have a very complicated analytical form and cannot be understood by simple physically motivated reasonings. Some examples: P86C: includes parameter fitted to the correlation energy of Ne. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 80 / 101 Approximate xc Functionals GGA Correlation in GGA EcGGA ’s have a very complicated analytical form and cannot be understood by simple physically motivated reasonings. Some examples: P86C: includes parameter fitted to the correlation energy of Ne. PW91C: based on xc hole investigation. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 80 / 101 Approximate xc Functionals GGA Correlation in GGA EcGGA ’s have a very complicated analytical form and cannot be understood by simple physically motivated reasonings. Some examples: P86C: includes parameter fitted to the correlation energy of Ne. PW91C: based on xc hole investigation. LYP: derived from an expression for the correlation energy of He from accurate ab initio calculations. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 80 / 101 Approximate xc Functionals GGA LSD/GGA results Exc for atoms Atom H He Li Be N Ne LSD −0.29 −1.00 −1.69 −2.54 −6.32 −11.78 GGA −0.31 −1.06 −1.81 −2.72 −6.73 −12.42 exact −0.31 −1.09 −1.83 −2.76 −6.78 −12.50 LSD: VWN for correlation, GGA: PBE for correlation and exchange Source: [Perdew and Kurt(2003)] Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 81 / 101 Approximate xc Functionals GGA LSD/GGA results Atomization energies for molecules Molecule H2 CH4 NH3 H2 O CO O2 LSD 0.18 0.735 0.537 0.426 0.478 0.279 GGA 0.169 0.669 0.481 0.371 0.43 0.228 exact 0.173 0.669 0.474 0.371 0.412 0.191 LSD: VWN for correlation, GGA: PBE for correlation and exchange Source: [Perdew and Kurt(2003)] Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 81 / 101 Approximate xc Functionals Hybrid Functionals Exact exchange Although we don’t know exact xc functional, it’s clear from numerical experience that the exchange dominates the correlation: |Ex | >> |Ec |. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 82 / 101 Approximate xc Functionals Hybrid Functionals Exact exchange Although we don’t know exact xc functional, it’s clear from numerical experience that the exchange dominates the correlation: |Ex | >> |Ec |. Thus, designing appropriate exchange functional is crucial to getting meaningful results from KS method. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 82 / 101 Approximate xc Functionals Hybrid Functionals Exact exchange Although we don’t know exact xc functional, it’s clear from numerical experience that the exchange dominates the correlation: |Ex | >> |Ec |. Thus, designing appropriate exchange functional is crucial to getting meaningful results from KS method. From HF theory we know the exact expression for the exchange resulting from single Slater determinant: Z Z 1 ρ(r1 ; r2 )ρ(r2 ; r1 ) 3 exact d r1 d3 r2 . Ex [ρ] = − 4 R3 R3 r12 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 82 / 101 Approximate xc Functionals Hybrid Functionals Exact exchange Although we don’t know exact xc functional, it’s clear from numerical experience that the exchange dominates the correlation: |Ex | >> |Ec |. Thus, designing appropriate exchange functional is crucial to getting meaningful results from KS method. From HF theory we know the exact expression for the exchange resulting from single Slater determinant: Z Z 1 ρ(r1 ; r2 )ρ(r2 ; r1 ) 3 exact d r1 d3 r2 . Ex [ρ] = − 4 R3 R3 r12 This exchange is termed exact in DFT jargon, though it’s different than the exchange in HF model as the one-matrix ρ(r; r0 ) used here is that of KS model, which doesn’t equal that of HF model. Also, as we remember, that exchange is non-local. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 82 / 101 Approximate xc Functionals Hybrid Functionals Exact exchange problems So, why not just mix the exact exchange with the GGA correlation: Exc = Exexact + EcGGA . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 83 / 101 Approximate xc Functionals Hybrid Functionals Exact exchange problems So, why not just mix the exact exchange with the GGA correlation: Exc = Exexact + EcGGA . This at first nice idea proves to yield results even worse than HF! Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 83 / 101 Approximate xc Functionals Hybrid Functionals Exact exchange problems So, why not just mix the exact exchange with the GGA correlation: Exc = Exexact + EcGGA . This at first nice idea proves to yield results even worse than HF! That’s because full exact exchange is incompatible with GGA correlation: Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 83 / 101 Approximate xc Functionals Hybrid Functionals Exact exchange problems So, why not just mix the exact exchange with the GGA correlation: Exc = Exexact + EcGGA . This at first nice idea proves to yield results even worse than HF! That’s because full exact exchange is incompatible with GGA correlation: the exact exchange hole in a molecule usually has a highly nonlocal, multi-center character. xc hole for H2 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 83 / 101 Approximate xc Functionals Hybrid Functionals Exact exchange problems So, why not just mix the exact exchange with the GGA correlation: Exc = Exexact + EcGGA . This at first nice idea proves to yield results even worse than HF! That’s because full exact exchange is incompatible with GGA correlation: the exact exchange hole in a molecule usually has a highly nonlocal, multi-center character. xc hole for H2 this is cancelled by an almost equal, but opposite, nonlocal and multicenter character in the exact correlation hole. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 83 / 101 Approximate xc Functionals Hybrid Functionals Exact exchange problems So, why not just mix the exact exchange with the GGA correlation: Exc = Exexact + EcGGA . This at first nice idea proves to yield results even worse than HF! That’s because full exact exchange is incompatible with GGA correlation: the exact exchange hole in a molecule usually has a highly nonlocal, multi-center character. xc hole for H2 this is cancelled by an almost equal, but opposite, nonlocal and multicenter character in the exact correlation hole. so, the exact xc hole is localized around the reference electron. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 83 / 101 Approximate xc Functionals Hybrid Functionals Exact exchange problems So, why not just mix the exact exchange with the GGA correlation: Exc = Exexact + EcGGA . This at first nice idea proves to yield results even worse than HF! That’s because full exact exchange is incompatible with GGA correlation: the exact exchange hole in a molecule usually has a highly nonlocal, multi-center character. xc hole for H2 this is cancelled by an almost equal, but opposite, nonlocal and multicenter character in the exact correlation hole. so, the exact xc hole is localized around the reference electron. the GGA-approximated exchange and correlation holes are more localized around the reference electron. . . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 83 / 101 Approximate xc Functionals Hybrid Functionals Exact exchange problems So, why not just mix the exact exchange with the GGA correlation: Exc = Exexact + EcGGA . This at first nice idea proves to yield results even worse than HF! That’s because full exact exchange is incompatible with GGA correlation: the exact exchange hole in a molecule usually has a highly nonlocal, multi-center character. xc hole for H2 this is cancelled by an almost equal, but opposite, nonlocal and multicenter character in the exact correlation hole. so, the exact xc hole is localized around the reference electron. the GGA-approximated exchange and correlation holes are more localized around the reference electron. . . and finally, mixing the full exact exchange hole with the local GGA correlation hole results in non-local xc hole, which can’t model the locality of the exact xc hole. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 83 / 101 Approximate xc Functionals Hybrid Functionals Hybrid functionals Adding the full exact exchange doesn’t work well, but we know that that exchange properly describes the non-interacting system. So, instead of full exact exchange, let’s just combine some fraction of it with the GGA counterparts. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 84 / 101 Approximate xc Functionals Hybrid Functionals Hybrid functionals Adding the full exact exchange doesn’t work well, but we know that that exchange properly describes the non-interacting system. So, instead of full exact exchange, let’s just combine some fraction of it with the GGA counterparts. That’s how we obtain the hybrid functionals. Generally, hyb Exc =a Exexact | {z } exact non-local exchange Łukasz Rajchel (University of Warsaw) + (1 − a) ExGGA | {z } GGA local exchange DFT + EcGGA | {z } , a < 1. GGA local correlation Warsaw, 2010 84 / 101 Approximate xc Functionals Hybrid Functionals Hybrid functionals Adding the full exact exchange doesn’t work well, but we know that that exchange properly describes the non-interacting system. So, instead of full exact exchange, let’s just combine some fraction of it with the GGA counterparts. That’s how we obtain the hybrid functionals. Generally, hyb Exc =a Exexact | {z } exact non-local exchange + (1 − a) ExGGA | {z } GGA local exchange + EcGGA | {z } , a < 1. GGA local correlation There are now plenty of hybrid functionals available. Some variations involve mixtures of three kinds of exchange: exact one, LSD one (called Slater exchange), and GGA local one. Accordingly, they involve more parameters than just one (a). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 84 / 101 Approximate xc Functionals Hybrid Functionals Examples of hybrid functionals B3 Exc a = 0.20 exact LSD B88 LSD PW91 = aEx + (1 − a)Ex + bEx + Ec + Ec , b = 0.72 c = 0.81 [Becke(1993)] Parameters a, b and c chosen to optimally reproduce the atomization and ionization energies and proton affinities from the G2 thermochemical database. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 85 / 101 Approximate xc Functionals Hybrid Functionals Examples of hybrid functionals B3LYP Exc a = 0.20 exact LSD B88 LYP LSD = aEx +(1−a)Ex +bEx +cEc +(1−c)Ec , b = 0.72 c = 0.81 [Stephens et al.(1994)Stephens, Devlin, Chabalowski, and Frisch] Parameters a, b and c take from the B3 functional. Particularly good results for vibrational spectra. Undeniably the most popular and widely used functional in DFT. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 85 / 101 Approximate xc Functionals Hybrid Functionals Examples of hybrid functionals PBE0 Exc = aExexact + (1 − a)ExPBE + EcPBE , a = 0.25 [Adamo and Barone(1999)] The value of a deducted from perturbation theory. Promising performance for all important properties. Competitive with the most reliable, empirically parameterized functionals. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 85 / 101 Approximate xc Functionals Hybrid Functionals Hybrid functionals results Properties of H2 O molecule: experimental values and deviation from experiment for different levels of theory Property Exp. HF MP2 ROH /Å νs /cm−1 νas /cm−1 µ/D hαi/Å3 0.957 3832 3943 1.854 1.427 −0.016 288 279 0.084 −0.207 0.004 −9 5 0.006 −0.004 SVWN 0.013 −106 −107 0.005 0.109 BLYP 0.015 −177 −186 −0.051 0.143 Functionals SLYP BVWN 0.019 0.010 −155 −132 −156 −142 0.007 −0.052 0.179 0.075 B3LYP 0.005 −33 −42 −0.006 0.026 MP2 — Møller–Plesset perturbation theory Source: [Koch and Holthausen(2001)] Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 86 / 101 Approximate xc Functionals Hybrid Functionals Hybrid functionals results Dipole moment for different molecules: calculations vs. experiment 0.15 (µcalculated - µexp)/au HF MP2 BLYP 0.1 HCTH B3LYP 0.05 0 -0.05 -0.1 -0.15 SO PH 3 2 F H3 N l H Li Li F C H H S H2 O H2 O C molecule Source: [Cohen and Tantirungrotechai(1999)] Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 86 / 101 Approximate xc Functionals Beyond GGA Meta-generalized gradient approximation (MGGA) The next step to improve functionals is to introduce the Laplacians and kinetic energy density into the functional — this is the meta-generalized gradient approximation scheme: Z MGGA MGGA Exc [ρα ; ρβ ] = fxc ρα ; ρβ ; ∇ρα ; ∇ρβ ; ∆ρα ; ∆ρβ ; τα ; τβ d3 r, R3 N/2 1X τσ = |∇ϕiσ |2 — kinetic energy density of the σ-occupied orbitals. 2 i=1 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 87 / 101 Approximate xc Functionals Beyond GGA Meta-generalized gradient approximation (MGGA) The next step to improve functionals is to introduce the Laplacians and kinetic energy density into the functional — this is the meta-generalized gradient approximation scheme: Z MGGA MGGA Exc [ρα ; ρβ ] = fxc ρα ; ρβ ; ∇ρα ; ∇ρβ ; ∆ρα ; ∆ρβ ; τα ; τβ d3 r, R3 N/2 1X τσ = |∇ϕiσ |2 — kinetic energy density of the σ-occupied orbitals. 2 i=1 Several meta-GGA’s have been constructed by a combination of theoretical constraints and fitting to chemical data. Some of them contain as many as 20 parameters! Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 87 / 101 Approximate xc Functionals Beyond GGA Meta-generalized gradient approximation (MGGA) The next step to improve functionals is to introduce the Laplacians and kinetic energy density into the functional — this is the meta-generalized gradient approximation scheme: Z MGGA MGGA Exc [ρα ; ρβ ] = fxc ρα ; ρβ ; ∇ρα ; ∇ρβ ; ∆ρα ; ∆ρβ ; τα ; τβ d3 r, R3 N/2 1X τσ = |∇ϕiσ |2 — kinetic energy density of the σ-occupied orbitals. 2 i=1 Several meta-GGA’s have been constructed by a combination of theoretical constraints and fitting to chemical data. Some of them contain as many as 20 parameters! Examples: PKZB (only one empirical parameter), TPSS (fully non-empirical). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 87 / 101 Approximate xc Functionals Beyond GGA Meta-generalized gradient approximation (MGGA) The next step to improve functionals is to introduce the Laplacians and kinetic energy density into the functional — this is the meta-generalized gradient approximation scheme: Z MGGA MGGA Exc [ρα ; ρβ ] = fxc ρα ; ρβ ; ∇ρα ; ∇ρβ ; ∆ρα ; ∆ρβ ; τα ; τβ d3 r, R3 N/2 1X τσ = |∇ϕiσ |2 — kinetic energy density of the σ-occupied orbitals. 2 i=1 Several meta-GGA’s have been constructed by a combination of theoretical constraints and fitting to chemical data. Some of them contain as many as 20 parameters! Examples: PKZB (only one empirical parameter), TPSS (fully non-empirical). They usually perform better than LSD’s and GGA’s, but there are exceptions (e.g., surface energies and lattice constants are less correct). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 87 / 101 Approximate xc Functionals Beyond GGA MGGA results Statistical summary of the errors of density functionals for various properties of molecules and solids Property Test set LSD Atomization en./(kcal/mol) Ionization en./eV Electron affinity/eV Bond length/Å Harmonic frequency G2 (148 mols.) G2 (86 species) G2 (58 species) 96 molecules 82 diatomics 83.8 0.22 0.26 0.013 48.9 GGA PBE PBE0 17.1 5.1 0.22 0.20 0.12 0.17 0.016 0.010 42.0 43.6 MGGA PKZB TPSS 4.4 6.2 0.29 0.23 0.14 0.14 0.027 0.014 51.7 30.4 Source: [Tao et al.(2003)Tao, Perdew, Staroverov, and Scuseria] Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 88 / 101 Approximate xc Functionals Beyond GGA Hyper-GGA and beyond In hyper-GGA the MGGA functional is appended with the exact exchange energy densities: HGGA Exc [ρα ; ρβ ] = Z HGGA fxc ρα ; ρβ ; ∇ρα ; ∇ρβ ; ∆ρα ; ∆ρβ ; τα ; τβ ; xα ; xβ d3 r, R3 1 xα (r) = − 2ρσ (r) Łukasz Rajchel (University of Warsaw) Z DFT R3 ρσ (r; r0 ) 3 0 d r. |r − r0 | Warsaw, 2010 89 / 101 Approximate xc Functionals Beyond GGA Hyper-GGA and beyond In hyper-GGA the MGGA functional is appended with the exact exchange energy densities: HGGA Exc [ρα ; ρβ ] = Z HGGA fxc ρα ; ρβ ; ∇ρα ; ∇ρβ ; ∆ρα ; ∆ρβ ; τα ; τβ ; xα ; xβ d3 r, R3 1 xα (r) = − 2ρσ (r) Z R3 ρσ (r; r0 ) 3 0 d r. |r − r0 | Semiempirical hyper-GGAs include the widely used global hybrid functionals such as B3LYP, B3PW91, or PBE0 that mix a fixed fraction of exact exchange with GGA exchange, and the local hybrids, though these functionals do not use all the ingredients prescribed above. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 89 / 101 Approximate xc Functionals Beyond GGA Hyper-GGA and beyond In hyper-GGA the MGGA functional is appended with the exact exchange energy densities: HGGA Exc [ρα ; ρβ ] = Z HGGA fxc ρα ; ρβ ; ∇ρα ; ∇ρβ ; ∆ρα ; ∆ρβ ; τα ; τβ ; xα ; xβ d3 r, R3 1 xα (r) = − 2ρσ (r) Z R3 ρσ (r; r0 ) 3 0 d r. |r − r0 | Semiempirical hyper-GGAs include the widely used global hybrid functionals such as B3LYP, B3PW91, or PBE0 that mix a fixed fraction of exact exchange with GGA exchange, and the local hybrids, though these functionals do not use all the ingredients prescribed above. Finally, to obtain the chemical accuracy, we can incorporate all the Kohn-Sham orbitals (occupied and virtual) into the functional. That requires huge basis sets and is not yet ready for practical use. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 89 / 101 Approximate xc Functionals Beyond GGA Jacob’s Ladder HEAVEN OF CHEMICAL ACCURACY virtual {ϕa } full orbital-based DFT x hyper-GGA ∇2 ρ, τ meta-GGA ∇ρ GGA ρ LSD HARTREE WORLD The xc functional approximations were arranged by J. P. Perdew with growing accuracy as rungs of a ladder. We can climb that ladder to get to the heaven of chemical accuracy, an analogy to biblical Jacob’s Ladder: Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 90 / 101 Approximate xc Functionals Problems of Approximate Functionals Self-interaction In the HF model the non-physical self-interaction of the Coulomb e-e repulsion is removed by the exchange, so for hydrogen atom we always get J[ρ] + Ex [ρ] = 0. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 91 / 101 Approximate xc Functionals Problems of Approximate Functionals Self-interaction In the HF model the non-physical self-interaction of the Coulomb e-e repulsion is removed by the exchange, so for hydrogen atom we always get J[ρ] + Ex [ρ] = 0. But it’s not the case for most of the approximate xc functionals: here are the results for hydrogen atom for several functionals: Functional SVWN BLYP B3LYP BP86 BPW91 HF J[ρ] 0.29975 0.30747 0.30845 0.30653 0.30890 0.31250 Łukasz Rajchel (University of Warsaw) Ex [ρ] −0.25753 −0.30607 −0.30370 −0.30479 −0.30719 −0.31250 DFT Ec [ρ] −0.03945 0.0 −0.00756 −0.00248 −0.00631 0.0 J[ρ] + Exc [ρ] 0.00277 0.00140 −0.00281 −0.00074 −0.00460 0.0 Warsaw, 2010 91 / 101 Approximate xc Functionals Problems of Approximate Functionals Self-interaction In the HF model the non-physical self-interaction of the Coulomb e-e repulsion is removed by the exchange, so for hydrogen atom we always get J[ρ] + Ex [ρ] = 0. But it’s not the case for most of the approximate xc functionals: here are the results for hydrogen atom for several functionals: Functional SVWN BLYP B3LYP BP86 BPW91 HF J[ρ] 0.29975 0.30747 0.30845 0.30653 0.30890 0.31250 Ex [ρ] −0.25753 −0.30607 −0.30370 −0.30479 −0.30719 −0.31250 Ec [ρ] −0.03945 0.0 −0.00756 −0.00248 −0.00631 0.0 J[ρ] + Exc [ρ] 0.00277 0.00140 −0.00281 −0.00074 −0.00460 0.0 To alleviate the problem, several solutions have been proposed, e.g. the self-interaction corrected KS in which the self-interaction is subtracted demanding that J[ρ] = −Exc [ρ] for one-electron systems. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 91 / 101 Approximate xc Functionals Problems of Approximate Functionals xc potential asymptotics An electron in infinite distance P r from other N − 1 electrons and M nuclei of total charge Z = M α=1 Zα sees the potential v(r) = Łukasz Rajchel (University of Warsaw) N −1−Z . r DFT Warsaw, 2010 92 / 101 Approximate xc Functionals Problems of Approximate Functionals xc potential asymptotics An electron in infinite distance P r from other N − 1 electrons and M nuclei of total charge Z = M α=1 Zα sees the potential v(r) = N −1−Z . r The asymptotics of n-e and Coulomb potentials: Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 92 / 101 Approximate xc Functionals Problems of Approximate Functionals xc potential asymptotics An electron in infinite distance P r from other N − 1 electrons and M nuclei of total charge Z = M α=1 Zα sees the potential v(r) = N −1−Z . r The asymptotics of n-e and Coulomb potentials: I lim vne (r) = − lim r→∞ Łukasz Rajchel (University of Warsaw) r→∞ M X Zα Z =− , |r − R | r α α=1 DFT Warsaw, 2010 92 / 101 Approximate xc Functionals Problems of Approximate Functionals xc potential asymptotics An electron in infinite distance P r from other N − 1 electrons and M nuclei of total charge Z = M α=1 Zα sees the potential v(r) = N −1−Z . r The asymptotics of n-e and Coulomb potentials: I I M X Zα Z lim vne (r) = − lim =− , r→∞ r→∞ |r − R | r α Z α=1 0 ρ(r ) 3 0 N lim ̂(r) = lim d r = . r→∞ r→∞ R3 |r − r0 | r Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 92 / 101 Approximate xc Functionals Problems of Approximate Functionals xc potential asymptotics An electron in infinite distance P r from other N − 1 electrons and M nuclei of total charge Z = M α=1 Zα sees the potential v(r) = N −1−Z . r The asymptotics of n-e and Coulomb potentials: I I M X Zα Z lim vne (r) = − lim =− , r→∞ r→∞ |r − R | r α Z α=1 0 ρ(r ) 3 0 N lim ̂(r) = lim d r = . r→∞ r→∞ R3 |r − r0 | r 1 So, the correct asymptotics of xc potential is lim vxc (r) = − . r→∞ r Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 92 / 101 Approximate xc Functionals Problems of Approximate Functionals xc potential asymptotics But that’s true for functionals satisfying the derivative discontinuity behaviour, i.e. potentials which are not continous for the integer electron numbers and continuous for fractional ones. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 92 / 101 Approximate xc Functionals Problems of Approximate Functionals xc potential asymptotics But that’s true for functionals satisfying the derivative discontinuity behaviour, i.e. potentials which are not continous for the integer electron numbers and continuous for fractional ones. The correct asymptotics of the continuous xc potential is 1 lim vxc (r) = − + I + HOMO , r r→∞ I — first ionization energy, HOMO — energy of the highest occupied KS orbital. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 92 / 101 Approximate xc Functionals Problems of Approximate Functionals xc potential asymptotics But that’s true for functionals satisfying the derivative discontinuity behaviour, i.e. potentials which are not continous for the integer electron numbers and continuous for fractional ones. The correct asymptotics of the continuous xc potential is 1 lim vxc (r) = − + I + HOMO , r r→∞ I — first ionization energy, HOMO — energy of the highest occupied KS orbital. Approximate xc functionals vanish exponentially which is too fast. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 92 / 101 Approximate xc Functionals Problems of Approximate Functionals xc potential asymptotics But that’s true for functionals satisfying the derivative discontinuity behaviour, i.e. potentials which are not continous for the integer electron numbers and continuous for fractional ones. The correct asymptotics of the continuous xc potential is 1 lim vxc (r) = − + I + HOMO , r r→∞ I — first ionization energy, HOMO — energy of the highest occupied KS orbital. Approximate xc functionals vanish exponentially which is too fast. That’s why they need the asymptotic correction to properly describe the properties depending on long-range parts of xc potentials (electron affinities, polarizabilities, excitation energies). Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 92 / 101 Approximate xc Functionals Problems of Approximate Functionals xc potential asymptotics Since vxexact has a correct long-range behaviour, the hybrid functionals (with Exexact ) have better asymptotics than the pure local functionals. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 92 / 101 Approximate xc Functionals Problems of Approximate Functionals xc potential asymptotics Since vxexact has a correct long-range behaviour, the hybrid functionals (with Exexact ) have better asymptotics than the pure local functionals. But the inclusion of too much exact exchange leads to problems. . . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 92 / 101 Approximate xc Functionals Problems of Approximate Functionals xc potential asymptotics Since vxexact has a correct long-range behaviour, the hybrid functionals (with Exexact ) have better asymptotics than the pure local functionals. But the inclusion of too much exact exchange leads to problems. . . Now, the idea is simple: preserve the GGA exchange at short-range and activate the exact exchange asymptotically through the range-separated Coulomb operator (ω — switching parameter): 1 = r12 1 − erf(ωr12 ) r12 | {z } short-range Coulomb-like potential Łukasz Rajchel (University of Warsaw) DFT + erf(ωr12 ) r12 | {z } . nonsingular long-range background potential Warsaw, 2010 92 / 101 Approximate xc Functionals Problems of Approximate Functionals xc potential asymptotics Since vxexact has a correct long-range behaviour, the hybrid functionals (with Exexact ) have better asymptotics than the pure local functionals. But the inclusion of too much exact exchange leads to problems. . . Now, the idea is simple: preserve the GGA exchange at short-range and activate the exact exchange asymptotically through the range-separated Coulomb operator (ω — switching parameter): 1 = r12 1 − erf(ωr12 ) r12 | {z } short-range Coulomb-like potential + erf(ωr12 ) r12 | {z } . nonsingular long-range background potential The functionals using this or similar ansatz are termed as long-range-corrected (e.g. CAM-B3LYP). They are often used in TDDFT to calculate excited states related properties. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 92 / 101 Approximate xc Functionals Problems of Approximate Functionals xc potential asymptotics Long-range corrected functionals improve over: linear and nonlinear optical properties of long-chain molecules, Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 92 / 101 Approximate xc Functionals Problems of Approximate Functionals xc potential asymptotics Long-range corrected functionals improve over: linear and nonlinear optical properties of long-chain molecules, the poor description of van der Waals bonds, Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 92 / 101 Approximate xc Functionals Problems of