* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download 2s - Chemistry
2-Norbornyl cation wikipedia , lookup
X-ray fluorescence wikipedia , lookup
Coupled cluster wikipedia , lookup
Aromaticity wikipedia , lookup
Hartree–Fock method wikipedia , lookup
Metastable inner-shell molecular state wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Atomic theory wikipedia , lookup
Woodward–Hoffmann rules wikipedia , lookup
Atomic orbital wikipedia , lookup
Chemical bond wikipedia , lookup
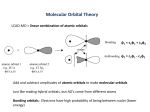
Chapter 10 Bonding and Molecular Structure: Orbital Hybridization and Molecular Orbitals Goals • Understand the differences between valence bond theory and molecular orbital theory. • Identify the hybridization of an atom in a molecule or ion. • Understand the differences between bonding and antibonding molecular orbitals. • Write the molecular orbital configuration for simple diatomic molecules. Orbitals and Bonding Theories VSEPR Theory only explains molecular shapes. It says nothing about bonding in molecules In Valence Bond (VB) Theory (Linus Pauling) atoms share electron pairs by allowing their atomic orbitals to overlap. Another approach to rationalize chemical bonding is the Molecular Orbital (MO) Theory (Robert Mulliken): molecular orbitals are spread out or “delocalized” over the molecule. Valence Bond (VB) Theory Covalent bonds are formed by the overlap of atomic orbitals. Atomic orbitals on the central atom can mix and exchange their character with other atoms in a molecule. Process is called hybridization. Hybrids are common: Pink flowers Mules Hybrid Orbitals have the same shapes as predicted by VSEPR. 1s 1s + H H bond 1s 1s + H H E 1s H bond + H H E H 1s H bond 2p 2p + F F bond F2 2p E 2s 1s F F 2p E 2s 1s F Methane CH4 2p E 2s 1s C Methane CH4 H H 2p E 2s 1s C Methane CH4 H H+ E 2p 2s 1s H C Methane CH4 H H+ E 2p 2s 1s H H– C Methane CH4 H Z Y H H– X H+ E 2p H 2s H 1s C C 90° H 90° H The approach is not correct, because… Methane CH4 H 109.5° C H H H Tetrahedral Geometry 4 Identical Bonds Problem and Solution C must have 4 identical orbitals in valence shell for bonding solution: hybridization (theoretical mixing of the four atomic orbitals of carbon atom, the 2s and the three 2p) Methane CH4 2p E 2s 1s Methane CH4 2s 2p E 2s 1s E 1s 2p Methane CH4 2s 2p E 2s 1s E 1s 2p Methane CH4 2s 2p E 2s 1s E 1s 2p Methane CH4 2p E 2s 1s four sp3 orbitals E 1s + – + + 2p 2s – + + + three 2p 2s = four sp3 hybrid orbitals 4 identical sp3 hybrid orbitals: they are four because there was the combination of one s and three p atomic orbitals (25% s, 75% p) tetrahedral geometry Methane CH4 H 2p E 2s 1s H H sp3 E 1s H Valence Bond (VB) Theory Regions of High Electron Density (BP+LP) Electronic Geometry Hybridization, Angles(°) 2 3 Linear Trigonal planar 4 Tetrahedral 5 Trigonal bipyramidal sp, 180 sp2 120 sp3 109.5 sp3d 120, 90, 180 6 Octahedral sp3d2 90, 180 Predict the Hybridization of the Central Atom in aluminum bromide Br Br Al Br Electron-pair shape 3 regions trigonal planar Hybridization: sp2 Trigonal Planar Electronic Geometry, sp2 Electronic Structures: BF3 B B F [He] 1s 1s 2s 2p 2s 2p 2p 2s 2p 1s 2 sp hybrid Trigonal Planar Electronic Geometry, sp2 BF3 Predict the Hybridization of the Central Atom in carbon dioxide CO2 O C O 2 regions Electron-pair shape, linear Hybridization: sp (50% s, 50% p) Linear Electronic Geometry, sp Electronic Structures: BeCl2 Be 1s 2s 2p Cl [Ne] 3s 3p 1s sp hybrid Predict the Hybridization of the Central Atom in Beryllium Chloride Two regions: electron-pair shape sp hybridization Predict the Hybridization of the Central Atom in PF5 Five regions: Trigonal Bipyramidal Electronic Geometry sp3d hybridization, five sp3d hybrid orbitals Predict the Hybridization of the Central Atom in xenon tetrafluoride Predict the Hybridization of the Central Atom in xenon