* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Document
Survey
Document related concepts
Transcript
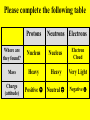
Democritus (400 B.C.) • World is made of 2 things empty space “atoms” Big Players in Atomic Theory • • • • • The Greeks (staring Democritus) John Dalton (1808) J.J. Thompson (1898) Rutherford (1909) Milliken (1909) More History Ernie’s (Rutherford) Big Adventure Thin Sheet of Gold Atoms Rutherford’s Conclusion • Atoms are made of mostly EMPTY SPACE! – Actually, the ratio of the size of the nucleus to the diameter of the orbits of electrons can be compared with placing a marble in the middle of a football stadium! LAVOISIER Found out that the mass of the products must equal the mass of the reactants (in a closed system) PROUST LAW OF DEFINITE PROPORTIONS Specific substances always contain elements in the same ratio BY MASS DALTON LAW OF MULTIPLE PROPORTIONS The ratio of masses of 1 element that combine with a constant mass of another element can be expressed in whole numbers Two major parts of an atom Nucleus (not to scale) Electron Cloud Three Major Sub-Atomic Particles • Protons • Neutrons • Electrons PROTON + (p ) a single, relatively large particle with a positive charge that is found in the nucleus THE PROTON • Fat (heavy) + p • Positive (charge) • Doesn’t move (lazy) NEUTRON (N°) a single, relatively large particle with a neutral charge that is found in the nucleus THE NEUTRON ° N • Fat (heavy) • Neutral (charge) • Doesn’t move (lazy) ELECTRON (e ) a single, very small particle with a negative charge that is found in a “cloud” around the nucleus THE ELECTRON • Skinny (very light) e • Negative (charge) • Moves a lot (runs around) Review: Subatomic Particles + p ° N e- Please complete the following table Protons Neutrons Electrons Where are they found? Nucleus Nucleus Electron Cloud Mass Heavy Heavy Very Light Charge (attitude) Positive Neutral Negative ATOMIC MASS # (A) The total mass of all of the subatomic particles in an atom (but really # of protons and neutrons) ATOMIC NUMBER (Z) the number of protons in an atom (assuming the atom is + neutral, # of p = # of e ) Example: Sodium Atomic Mass # = + p & 22.99 Na 11 Atomic # = # of protons ° N Another Notation Atomic Mass # = + p & Atomic # = # of protons ° N To calculate the number of neutrons, subtract the atomic number (smaller) from the atomic mass number (larger) A – Z = # of neutrons Ex: How many neutrons does Sodium have? Mass # - Atomic # = #N° (You may need to round the atomic #) 22.99 Na 11 23 - 11 = 12 N° ION Atoms of the same element that differ in charge. (They have the same # of + p , but different # of e ) Positive Ions Negative Ions (cations) (anions) • 2+ • Ca (lost 2 e ) 3+ • Al (lost 3 e ) 4+ • Pb (lost 4 e ) + • H (lost 1 e ) • 2• O (gain 2 e ) 3• P (gain 3 e ) 2• S (gain 2 e ) • OH (gain 1 e ) + Na (lost 1 e ) Cl (gain 1 e ) If an atom GAINS electrons, its overall charge becomes more negative. If it LOSES electrons, its charge becomes more positive ISOTOPE Atoms of the same element that differ in mass. (They have the same # of + p , but different # of N°) Isotopes are CHEMICALLY the SAME as atoms, but DIFFER PHYSICALLY because they have different masses. A few examples of isotopes… Complete the following table Protons Na+ Br w/ mass 84 O2- with mass 13 Neutrons Electrons So, why do the elements on the PT have masses with decimals??? AVERAGE ATOMIC MASS The average mass of all of the isotopes of a substance Example using exam scores! To calculate the average atomic mass: 1. Mass each isotope and multiply by # present 2. Add these products 3. Divide this sum by the total # present To calculate the average atomic mass: 1. Multiply the mass of each isotope by the % of the isotope 2. Add the products Carbon has two isotopes. 99% of carbon has a mass of 12 amu, and 1% has a mass of 13 amu. Calculate the average atomic mass. 12.01 amu 76% of chlorine has a mass of 35 amu. The other 24% has a mass of 37 amu. Calculate the average atomic mass of chlorine. 35.48 amu Sulfur has three main isotopes. 95% is Sulfur-32, 4% is Sulfur-34, and 1% is Sulfur- 33. Calculate the average atomic mass of sulfur. 32.09 amu