* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Ibuprofen
Survey
Document related concepts
Transcript
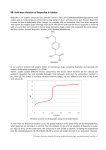
Public Assessment Report Scientific discussion Ibunin 200mg and Ibunin 400mg film-coated tablets (Ibuprofen) This module reflects the scientific discussion for the approval of Ibunin 200mg and Ibunin 400mg film-coated tablets. The procedure was finalised at day 210. For information on changes after this date please refer to the module ‘Update’. 1/6 I. INTRODUCTION This is an application for Ibuprofen 200mg and 400mg film-coated tablets (OTC), under the trade names Ibunin 200mg and Ibunin 400mg based on Article 10 (generics) of Directive 2001/83/EC as amended. The active substance, ibuprofen is not considered a new active substance. The original medicinal product which has been authorised for not less than 6/10 years in the EEA is Brufen 400 mg film-coated tablets by Abbott Laboratories A/S, registered in Denmark since 1972. Since Brufen is not authorised in Malta, the applicant has chosen Brufen (Abbott Scandinavia AB) as the European Reference Medicinal Product. With Malta as the Reference Member State in this Decentralised Procedure the applicant (Alchemia Limited, United Kingdom) is applying for Marketing Authorisations in the following Member States: AT, BE, BG, CZ, DE, DK, EE, FI, IE, IS, IT, LT, LV, NL, NO, PL, PT, RO, SE and SK. Ibunin 200mg and Ibunin 400mg was approved for the treatment of • mild to moderate pain, such as headache including migraine headache, dental pain; • primary dysmenorrhoea; and • fever II. QUALITY ASPECTS II.1 Introduction Pharmaceutical form: Film-coated tablets Formulation: Active ingredient: Ibuprofen Excipients: Tablet core Cellulose, microcrystalline Silica, colloidal anhydrous Hydroxypropylcellulose Sodium laurylsulfate Croscarmellose sodium Talc Film coating (Opadry (white) 06B28499) Hypromellose Macrogol 400 Titanium dioxide (E171) Container system: Opaque PVC/Aluminium blister packs. Clear PVC/Aluminium blister packs. 2/6 Tablet containers (polyethylene) with polypropylene caps. Pack sizes: Blisters: 6, 10, 12, 20, 24, 30 and 50 film-coated tablets. Tablet containers: 10, 20, 30 and 50 film-coated tablets. II.2 Drug Substance Pharmacological classification: Anti-inflammatory and anti-rheumatic products, non-steroids; propionic acid derivatives. ATC code: M01 AE01 Ibuprofen is a well known substance described in the European Pharmacopeia (Ph. Eur.) Version 6.1 Monograph 0721. It is practically insoluble in water. The manufacturer of the substance is Shasun Chemicals and Drugs Ltd, India who has been granted the certificate of suitability R1-CEP1996-061-Rev 03, therefore the route of synthesis is covered by this CEP. Ibuprofen quality can be suitably controlled according to its Ph. Eur. monograph complemented by a control on residual solvents and particle size distribution. The drug substance specifications adopted by the applicant, are in line with the current Ph. Eur. monograph 6.1 and with the additional tests given in the CEP. Issues raised to the applicant regarding particle size have been adequately addressed and the applicant has also adopted all additional parameters of the CEP as his own set of specifications as requested. All batch analytical data complied with the limits set out in the CEP and with the new Ph.Eur. monograph 6.1 for ibuprofen. Stability studies have been performed with the drug substance and no significant changes in any parameters were observed. II.3 Medicinal Product The development of the product has been described. Excipients common to pharmaceutical manufacture have been selected and each is described in the Ph.Eur. All excipient functions are explained. Comparative dissolution of the different strengths of the tablets manufactured by Alchemia Limited and reference products sourced from several EU countries have been provided and indicate that Ibuprofen Alchemia tablets are comparable to the corresponding Brufen® (Abbott Laboratories Limited, Kent, United Kingdom) tablets used in the Bioequivalence study and to the European reference product in the RMS. The product specifications cover appropriate parameters for this dosage form. Subdivision of tablets has also been included in the product specifications as requested since the purpose of the score line is