* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Marginal zone cells-the primitive streak
Survey
Document related concepts
Transcript
/. Embryol. exp. Morph. Vol. 52, pp. 79-88, 1979
Printed in Great Britain © Company of Biologists Limited 1979
79
Marginal zone cells-the primitive streak-inducing
component of the primary hypoblast in the chick
By YEHUDIT AZAR 1 AND HEFZIBAH EYAL-GILADI 1
From the Department of Zoology, The Hebrew University of Jerusalem
SUMMARY
(1) Removal of both the area opaca and the marginal zone of the area pellucida from a
blastoderm stripped of its hypoblast (type-lV operation) prevents the regeneration of a
normally functioning primary hypoblast.
(2) Stage-XIIl E.G & K blastoderms (prior to the appearance of PS) after a type-lV
operation do not form a primitive streak.
(3) In older type-lV operated blastoderms in which the primitive streak has already begun
to appear, the regeneration of a non-functional hypoblast did not support the normal maturation of the primitive streak, and in many cases the already existing rudimentary streak was
absorbed.
(4) Type-IV operated blastoderms from stage 3 + H & H and on developed normally.
(5) It is concluded that the cellular contribution of the marginal zone to the primary
hypoblast is instrumental in the latter's capacity to induce a PS.
INTRODUCTION
The chick blastoderm at about 10 h of incubation consists of two germ layers:
an upper layer, the epiblast, and a lower layer, the primary hypoblast. Somewhat later the primitive streak (PS) appears as the first axial tissue and cells
from the epiblast invaginate through it, either into the forming mesoblast, or
deeper down into the primary hypoblast, to form the definitive entoblast
(secondary hypoblast). The new entodermal cells occupy at stage 30 H & H
(Hamburger & Hamilton, 1951) a limited central position in the lower layer
(Vakaet, 1962, 1970), but as they increase in number, they spread centrifugally
and push the primary hypoblast to the periphery. The cells of the primary
hypoblast later form only extra-embryonic structures such as the yolk-sac
entoderm (Rosenquist, 1972; Wolk & Eyal-Giladi, 1977). Despite the fact that
its cells do not participate in the formation of embryonic organs (Vakaet, 1962;
Rosenquist, 1966; Modak, 1966; Nicolet, 1970), it was proposed (Eyal-Giladi
& Wolk, 1970; Eyal-Giladi, 1970) that the young primary hypoblast has a
major role in inducing the primitive streak and in stabilizing the embryonic axis.
1
Authors' address: Department of Zoology, The Hebrew University of Jerusalem,
Jerusalem, Israel.
82
Y. AZAR AND H. E Y A L - G I L A D I
(a)
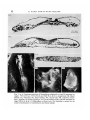
Fig. 1. (a-c) Transverse sections of blastoderms subjected to type-lV operation at
stages: 3~H & H (a); 2+H & H (b) and X1IL E.G & K (c) respectively. BI, Blood
islands; Ch, notocord; PL, lateral plates; NT, neural tube; PS, primitive streak;
Som., somites, (d) Control embryo, (e) Curved embryo after type-Ill operation at
stage XIII E.G & K. (/) Blastoderm without axis; the thickness is caused by the
massive development of mesenchyme and blood islands.
83
Marginal zone cells in chick
Table 1. Scheme of operations and results of experiments with stage-XIIIE.G & K
blastoderms
Stage Mil T y p e o f
h.Ci&K. Ar...i"itirm
blastoderm o p u d t I ° '
Steps of operation
Fragments discarded
Fragments
cultured
No. of
No. of
blasto- blastoderms
derms witli axial
development
15
12
18
15
19
37
IV
Area opaca
t(
^Marginal zone ffiffl Ilypoblast
f.:[) Hpiblast
----- Incision
Table 2. Results of type-IV operations carried out at four consecutive developmental stages
Group
(a)
(6)
+
(d)
(c)
+
Stage of operation
XIII E.G &K
2-2 H & H
3"-3 H & II
3 H & 11
No. of blastoderms
37
19
19
8
No. of blastoderms
with embryo
2
7
i.
8
No. of blastoderms
with P.S.
