* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download cis- trans - KSU Web Home
Survey
Document related concepts
Transcript
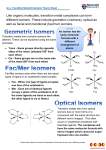
Chapter 12 Alkenes, Alkynes, and Aromatic Compounds 12.2 Cis–Trans Isomers General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 1 Cis and Trans Isomers In an alkene, cis and trans isomers are possible because the double bond is rigid cannot rotate has groups attached to the carbons of the double bond that are fixed relative to each other General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 2 Cis–Trans Isomers Cis–trans isomers can be modeled by making a “double bond” with your fingers with both thumbs on the same side (cis isomer) or fingers with thumbs on opposite sides from each other (trans isomer) General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 3 Cis–Trans Isomers (continued) Cis–trans isomers occur when different groups are attached to the double bond. In a cis isomer, groups are attached on the same side of the double bond. In the trans isomer, the groups are attached on opposite sides of the double bond. General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 4 Naming Cis–Trans Isomers The prefixes cis- or trans- are placed in front of the alkene name when there are cis–trans isomers. cistrans- Br Br C C H H Br C C H cis-1,2-Dibromoethene General, Organic, and Biological Chemistry H Br trans-1,2-Dibromoethene Copyright © 2010 Pearson Education, Inc. 5 Cis–Trans Isomerism Cis–trans isomers do not occur if a carbon atom in the double bond is attached to identical groups. (not cis or trans) General, Organic, and Biological Chemistry (not cis or trans) Copyright © 2010 Pearson Education, Inc. 6 Pheromones A pheromone is a chemical messenger emitted by insects in tiny quantities called bombykol is emitted by the silkworm moth to attract other moths and has one cis and one trans double bond General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 7 Learning Check Name each, using cis–trans prefixes when needed. Br Br C C A. H H H CH3 C C B. H CH3 Cl CH3 C. C C H Cl General, Organic, and Biological Chemistry Copyright © 2010 Pearson Education, Inc. 8 Solution Br Br cis-1,2-Dibromoethene C C A. H H H CH3 trans-2-Butene C C B. H CH3 Cl CH3 C. C C H Cl General, Organic, and Biological Chemistry 1,1-Dichloropropene Identical atoms; no cis or trans Copyright © 2010 Pearson Education, Inc. 9