for tablet division into equal halves as stated by the applicant. Tightened dissolution limits in line with the results obtained for the batches used in the Bioequivalence study have been adopted by the applicant. Validation results of the analytical methods have been presented. Batch analysis has been performed on two pilot scale size batches for each strength and one full scale production batch. The batch analysis results show that the finished products meet the specifications proposed. Stability data has been provided for two pilot scale batches and one production scale batch. The conditions applied in the stability studies are according to the ICH stability guideline. The control tests and specifications for drug product are adequately drawn up. The data submitted (for all tablet strengths) support the proposed shelf-life of 24 months for the product as stored under no special 3/6 conditions. III. NON-CLINICAL ASPECTS III.1 Introduction Pharmacodynamic, pharmacokinetic and toxicological properties of ibuprofen are well known. As ibuprofen is a widely used, well-known active substance, the applicant has not provided additional studies and further studies are not required. Overview based on literature review is, thus, appropriate. The non-clinical overview on the pre-clinical pharmacology, pharmacokinetics and toxicology is adequate. III.2 Environmental Risk Assessment No Environmental Risk Assessment Report has been submitted. This is acceptable since ibuprofen is a well-known active ingredient and ibuprofen drug products have been marketed worldwide for more than 30 years. IV. CLINICAL ASPECTS IV.1 Introduction Ibuprofen is a non-steroidal anti-inflammatory drug (NSAID) that possesses anti-inflammatory, analgesic and antipyretic activity. Animal models for pain and inflammation indicate that ibuprofen effectively inhibits the synthesis of prostaglandins. In humans, ibuprofen reduces pain possibly caused by inflammation or connected with it, swelling and fever. In addition ibuprofen has an inhibitory effect on adenosine diphosphate (ADP) or collagen-stimulated platelet aggregation. IV.2 Pharmacokinetics Racemic ibuprofen is absorbed from the gastrointestinal tract and peak plasma concentrations occur about 1 to 2 hours after ingestion. It is 90 – 99% bound to plasma proteins and has a plasma halflife of about 2 hours. It is rapidly excreted in the urine mainly as metabolites and their conjugates. About 1% is excreted in urine as unchanged ibuprofen and about 14% as conjugated ibuprofen. There appears to be little if any distribution in the breast milk. Ibuprofen’s disposition is stereoselective and there is some metabolic conversion of the inactive R (-) enantiomer to the active S (+) enantiomer. IV.3 Pharmacodynamics Ibuprofen inhibits the prostaglandin synthesis in the uterus, thereby reducing intrauterine rest and active pressure, the periodic uterine contractions and the amount of prostaglandins released into the circulation. These changes are assumed to explain the alleviation of menstrual pain. Ibuprofen inhibits renal prostaglandin synthesis which can lead to renal insufficiency, fluid retention and heart failure in risk patients. Prostaglandins are connected with ovulation and the use of medicinal products inhibiting prostaglandin synthesis may therefore affect the fertility of women. 4/6 IV.4 Clinical efficacy All issues concerning indications and posology which were raised by the RMS or the CMS(s) have been adequately resolved by the applicant. IV.5 Clinical safety All issues concerning clinical safety which were raised by the RMS or the CMS(s) have been adequately resolved by the applicant. IV.6 Discussion on the clinical aspects To support this application, the applicant has submitted one randomized, open label, two treatment, two period, two sequence, single dose, crossover, bioequivalence study of Ibuprofen 200 mg tablets of Actavis Group PTC ehf, Iceland and Brufen® (Ibuprofen) 200 mg tablets of Abbott Laboratories Limited, Kent, United Kingdom, in 30 healthy adult males, under fasting conditions, with a washout period of 10 days. The applicant requested a biowaver for the higher 400 mg strength. The references supplied by the applicant indicate that at higher doses, ibuprofen exhibits nonlinear kinetics due to nonlinear plasma protein binding with a