1
2
3
-
No. of blastoderms
in which P.S.
disappeared
-
10
5
-
Fragment cultured
delineated by
broken line
84
Y. AZAR AND H. EYAL-GILADI
tion was made between blastoderms which developed an embryo and others in
which only a primitive streak developed.
Subgroup (a) consisted of 37 stage-XIII E.G & K blastoderms, only two of
which developed an embryo and one a primitive streak. Thirty-five blastoderms
developed into relatively compact fragments with mesenchyme and blood
islands (Fig. lc,f).
Subgroup (b) consisted of 19 stage-2 to 2+ H & H blastoderms, of which seven
developed an embryo. Two blastoderms succeeded in completing the rudimentary PS present at the beginning of the experiment (Fig. \b), as compared
to ten of the blastoderms in which the existing small PS disappeared at the
end of incubation.
Subgroup (c) included 19 stage-3~ to 3 H & H blastoderms in 11 of which
embryonic development took place (Fig. la). Additional three completed the
formation of the PS and in five the PS present at the time of operation disappeared at the end of the experiment.
Subgroup (d) included eight stage-3+ H & H blastoderms with a 100%
embryonic development.
The results were statistically analysed for x2 distribution. The difference
between group a and each of the other groups was highly significant (0-003 or
less) while between b and c and c and d the significance was 0-05.
DISCUSSION
The aim of the present study was to follow the dynamics of the inductive
influence of the primary hypoblast on the epiblast by removing the entire
hypoblast from blastoderms of stages XIII E.G & K to 3 H & H, which covers
the period starting just prior to the formation of the PS until about two-thirds PS.
Waddington (1932, 1933) and Vakaet (1967) showed, by horizontally turning
the hypoblast relative to the epiblast in blastoderms with PS of various lengths,
that the orientation of the developing embryo was influenced by the polarity of
the rotated hypoblast. Waddington suspected that an induction might be
involved. However, he made no distinction between the primary and secondary
hypoblasts.
Eyal-Giladi & Wolk (1970), using pre-streak developmental stages, showed
by making trans-millipore filter combinations of epiblasts and primary hypoblasts that the primary hypoblast is indeed the inductor of the PS.
Two processes are involved in the formation of a primary hypoblast as well as
in the regeneration of a removed primary hypoblast. The process of polyinvagination, which proceeds simultaneously at many isolated spots, and an
anteriorly directed growth of marginal zone cells from Roller's sickle (Vakaet,
1962, 1967, 1970; Spratt & Haas, 1965; Eyal-Giladi & Kochav, 1976).
At stages older than stage XIII E.G & K the regeneration of a lower layer is
quite different, the cells being derived mostly from the PS, from which they
Marginal zone cells in chick
85
spread centrifugally (Vakaet, 1962, 1967, 1970; Modak, 1966; Nicolet, 1970,
1971; Rosenquist, 1972). At the same time the contribution to such a regenerating hypoblast by the processes of polyinvagination and posterio-anterior
growth from the marginal zone is gradually suppressed. Thus, the two different
modes of the regeneration of the lower layer in stage XIII E.G & K and at the
PS stages is obviously based on the normal development of the same stages
(Modak, 1966; Vakaet, 1967) where the cells of the definitive endoblast or the
secondary hypoblast which will form the intestinal tract of the chick invaginate
from the streak, invade the primary hypoblast and gradually push its cells to the
periphery of the lower layer.
With the above information in mind we tried to remove the inductive lower
layer at different developmental stages and also to interfere with its normal
regeneration which would inevitably result, after a certain lag, in the development of a normal embryonic axis (Modak, 1966; Wolk, 1968; and results of
type-I experimental group).
Of the two processes involved in the formation of the inductive-primary
hypoblast, the process of polyinvagination taking place at stages X-XIII E.G &
K seemed difficult if not impossible to tackle. However, the centripetal growth
from the marginal zone, predominantly expressed along the posterio-anterior
axis, seemed a suitable candidate for blocking.