tetrafluoride F F Xe F F 6 regions electron-pair shape octahedral Predict the Hybridization of the Central Atom in xenon tetrafluoride F F Xe F F 3 2 sp d 6 regions electron-pair shape octahedral hybridization Predict the Hybridization of the Central Atom in SF6 Six regions: Octahedral Electronic Geometry - sp3d2 hybridization, six sp3d2 hybrid orbitals Consider Ethylene, C2H4 Consider Ethylene, C2H4 H H C H C H Consider Ethylene, C2H4 H H C C H 3 regions trigonal planar H Consider Ethylene, C2H4 H H C C H H 3 regions trigonal planar 2 sp hybridization Consider Ethylene, C2H4 H H C H C H 3 regions trigonal planar sp2 hybridization 2p E 2s 1s 2s 2p E 2s 1s E 1s 2p 2p E 2s 1s sp2 E 1s 2p sp2 2p sp2 sp2 2p sp2 sp2 sp2 bond framework bond bond Compounds Containing Double Bonds Thus a C=C bond looks like this and is made of two parts, one and one bond. Consider Acetylene, C2H2 H C C H Consider Acetylene, C2H2 H C 2 regions linear C H Consider Acetylene, C2H2 H C C H 2 regions linear sp hybridization Consider Acetylene, C2H2 H C C H 2 regions linear sp hybridization 2s 2p E 2s 1s E 1s 2p 2p E 2s 1s sp E 1s 2p 2p sp 2p sp bond framework bonds Compounds Containing Triple Bonds A bond results from the head-on overlap of two sp hybrid orbitals. The unhybridized p orbitals form two bonds (side-on overlap of atomic orbitals.) Note that a triple bond consists of one and two bonds. bonds Generally • single bond is a bond • double bond consists of 1 and 1 bond • triple bond consists of 1 and 2 bonds Molecular Orbital (MO) Theory When atoms combine to form molecules, atomic orbitals overlap and are then combined to form molecular orbitals. # of orbitals are conserved. A molecular orbital is an orbital associated with more than 1 nucleus. Like any other orbital, an MO can hold 2 electrons. Consider 2 hydrogen atoms bonding to form H2 Molecular Orbital Theory • Combination of atomic orbitals on different atoms forms molecular orbitals (MO’s) so that electrons in MO’s belong to the molecule as a whole. • Waves that describe atomic orbitals have both positive and negative phases or amplitudes. • As MO’s are formed the phases can interact constructively or destructively. Molecular Orbitals There are two simple types of molecular orbitals that can be produced by the overlap of atomic orbitals. Head-on overlap of atomic orbitals produces (sigma) orbitals. Side-on overlap of atomic orbitals produces (pi) orbitals. Two 1s atomic orbitals that overlap produce two molecular orbitals designated as: 1s or bonding molecular orbital 1s* or antibonding molecular orbital. + H H subtract add subtract antibonding add bonding subtract antibonding *1s add bonding 1s Molecular Orbital Energy Level Diagram Now that we have seen what these MO’s look like and a little of their energetics, how are the orbitals filled with electrons? Order of filling of MO’s obeys same rules as for atomic orbitals. Including Aufbau principle: increasing energy Pauli’s Excluion: two unaligned e- per orbital, with opposite spins (+1/2 and -1/2) Hund’s Rule: maximum spin; unpaired electrons in degenerate orbitals have same spin (+1/2 or -1/2) Thus the following energy level diagram results for the homonuclear diatomic molecules H2 and He2. *1s E E 1s 1s 1s H H2 H *1s E E 1s 1s 1s H H2 H *1s E E 1s 1s 1s H H2 H *1s E E 1s 1s 1s H H2 H (1s ) 2 *1s E E 1s 1s 1s H H2 H (1s ) 2 total spin = 0 *1s E E 1s 1s 1s H H2 H • Diamagnetic: slightly repelled by a magnetic field – total spin = 0 • paramagnetic: attracted to a magnetic field – total spin not 0 (bonding e– – antibonding e–) • Bond Order = ──────────────────── 2 Bond Order and Bond Stability The larger the bond order, the more stable the molecule or ion is. Bond order = 0 implies there are equal numbers of electrons in bonding and antibonding orbitals, ~ same stability as separate atoms: no bond formed Bond order > 0 implies there are