* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chemical Quantities
Particle-size distribution wikipedia , lookup
Inductively coupled plasma mass spectrometry wikipedia , lookup
Isotopic labeling wikipedia , lookup
History of molecular theory wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Size-exclusion chromatography wikipedia , lookup
Mass spectrometry wikipedia , lookup
Stoichiometry wikipedia , lookup
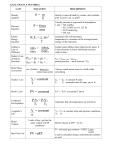
Chemical Quantities Unit 9D and 10 How were the molecules “figured out?” • Dalton used experiments to determine that water was 1 part mass of hydrogen gas to 8 parts mass of oxygen – Regardless what type of water he used, it was ALWAYS a 1:8 ratio (Volume was always 2:1) • “Law of Definite Proportions” – In samples of the same chemical compound, the ratio of masses of the elements will always be the same. Different compounds, different ratios • If the ratios are different, it MUST be a different compound – “Law of Multiple Proportions” • To determine if the ratios are the same: – Put the same elements on the same side of the ratios (hydrogen on top, oxygen on bottom) – Either: Simplify the two ratios and see if they’re the same, OR cross multiple and see if the products are the same. For example… • If you have a ratio of 10 players and 1 goalie, it is soccer. • If you have a ratio of 5 players and 1 goalie, it is NOT soccer. Let’s Try It • Sample A: – 13.18g of C – 8.25g of H • Sample B: – 39.40g of C – 33.15g of H • Are they the same ratios? • Sample A: – 13.18g of C – 8.25g of H • Sample B: – 39.40g of C – 33.15g of H • Are they the same ratios? – Sample A: 1.6 C:1 H – Sample B: 1.2 C:1 H – Or 437 = 325 Let’s Try It Try it on your own • Are these the same? • Sample A: – 29.35 g of Aluminum – 1.3g of Sulfur • Sample B: – 557.7g of Aluminum – 24.80g of Sulfur No, but YES. • They aren’t PERFECT ratios, but they are very close – Within the last significant digit, which is an estimate anyways Determining the formula by name • Are there pre-fixes before the name of an element? Are there no metals (or ammonium)? – Yes: Covalent. • Use prefixes for the subscript numbers and element names for the symbols – No: Ionic • Cations first: If group 1A-3A, the charge is the group number. If not, use the Roman Numerals • Anions second: If group 4A-7A, subtract 8-(group #) to determine charge. If it’s a polyatomic, it has its own charge. • BALANCE the charges at end. Just a reminder: • Only one element: (Ex: Hg or N2) – It’s a pure element: Name it after the element. • Two metals: (Ex: NaK) – It’s a metallic bond. Name it as an alloy. • One metal, one non-metal: (Ex: NaCl or BaOH2) – It’s an ionic bond. Name it with the two ions. • Two non-metals: (Ex: SO4, CO2) – It’s a covalent bond (molecule). Name it with prefixes. • If you’re not sure, use a flow chart. Try These: • Name these: – Na2O – S2O5 – Fe2O3 Try These: • Name these: – Na2O • Ionic: Sodium oxide – S2O5 • Covalent molecule: Disulfur pentaoxide – Fe2O3 • Ionic: Iron (III) oxide Determine the formulas • Tetrahydrogen monocarbide • Aluminum tetraborate • Ammonium Oxide Determine the formulas • Tetrahydrogen monocarbon – Covalent: H4C • Aluminum tetraborate – Ionic: Al3+2(B4O72-)3 • Ammonium Oxide – Ionic: (NH4+)2O2- How you can measure matter • Volume – The amount of space matter takes up • Mass – The amount of matter there is • Count – How many “things” there are. Measuring all 3 for paperclips. (And their intrinsic properties.) • • • • • • • Get 50 paperclips. Get a graduated cylinder Measure their mass Measure their volume Determine the mass per paperclip. Determine the mass per volume (density). Determine the volume per paperclip. The Mole • A mole is a special count used by chemists so they don’t have to use the HUGE counts of atoms and molecules in normal things – You are about 2,000,000,000,000,000,000,000,000,000 atoms or about 4,000 moles of water. – Like a dozen, but much bigger. • A mole can be a count of atoms, molecules, or formula units. – “Representative particles”. How much is a mole? • 602,000,000,000,000,000,000,000 particles. • 6.02x1023 particles – “Avogadro’s Number” • Why that number? That’s insane! – A mole of protons or neutrons has a mass of (almost exactly) 1g. – A mole of Carbon-12 has a mass of EXACTLY 12g. – One mole of ANYTHING equals its atomic mass in grams. • http://www.youtube.com/watch?v=g_BelGwR xG8 Okay, let’s try… • How many moles are in 3.00x1025 atoms of silicon? • How many molecules are in 51.0 moles of water? 