Spratt & Haas (1960) prevented growth from the marginal zone mechanically,
by putting large clumps of carmine powder on the front of the anteriorly moving
cells. Eyal-Giladi & Wolk (1970) prevented the formation of normal primary
hypoblasts capable of PS induction both in unincubated stage-X E.G & K
blastoderms and in stage-XIII blastoderms stripped of their already existing
primary hypoblast, by applying a millipore filter to their lower surface. However,
from other experiments of the same series, it appeared that the filter might have
interfered mechanically with the normal differentiation of the mesodermal
tissues in experiments in which primitive streaks were induced. In the present
study we therefore decided to discard the marginal contribution to the primary
hypoblast by excising the entire marginal zone from the rest of the area pellucida,
which also required the removal of the area opaca external to it. Bellairs,
Bromham & Wylie (1967) studied the effect of the removal of the area opaca on
the differentiation of blastoderms of stage 4 H & H and concluded that after
24 h of incubation these embryos were poorly differentiated and smaller as
compared to their controls. We checked the development of blastoderms with
their area opaca removed either at stage XIII E.G & K or at stages 2-3 + H & H
grown for 48 h in culture, and found that the blastoderms were small, and the
embryos short and curved, probably due to the lack of the normal tension
exerted on the area pellucida by the centrifugal growth of the area opaca
(Bellairs, Boyde & Heaysman, 1969). However, more than 70 % of axial
developments in these blastoderms justified the application of the above experimental procedure.
86
Y. AZAR AND H. EYAL-GILADJ
The experimental results were compared along two different parameters. The
first approach was the comparison of the differentiation of stage-XHI E.G & K
blastoderms following different types of operation procedures (Table 1). The
group subjected to type-I procedure merely confirms that the removal of the
primary hypoblast without any further interference does not affect axial
development except for a 20 h delay, needed for the regeneration process to take
place. With both the hypoblast and the area opaca removed (type II) the percentage of axial development and the 20 h delay remained the same although
the blastoderms were much smaller in diameter. More revealing is the comparison of the two following pairs of experimental types.
(1) Operation types III and IV: Whereas the cultured central area of the epiblast with the hypoblast adhering to it (type 111) developed an axis, an identical
epiblastic fragment without an hypoblast (type IV) did not. This, like the transfilter experiments of Eyal-Giladi & Wolk (1970), indicates that the primary
hypoblast is indeed the inductor of the primitive streak.
(2) On comparing the results of operation types II and IV the importance of
the marginal zone in the regeneration of an inducing hypoblast becomes obvious. The only initial difference between the two is the presence of a marginal
zone in type II which during the regeneration process probably allows the
inclusion of the inductive cellular component into the regenerative hypoblast.
We therefore think that cells derived from the marginal zone participate in
the formation of the primary hypoblast by invading it mainly from the posterior
side and gradually moving anteriorly. Those marginal cells seem to be the only
component of the hypoblast capable of inducing a PS. The stock of the inductive marginal cells does not seem to be exhausted during the formation of the
primary hypoblast, so that after the latter's removal at stage XIII a new 'primary
hypoblast' with an inductive capacity usually regenerates. The lower layer of the
embryo-less blastoderms of type-lV experiments, as well as those of the transfilter experiments of Eyal-Giladi & Wolk (1970), although resembling a primary
hypoblast, was devoid of PS-inducing capacity. We assume that this layer was
formed by the cells which moved down from the epiblast by the process of polyinvagination. It is interesting that although no organized formation of a PS
could take place, dispersed mesodermal cells were found between the epiblast
and the defective hypoblast. They sometimes disclosed their identity by forming
blood islands. The temporal aspect of the inductive effect of the primary hypoblast was checked in type-IV experiments (Table 2). Here, a clear correlation
was shown between the stage at which the primary hypoblast was removed and
the ability of the denuded marginless epiblast to continue normal development.
At stage 3 + H & H which is about two-thirds of a PS the removal of the hypoblast no longer has any effect on the epiblast, the inductive process is completed
and development can continue undisturbed. It is interesting that after the
removal of the hypoblast in young PS stages (2-3 H & H), there was a definite
tendency for the already existing PS to disappear (Table 2, groups b and c).
Marginal zone cells in chick
87
This tendency was more pronounced the earlier the stage. The absorption
tendency supports the conclusion of Eyal-Giladi (1970) that the hypoblast
not only induces but also stabilizes the embryo-forming potencies of the
blastoderm.
This paper is part of a Ph.D. thesis by one of us (Y. A.).