more electrons in bonding than antibonding orbitals. Molecule is more stable than separate atoms. The greater the bond order, the shorter the bond length and the greater the bond energy. (1s ) 2 total spin = 0 diamagnetic *1s E E 1s 1s 1s H H2 H BO = 1/2 ( 2 – 0) = 1 *1s E E 1s 1s 1s H H2 H Consider He2 *1s E E 1s 1s 1s He He2 He *1s E E 1s 1s 1s He He2 He (1s ) 2 ( *1s ) 2 *1s E E 1s 1s 1s He He2 He diamagnetic *1s E E 1s 1s 1s He He2 He BO = 1/2 ( 2 – 2 ) = 0 He2 does not exist *1s E E 1s 1s 1s He He2 He Combination of p Atomic Orbitals Molecular Orbitals The head-on overlap of two corresponding p atomic orbitals on different atoms, say 2px with 2px produces: 2px bonding orbital 2px* antibonding orbital 2p 2p subtract add antibonding MO subtract add bonding MO subtract antibonding MO *2p add bonding MO 2p Molecular Orbitals Side-on overlap of two corresponding p atomic orbitals on different atoms (say 2py with 2py or 2pz with 2pz) produces: π 2p π * y 2p y or π 2p (both are bonding orbitals) z or π*2pz (both are nonbonding orbitals) 2p 2p subtract add subtract antibonding MO add bonding MO subtract *2p add 2p subtract *2p add 2p Consider Li2 *2p *2p 2p E 2p 2p E 2p *2s 2s 2s Li 2s Li2 Li *2p *2p 2p E 2p 2p E 2p *2s 2s 2s Li 2s Li2 Li *2p *2p 2p E 2p 2p E 2p *2s 2s 2s Be 2s Be2 Be *2p *2p 2p E 2p 2p E 2p *2s 2s 2s Be 2s Be2 Be *2p *2p 2p E 2p 2p E 2p *2s 2s 2s B 2s B2 B *2p *2p 2p E 2p 2p E 2p *2s 2s 2s B 2s B2 B *2p *2p 2p E 2p 2p E 2p *2s 2s 2s C 2s C2 C *2p *2p 2p E 2p 2p E 2p *2s 2s 2s N 2s N2 N Homonuclear Diatomic Molecules In shorthand notation we represent the configuration of N2 as N2 2 1s *2 1s 2 2s *2 2s 2 2 py 2 2 pz 2 2p Bond Order of N2 N2 2 1s *2 1s 2 2s *2 2s 2 2 py 2 2 pz 2 2p The greater the bond order of a bond the more stable we predict it to be. For N2 the bond order is 10 - 4 bo 2 6 2 3 correspond ing to a triple bond in VB theory *2p *2p 2p E 2p 2p E 2p *2s 2s 2s O 2s O2 O Homonuclear Diatomic Molecules In shorthand notation we represent the configuration of O2 as O2 2 1s *2 1s 2 2s *2 2s 2 2 py 2 2 pz 2 2 px *1 2 py 10 - 6 bo = 2 2 We can see that O2 is a paramagnetic molecule (two unpaired electrons). *1 2 pz *2p *2p 2p E 2p 2p E 2p *2s 2s 2s F 2s F2 F *2p *2p 2p E 2p 2p E 2p *2s 2s 2s Ne 2s Ne2 Ne Bond Order for Ne2 24 - 24 BO = ─────── = 0 2 We can see that Ne2 is not stable. It does not exist. Delocalization and Shapes of Molecular Orbitals Molecular orbital theory describes shapes in terms of delocalization of electrons. Carbonate ion (CO32-) is a good example. VB Theory MO Theory Delocalization and Shapes of Molecular Orbitals Benzene, C6H6, Resonance structure - VB theory Delocalization and Shapes of Molecular Orbitals This is the picture of the valence bond (VB) theory Delocalization and Shapes of Molecular Orbitals The structure of benzene is described well by molecular orbital theory. Heteronuclear Diatomic Molecules • the more electronegative atom has lower energy orbitals • when the combining atomic orbitals are identical and equal energy, the weight of each atomic orbital in the molecular orbital are equal • when the combining atomic orbitals are different kinds and energies, the atomic orbital closest in energy to the molecular orbital contributes more to the molecular orbital – lower energy atomic orbitals contribute more to the bonding MO – higher energy atomic orbitals contribute more to the antibonding MO • nonbonding MOs remain localized on the atom donating its atomic orbitals NO Free-Radical 2s Bonding MO mainly O’s 2s atomic orbital HF Polyatomic Molecules • when many atoms are combined together, the atomic orbitals of all the atoms are combined to make a set of molecular orbitals which are delocalized over the entire molecule • gives results that better match real molecule properties than either Lewis or Valence Bond theories Ozone, O3 Delocalized bonding orbital of O3