Approximate Functionals xc potential asymptotics Long-range corrected functionals improve over: linear and nonlinear optical properties of long-chain molecules, the poor description of van der Waals bonds, barrier heights, Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 92 / 101 Approximate xc Functionals Problems of Approximate Functionals xc potential asymptotics Long-range corrected functionals improve over: linear and nonlinear optical properties of long-chain molecules, the poor description of van der Waals bonds, barrier heights, charge transfer and Rydberg excitation energies, Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 92 / 101 Approximate xc Functionals Problems of Approximate Functionals xc potential asymptotics Long-range corrected functionals improve over: linear and nonlinear optical properties of long-chain molecules, the poor description of van der Waals bonds, barrier heights, charge transfer and Rydberg excitation energies, and the corresponding oscillator strengths in time-dependent DFT calculations. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 92 / 101 Approximate xc Functionals Problems of Approximate Functionals Points to remember: There is a clear gradation of approximate xc functionals, from LSD to hyper-GGA. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 93 / 101 Approximate xc Functionals Problems of Approximate Functionals Points to remember: There is a clear gradation of approximate xc functionals, from LSD to hyper-GGA. Approximate xc functionals include unphysical self-interaction contribution. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 93 / 101 Approximate xc Functionals Problems of Approximate Functionals Points to remember: There is a clear gradation of approximate xc functionals, from LSD to hyper-GGA. Approximate xc functionals include unphysical self-interaction contribution. Also, their asymptotics is not correct and needs some patches if long-range properties are of interest. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 93 / 101 Approximate xc Functionals Problems of Approximate Functionals Points to remember: There is a clear gradation of approximate xc functionals, from LSD to hyper-GGA. Approximate xc functionals include unphysical self-interaction contribution. Also, their asymptotics is not correct and needs some patches if long-range properties are of interest. The End (for today) Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 93 / 101 Supplement back to symbols Permutation examples: f (x1 ; x2 ) = cos (x1 − x2 ), Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 94 / 101 Supplement back to symbols Permutation examples: f (x1 ; x2 ) = cos (x1 − x2 ), P12 f (x1 ; x2 ) = cos (x2 − x1 ) = cos (x1 − x2 ), Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 94 / 101 Supplement back to symbols Permutation examples: f (x1 ; x2 ) = cos (x1 − x2 ), P12 f (x1 ; x2 ) = cos (x2 − x1 ) = cos (x1 − x2 ), P12 f (x1 ; x2 ) = f (x1 ; x2 ) : symmetric. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 94 / 101 Supplement back to symbols Permutation examples: f (x1 ; x2 ) = cos (x1 − x2 ), P12 f (x1 ; x2 ) = cos (x2 − x1 ) = cos (x1 − x2 ), P12 f (x1 ; x2 ) = f (x1 ; x2 ) : symmetric. g(x1 ; x2 ) = sin (x1 − x2 ), Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 94 / 101 Supplement back to symbols Permutation examples: f (x1 ; x2 ) = cos (x1 − x2 ), P12 f (x1 ; x2 ) = cos (x2 − x1 ) = cos (x1 − x2 ), P12 f (x1 ; x2 ) = f (x1 ; x2 ) : symmetric. g(x1 ; x2 ) = sin (x1 − x2 ), P12 g(x1 ; x2 ) = sin (x2 − x1 ) = − sin (x1 − x2 ), Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 94 / 101 Supplement back to symbols Permutation examples: f (x1 ; x2 ) = cos (x1 − x2 ), P12 f (x1 ; x2 ) = cos (x2 − x1 ) = cos (x1 − x2 ), P12 f (x1 ; x2 ) = f (x1 ; x2 ) : symmetric. g(x1 ; x2 ) = sin (x1 − x2 ), P12 g(x1 ; x2 ) = sin (x2 − x1 ) = − sin (x1 − x2 ), P12 g(x1 ; x2 ) = −g(x1 ; x2 ) : antisymmetric. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 94 / 101 Supplement back to electron density 1 X σ Łukasz Rajchel (University of Warsaw) → 2 X . σ=− 21 DFT Warsaw, 2010 95 / 101 Supplement back to electron density 1 X → σ Z Z 3 ∞ 2 X Z ∞ Z ∞ f (r) d r = R3 . σ=− 21 f (r) dx dy dz = −∞ −∞ −∞ Z ∞ Z π Z 2π f (r)r2 sin θ dr dθ dϕ = = 0 0 0 = (or other coordinates) . . . Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 95 / 101 Supplement back to expectation values Brakets are shorthand notation for the integration over all electronic coordinates: hψ|Â|ψi = X Z = σ1 ...