1 mole 6.02x1023 particles How about these? • How many atoms are in 2.712 moles of neon? • How many moles are in 120,000,000 atoms? Two step molar conversions • You can convert moles into molecules, and then the molecules into atoms. • How many atoms of carbon are in a 2 moles of buckyballs (C60)? How about these two? • How many total atoms are in 425 moles of Na2SO4? • How many moles are in 3.52x1018 molecules of ammonia (NH3)? Why do all this madness?!? • MASS is the reason for the season mole thing. • The mass of a mole of any particle is equal to the atomic mass of the particle. – The mass of a mole is called its “molar mass” • The molar mass of atomic hydrogen is 1.0079g • The molar mass of atomic uranium is 238.03g • It doesn’t matter what element it is, a mole still always has 6.02x1023 particles. Allllllllllllwaaaaaaysssss! So, what is the mass of a mole of these? • Helium? • Bismuth? • Cesium? Okay, how about this? • What is the mass of 0.50 moles of atomic oxygen? • What is the mass of 5.00 moles of gold? The Molar Mass of a Compound • The molar mass of a compound (or molecule) is just the mass of the elements added together. – EX) Water has a molar mass of 18 because 15.9994+1.0079+1.0079= 18.0152 (O+H+H=H20) – EX) NaCl is 35.453+29.990= 65.443 Okay, try these. • What is the molar mass of propane (C3H8)? • What is the molar mass of nitrogen gas (N2)? Try these on your own. • What is the molar mass of ethanol (C2H6O)? • What is the molar mass of iron III oxide? • Challenge: What is the mass of 0.150 moles of ammonium tetraborate? Converting moles to mass • Use the molar mass of an element or compound to convert between the mass of a substance and the moles of the substance. – Mass = (moles) x (molar mass) • Or use the mass and the molar mass to determine the number of moles. – Moles = (mass) / (molar mass) Let’s Give it a Shot. • What is the mass of 0.0718 moles of CO2? • How many moles are in 37.8g of ozone (O3)? Try these on your own. • What is the mass of 1.48x10-9 moles of Fe3N2? • How many moles are in 1.758g of sodium bicarbonate (baking soda)? • https://www.youtube.com/watch?v=rWCvymhpfY A Mole of a Gas is only ONE volume! • A mole of any gas has a volume of 22.4 liters. • “Avogadro’s hypothesis” – It doesn’t matter what gas it is! – Must be at “standard temperature and pressure” (STP) • Basically at freezing and normal pressure at sea level. • Moles = volume (in liters) / 22.4 – Volume = Moles * 22.4 – At STP of course. Alright, now to try some of these. • What is the volume of 0.85 moles of N2 gas at STP? • How many moles of gas would fill 2265.35 liters? If we have mass and volume, we can get density! • Molar mass = density x 22.4 – Density is g/L • Density = Molar mass / 22.4 • Try this: – What is the density of CH4? • At STP, of course. – What is the molar mass of an unknown gas with a density of 0.80 g/L? One more problem? • What is the mass, density, and volume of 3.00 moles of chlorine gas at STP? • https://www.youtube.com/watch?v=rWCvymhpfY Percent Composition • Chemical compounds are “figured out” by using percent composition. – For instance, if you win 20% of a 10 game season, how many games did you win? How many did you lose? • To determine % composition for an element: – (mass of element)x100 mass of compound A percent mass composition problem! • No way! We totally weren’t expecting that!! • You have a sample from an alien planet. It is one where all the squirrels are 10 feet tall and pink. • The compound that makes the squirrels tall and pink is found to have 15.33g of carbon, 22.75g of hydrogen, and 115.91g of bismuth. – What is the total mass of the compound? – What is the % carbon, hydrogen and bismuth of the compounds? Wanna try one (or two) on your own? No? Well, too bad. • What is the percent carbon if the sample is 75.3g total, and 51g of that is carbon? • What is the mass of iodine in a sample if the total sample is 350g and it is 11% iodine? You can also use chemical formula to determine percent composition! • % mass = mass of element in compound x 100 molar mass of compound • For example: – H2O: H = 2(1.00794) = 2.01588g O = 15.9994g Total mass = 18.0153g % Hydrogen = 2.01588g/18.0153g = 11.1898% % Oxygen = 100 – 11.1898% = 88.8102% **Hooray, a shortcut! Try these. Or else! Or else what? Exactly. • What is the % composition of ethene (C2H4)? • What is the % composition of sodium phosphide? Solving Formulas • You can also determine the ratio of one element to the other – The “empirical formula” • The simplified (whole number) ratio of elements in a compound – But it is impossible to tell the difference of C2H4 and C4H8. – Or like “I have an equal number of children as cats.” But you can’t know how many of each. • http://www.youtube.com/watch?v=7OoDhM WDBDQ Determining Empirical Formulas • Start with the %, grams, or moles of a sample – If it is %, convert it to grams, then to moles – If it is grams, convert it to moles • Divide the moles of the larger numbers by the smallest number. • Turn it into a whole number ratio. Hmmm, maybe we should try some. • You have 12.44 moles of hydrogen and 6.22 moles of nitrogen. – What is the whole number ratio of H to N? – What would be the empirical formula? • You have a sample of 94.1% O and 5.9% H. What is the empirical formula? Determining Molecular Formulas • To find the actual molecular formula: 1. First determine the empirical formula. 2. Determine the molar mass of the empirical formula. 3. Then you need to know the molar mass of molecule. 4. Divide the molar mass of the molecule by the molar mass of the empirical formula. 5. Get this answer, and multiply it by the subscripts of the empirical formula to get the molecular formula. • Yes, this makes my brain hurt, too. But once you try a few it will make more sense. Let’s try one. • Calculate the molecular formula of a compound whose molar mass is 60.0g/mol and the empirical formula is CH3N. How about this one? • Determine the molecular formula for a compound that is 50.7% carbon, 4.2% hydrogen, and 45.1% oxygen. The molar mass of the molecule is 142g.