resulting less than proportionate increase in AUC with the given dose. Under these circumstances the largest sensitivity to detect differences between two products would be with a single-strength study conducted on the lowest dose (or a dose in the linear range). Results of the study show that the 90% confidence intervals of the test/reference (T/R) ratio lie well within the prospectively defined 80 -125% acceptance for AUCt, AUC0-∞, and Cmax for both S (+) and R (-) enantiomers of ibuprofen. The RMS considers that the request for a Biowiaver for the 400mg strength is justified. There were no safety issues identified during the BE study. In conclusion bioequivalence between Ibuprofen 200 mg tablets of Actavis Group PTC ehf, Iceland and Brufen® (Ibuprofen) 200 mg tablets of Abbott Laboratories Limited, Kent, United Kingdom has been appropriately demonstrated in this study. V. OVERALL CONCLUSION, BENEFIT/RISK ASSESSMENT AND RECOMMENDATION Ibuprofen is a well established non-steroidal anti-inflammatory drug (NSAID) that possesses antiinflammatory, analgesic and anti-pyretic activity. Its clinical efficacy and safety are welldocumented. This application deals with a generic version of the original product Brufen, which has been registered in Denmark since 1972. Bioequivalence has been shown between Ibuprofen 200 mg tablets of Actavis Group PTC ehf, Iceland and Brufen® (Ibuprofen) 200 mg tablets of Abbott Laboratories Limited, Kent, United Kingdom in a well-designed and well-conducted Bioequivalence study. The RMS considers that the request for a BW for the 400mg strength is justified. 5/6 The final SPC is acceptable to the RMS. In conclusion the benefit/risk ratio for the product Ibunin 200 and Ibunin 400mg is positive. The application is approvable. The applicant committed to perform postauthorisation follow-up measures to be reported back to the RMS and CMSs as listed below. Follow-up measures: Area1 Description Quality The Applicant hereby commits to perform an in-use stability study for tablets packed in polyethylene containers, and to provide the results as soon as available. Quality Quality 1. Areas: Quality, Non-clinical, Clinical, Pharmacovigilance User consultation The package leaflet has been evaluated via a user consultation study in accordance with the requirements of Articles 59(3) and 61(1) of Directive 2001/83/EC. The language used for the purpose of user testing the Patient Leaflet was English. The results show that the package leaflet meets the criteria for readability as set out in the Guideline on the readability of the label and package leaflet of medicinal products for human use. 6/13 Public Assessment Report Update Ibufen 200mg and 400mg Ibuprofen Ibufen 200 mg film-coated tablet and Ibufen 400 mg film-coated tablet 7/13 This module reflects the procedural steps and scientific information after the finalisation of the initial procedure. 8/13 Scope Procedure number Product Information affected Renewal of the Marketing Authorization. MT/H/0100/ The product 001information 002/R/001 was updated during the renewal procedure. 9/13 Date of Date of end Approval/ start of the of procedure procedure non approval Assessment report attached 08/01/2014 Y, Annex I 14/07/2014 Approved Annex I – Renewal Marketing Authorization I Recommendation Based on the review of the data submitted for the renewal application, the RMS is of the opinion that the benefit/risk balance of Ibufen 200mg and 400mg, Ibuprofen, MT/H/0100/001-0002 is positive. The RMS therefore recommends the renewal of the Marketing Authorisation for Ibufen 200mg and Ibufen 400mg granted with unlimited validity. II Scope For this renewal, the MAH has submitted the following documents: - Cover Letter - Details on contact persons: • Qualified person in the EEA for Pharmacovigilance • Contact person in the EEA with overall responsibility for product defects and recalls • Contact person for scientific service in the EEA in charge of information about the medicinal product - Comprehensive table of content (not applicable for centrally authorised medicinal products) Renewal Application Form with the following annexes: A list of all authorised product presentations for which renewal is sought in tabular format - List of EU Member States / Norway / Iceland where the product is on the market and indicating for each