σN R3 Z ... ψ(r1 ; . . . ; qN )∗ Âψ(r1 ; . . . ; qN )d3 r1 . . . d3 rN . R3 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 96 / 101 Supplement back to Fock operator Functional F maps a function f to a number α: f 7→ F [f ] = α ∈ C. Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 97 / 101 Supplement back to Fock operator Functional F maps a function f to a number α: f 7→ F [f ] = α ∈ C. Examples of functionals: Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 97 / 101 Supplement back to Fock operator Functional F maps a function f to a number α: f 7→ F [f ] = α ∈ C. Examples of functionals: Z b F [f ] = |f (x)| dx — area under the curve f (x) for x ∈ ha; bi a Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 97 / 101 Supplement back to Fock operator Functional F maps a function f to a number α: f 7→ F [f ] = α ∈ C. Examples of functionals: Z b F [f ] = |f (x)| dx — area under the curve f (x) for x ∈ ha; bi a Z bp F [f ] = 1 + [f 0 (x)]2 dx — length of curve f (x) for x ∈ ha; bi. a Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 97 / 101 Supplement back to Fock operator Functional F maps a function f to a number α: f 7→ F [f ] = α ∈ C. Examples of functionals: Z b F [f ] = |f (x)| dx — area under the curve f (x) for x ∈ ha; bi a Z bp F [f ] = 1 + [f 0 (x)]2 dx — length of curve f (x) for x ∈ ha; bi. a hψ|Â|ψi = hAi — every physical observable is a functional of hψ|ψi the wavefunction. A[ψ] = Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 97 / 101 Supplement back to Fock operator Density function and the density in HF: 0 ρ(r; r ) = 2 N/2 X ϕi (r)ϕ∗i (r0 ), ρ(r) = ρ(r; r) = 2 i=1 Łukasz Rajchel (University of Warsaw) N/2 X |ϕi (r)|2 . i=1 DFT Warsaw, 2010 98 / 101 Supplement back to Fock operator Density function and the density in HF: 0 ρ(r; r ) = 2 N/2 X ϕi (r)ϕ∗i (r0 ), ρ(r) = ρ(r; r) = 2 i=1 1 Kinetic energy: T [ρ] = − 2 Łukasz Rajchel (University of Warsaw) N/2 X |ϕi (r)|2 . i=1 Z R3 ∆r ρ(r; r0 ) r0 =r d3 r. DFT Warsaw, 2010 98 / 101 Supplement back to Fock operator Density function and the density in HF: 0 ρ(r; r ) = 2 N/2 X ϕi (r)ϕ∗i (r0 ), ρ(r) = ρ(r; r) = 2 i=1 N/2 X |ϕi (r)|2 . i=1 Z 1 ∆r ρ(r; r0 ) r0 =r d3 r. Kinetic energy: T [ρ] = − 2 3 Z R vne (r)ρ(r) d3 r. n-e attraction: Vne [ρ] = R3 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 98 / 101 Supplement back to Fock operator Density function and the density in HF: 0 ρ(r; r ) = 2 N/2 X ϕi (r)ϕ∗i (r0 ), ρ(r) = ρ(r; r) = 2 i=1 N/2 X |ϕi (r)|2 . i=1 Z 1 ∆r ρ(r; r0 ) r0 =r d3 r. Kinetic energy: T [ρ] = − 2 3 Z R vne (r)ρ(r) d3 r. n-e attraction: Vne [ρ] = R3 Z Z 1 ρ(r1 )ρ(r2 ) 3 d r1 d3 r2 . Coulomb e-e repulsion: J[ρ] = 2 R3 R3 r12 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 98 / 101 Supplement back to Fock operator Density function and the density in HF: 0 ρ(r; r ) = 2 N/2 X ϕi (r)ϕ∗i (r0 ), ρ(r) = ρ(r; r) = 2 i=1 N/2 X |ϕi (r)|2 . i=1 Z 1 ∆r ρ(r; r0 ) r0 =r d3 r. Kinetic energy: T [ρ] = − 2 3 Z R vne (r)ρ(r) d3 r. n-e attraction: Vne [ρ] = R3 Z Z 1 ρ(r1 )ρ(r2 ) 3 d r1 d3 r2 . Coulomb e-e repulsion: J[ρ] = 2 R3 R3 r12 e-e exchange: Z Z 1 ρ(r1 ; r2 )ρ(r2 ; r1 ) 3 K[ρ] = −Ex [ρ] = d r1 d3 r2 . 4 R3 R3 r12 Łukasz Rajchel (University of Warsaw) DFT Warsaw, 2010 98 / 101 Bibliography I R. G. Parr and W. Yang, Density-Functional Theory of Atoms and Molecules (Oxford University Press, 1989). J. P. Perdew and S. Kurt, A Primer in Density Functional Theory (Springer Berlin / Heidelberg, 2003), vol. 620 of Lecture Notes in Physics, chap. Density Functionals for Non-relativistic Coulomb Systems in the New Century. A. D. Becke, Phys. Rev. A 38, 3098 (1988). J. P. Perdew, J. A. Chevary, S. H. Vosko, K. A. Jackson, M. R. Pederson, D. J. Singh, and C. Fiolhais, Phys. Rev. B 46, 6671 (1992). J. P. Perdew, K. Burke, and M. Ernzerhof, Phys. Rev. Lett. 77, 3865 (1996). A. Becke, J. Chem. Phys. 98, 5648 (1993). Bibliography II P. J. Stephens, F. J. Devlin, C. F. Chabalowski, and M. J. Frisch, J. Phys. Chem. 98, 11623 (1994). C. Adamo and V. Barone, J. Chem. Phys. 110, 6158 (1999). W. Koch and M. C. Holthausen, A Chemist’s Guide to Density Functional Theory (Wiley-VCH Verlag GmbH, Weinheim, 2001). A. J. Cohen and Y. Tantirungrotechai, Chem. Phys. Lett. 299, 465 (1999). J. Tao, J. P. Perdew, V. N. Staroverov, and G. E. Scuseria, Phys. Rev. Lett. 91, 146401 (2003). Thank you for your attention . . .