REFERENCES
BELLAIRS, R., BROMHAM, D.
R. & WYLIE, C. C. (1967). The influence of the area opaca on the
development of the young chick embryo. /. Embryo/, exp. Morph. 17, 195-212.
BELLAIRS, R., BOYDE, A. & HEAYSMAN, J. E. M. (1969). The relationship between the edge
of the chick blastoderm and the vitelline membrane. Wilhelm Roux Arch. EntwMech. Org.
163, 113-121.
EYAL-GILADI, H. (1970). Differentiation potencies of the young chick blastoderm as revealed
by different manipulations. II. Localized damage and hypoblast removal experiments.
/. Embryol. exp. Morph. 23, 739-749.
EYAL-GILADI, H. & KOCHAV, S. (1976). From cleavage to primitive streak formation: A
complementary normal table and a new look at the first stages of the development of the
chick. I. General morphology. Devi Biol. 49, 321-337.
EYAL-GILADI, H. & WOLK, M. (1970). The inducing capacities of the primary hypoblast as
revealed by transfilter induction studies. Wilhelm Roux Arch. EntwMech. Org. 165, 226-241.
HAMBURGER, V. & HAMILTON, H. L. (1951). A series of normal stages in the development of
the chick embryo. /. Morph. 88, 49-92.
MODAK, S. P. (1966). Analyse experimental de l'origine de Pendoblaste embryonnaire chez
les oiseaux. Rev. suisse Zool. 73, 877-910.
NEW, D. A. T. (1955). A new technique for the cultivation of the chick embryo in vitro.
J. Embryol. exp. Morph. 3, 326-331.
NICOLET, G. (1970). Analyse autoradiographique de la localisation des differentes ebauches
presomptives dans la ligne primitive de l'embryon de Poulet. /. Embryol. exp. Morph. 23,
79-108.
NICOLET, G. (1971). Avian gastrulation. Advances in Morphogenesis 9, 231-262.
ROSENQUIST, G. C. (1966). A radioautographic study of labelled grafts in the chicks blastoderm. Contr. Embryol. Carneg. lnst. 38, 71-110.
ROSENQUIST, G. C. (1972). Endoderm movements in the chick embryo between the early
short streak and head process stages. /. exp. Zool. 180, 95-104.
SPRATT, N. T. (1947). A simple method for explanting and cultivating early chick embryos
in vitro. Science, N. Y. 106, 452.
SPRATT, N. T. & HAAS, H. (1960). Importance of morphogenetic movements in the lower
surface of the young chick blastoderm. /. exp. Zool. 144, 257-276.
SPRATT, N. T. & HAAS, H. (1965). Germ layer formation and the role of primitive streak in
the chick. I. Basic architecture and morphogenetic tissue movements. /. exp. Zool. 158,
9-38.
VAKAET, L. (1962). Some new data concerning the formation of the definitive Endoblast in the
chick embryo. /. Embryol. exp. Morph. 10, 38-57.
VAKAET, L. (1967). Contribution a l'etude de la pregastrulation et de la gastrulation
de l'embryon de poulet en culture 'in vitro'. Mem. Acad. Roy. Med. Belg. 2e ser. 5,
235-257.
VAKAET, L. (1970). Cinephotomicrographic investigations of gastrulation in the chick
blastoderm. Archs Biol., Liege 81, 387-426.
WADDINGTON, C. H. (1932). Experiments on the development of chick and duck embryos,.
cultivated in vitro. Phil. Trans. R. Soc. Lond. B. Ill, 179-230.
WADDINGTON, C. H. (1933). Induction by the endoderm in birds. Wilhelm Roux Arch.
EntwMech. Org. 128, 502-521.
88
Y. AZAR AND H. EYAL-GILADI
WOLK, M. (1968). The mutual interaction between epiblast and hypoblast in early stage of
chick development. M.Sc. thesis (Hebrew).
WOLK, M. & EYAL-GILADI, H. (1977). The dynamics of antigenic changes in the epiblast and
hypoblast of the chick during the processes of hypoblast, primitive streak and head
process formation, as revealed by immunofiuorescence. Devi Biol. 55, 33-45.
{Received 24 October 1978, revised 27 January 1979)