country which presentations are marketed and the launch date Chronological list of all post-authorisation submissions since grant of the Marketing authorisation: a list of all approved or pending Type IA/IB variations, Art 61(3) Notifications, giving the procedure number, date of submission, date of approval/Refusal and brief description of the change. A statement, or when available, a certificate of GMP compliance, not more than three years old or reference to the EudraGMP database, for the manufacturer(s) of the medicinal product listed in the application issued by an EEA competent authority or MRA partner authority For manufacturing sites of the medicinal product not located in the EEA or in the territory of an MRA partner, a list of the most recent GMP inspections carried out by other authorities indicating the date, inspection team and outcome A declaration by the Qualified Person (QP) of each of the manufacturing authorisation holders (i.e. located in the EEA) listed in the application form where the active substance(s) is used as a - - - - 10/13 - starting material, that the active substance(s) is manufactured in accordance with the guidelines on good manufacturing practice for starting materials as adopted by the EU 1 SmPC, Labelling and Package Leaflet Information about the expert Quality (incl. Signature + CV) Clinical (incl. Signature + CV) Summary of Pharmacovigilance System Risk Management Plan Addendum to Quality Overall Summary Addendum to Clinical Overview III Summary of authorities comments and MAH’s response Issues raised by the RMS pertained to: • • • • Update to the product information, Clarification or documentation for the Pharmacovigilance inspections/outcomes for Dr. Max Pharma, A request for an updated QP declaration/GMP certificate An update to the quality expert statement. Comments were received by MS during Day 55, 85 and Day 89. The comments/clarifications from CMSs pertained to the product information, variation history list, CEP and application forms and an update to the quality expert report. All comments that were raised by RMS and CMSs were addressed and all issues resolved. The following commitments were made by the applicant: Commitments 1. The outcomes of the text harmonisation of the ibuprofen product information in DE/H/1482/01/DC and DE/H/2605/01-02/DC can be added on by further variations following conclusion of this renewal. 2.The next PSUR should be submitted after 3 years. See List of substances under PSUR Work Sharing scheme and other substances contained in Nationally Authorised Products with DLP synchronised . http://www.hma.eu/fileadmin/dateien/Human_Medicines/CMD_h_/Pharmacovigilance_Legislation/PS UR/CMDh_2014_03_a_List_of_substances.xls 11/13 Changes to the Product information during the renewal: • QRD updates: SmPC, PIL, labelling • Changes resulting from the paediatric WS (DE/W/040/pdWS/001): SmPC sections 4.2 & 4.4, top of PIL and sections 2 & 3, and labelling • Additional side effects as per originator: SmPC, PIL • IMB commitment (to exclude use of 400 mg in children under 12 years): 400 mg SmPC section 4.2, 400 mg PIL section 2 & 3 • Minor typographical updates: SmPC, PIL IV Conclusions The products are still in compliance with the current requirements regarding quality. No new clinical data have become available that changed the benefit risk assessment. Also no new safety issues were identified based on spontaneous reports, literature or published studies. Therefore the RMS is of the opinion that the renewal can be granted with unlimited validity. The MAH should continue to submit PSURs in accordance with the List of substances under PSUR Work Sharing scheme and other substances contained in Nationally Authorised Products with DLP synchronized. http://www.hma.eu/fileadmin/dateien/Human_Medicines/CMD_h_/Pharmacovigilance_Legislation/PS UR/CMDh_2014_03_a_List_of_substances.xls The DLP for ibuprofen PSUR is 18/02/2014 and submission 19/05/2014. The common renewal date is 22nd March 2014 The renewal procedure ended positively on the 14th July 2014. LIST OF ABBREVIATIONS USED IN ANNEXES 12/13 CV Curriculum Vitae CMS Concerned Member State DLP Data Lock Point EEA European Economic Area PSUR Periodic Safety Update Report MAH Marketing Authorisation Holder MS Member State QP Qualified Person RMS Reference Member State 13/13