* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download density lab pictures and explanation
Survey
Document related concepts
Transcript
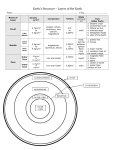
SMS 491/EDW742 – DENSITY LAB Spring 2007 Follow the instructions for each activity and ADDRESS ALL QUESTIONS IN YOUR JOURNAL Density (ρ) is defined as mass divided by volume ( ρ = m[ M ] ). The SI unit of density V [ L3 ] is kg/m3 and the cgs unit is g/cm3. Density is central to the understanding of many oceanic processes: from the formation of ocean basins, through ocean circulation to the transport of carbon from ocean surface to depth. As a property of matter, density is a central topic in secondary school physical science. While most adults have heard about the concept of density both school-age students and adults hold a number of misconceptions about density. Examples of misconceptions include (the list was compiled by the Operation Physics Elementary/middle school physics education outreach project of the American Institute of Physics): - Large objects sink and small object floats Objects float in water because they are lighter than water. Objects sink in water because they are heavier than water. Wood floats and metal sinks. All objects containing air float. Mass/volume/weight/heaviness/size/density may be perceived as equivalent. Note: while weight is commonly used as an equivalent to mass, in physics it is used to describe a particular force arising from the gravitational pull between objects (e.g. the Earth and the mass). Station 1: Will it float? Materials: One cube of balsa (from sciencekit.com) One cube of lignum vitae wood (from sciencekit.com) A large hollow metal ball (from sciencekit.com) Small Delrin® (or other plastic) ball (any hardware store) A container with water (use a big container for the large ball and a small one for the wooden cubes and the small ball). A ruler or caliper Scale (not shown) SMS491/EDW742 – Density Lab Last Updated: 01/22/2007 Page 1 © Karp-Boss, Weller and Boss, 2007 Materials for Station One -----------------------------------------------------------------------------------------------------------Experiment: You have two cubes of wood of the same size and two spheres (a large metal one and a small plastic one). 1. Predict which of the items will float in water and which one will sink and write your prediction in your journal. What was the reasoning behind your prediction? 2. Discuss your prediction with your group (and reflect upon it in your journal). 3. Test your predictions with the wooden cubes. Does the observation support your prediction? What are the similarities between the cubes? What are the differences between the cubes? 4. Now test your prediction with respect to the balls. Does the observation support your prediction? If not, how can you explain it. 5. Measure the densities of the cubes and the balls. Can that explain your observations? (volume of a sphere: V= 4/3πr3) SMS491/EDW742 – Density Lab Last Updated: 01/22/2007 Page 2 © Karp-Boss, Weller and Boss, 2007 Michelle attempts to measure density by hand. Will it float or sink? EXPLANATION: This activity can be used to address some of the misconceptions stated above (e.g., large object sinks, small object floats, wood floats, metal sinks). There is a wide range in the densities of wood found throughout the world. The range of density extends from balsa (0.1-0.17 g/cm3) to some tropical hardwoods that have density that exceeds that of water (1.04-1.37 g/cm3) Lignum vitae has a density of 1.171.29 g/cm3. The volume of the metal ball is 1023 cm3 and its mass is 144g. Its density is therefore 144/1023= 0.14 g/cm3. The volume of the Delrin ball is 1.15 cm3 cm and its mass is 1.5 g. Its density is therefore 1.5/1.15= 1.3 g/cm3. Given that the density of water is ~1 g/cm3 (depends on temperature, see reading material) the balsa and large metal ball will float and the Lignum vitae cube and Delrin ball will sink. SMS491/EDW742 – Density Lab Last Updated: 01/22/2007 Page 3 © Karp-Boss, Weller and Boss, 2007 Station 2: Density rods Materials: A set of density rods (from sciencekit.com) A ruler or caliper Calculator Graphing paper (or graphing program e.g., Excel) Materials for Station Two ----------------------------------------------------------------------------------------------------------Experiment: 1. You have a set of 12 black rods of different lengths but the same diameter. Do you think all the rods are made out of the same material? Feel free to use any sensory means to help you evaluate the rods. If you don’t think they are all made out of the same material, can you arrange them in groups according to their material? What were your category criteria for determining whether a rod belongs to one group or another? 2. Obtain the mass and the volume of the 12 rods and plot their volume (X) vs. mass (Y). Do all points fall on the same line? Do you see any pattern in the data? SMS491/EDW742 – Density Lab Last Updated: 01/22/2007 Page 4 © Karp-Boss, Weller and Boss, 2007 (Volume of a cylinder: V=π*r2*h, where r is the radius of the cylinder and h is the height of the cylinder) 3. What does the slope(s) of the line(s) represent? What does it tell you about the rods? Students use sensory means to help evaluate differences among the density rods EXPLANATION: Table 1 summarizes the mass, volume and density for each of the rods. SMS491/EDW742 – Density Lab Last Updated: 01/22/2007 Page 5 © Karp-Boss, Weller and Boss, 2007 3 Volume (cm ) Length of rod (cm) Table 1 Mass (g) 1.16 1.42 1.17 1.42 1.15 1.40 1.40 1.40 1.40 1.41 1.15 1.41 3.7 5.4 5.2 7.2 6.6 8.9 9.8 10.7 11.6 12.5 11 14.3 3.2 3.8 4.4 5.1 5.7 6.4 7.0 7.6 8.3 8.9 9.5 10.2 2.5 3 3.5 4 4.5 5 5.5 6 6.5 7 7.5 8 Density (g/cm3) 16 y = 1.3983x + 0.0551 R2 = 0.9999 14 12 Mass (g) 10 y = 1.1467x + 0.0679 R2 = 0.9999 8 6 4 2 0 0 2 4 6 8 10 12 Volume (cm^3) The data points are arranged along two linear lines, where the slope of each line provides the density (1.4 and 1.15 g/cm3. Compare the slopes with the range of calculated densities in the table. While the derived densities are not identical the small difference within each group is not significant given the uncertainties in the measurements). The data suggest that the rods are made of two different materials. (Caution: if two items have the same density it does not necessarily mean that they are made of the exact same material, however, density can be used as a method to distinguish between different materials). SMS491/EDW742 – Density Lab Last Updated: 01/22/2007 Page 6 © Karp-Boss, Weller and Boss, 2007 Station 3: Effects of temperature on density Materials: Two glass beakers: one with cold water (~6-15oC) and one with warm water ~40oC A set of reverse density rods (From Arbor Scientific): 1 Aluminum rod, 1 plastic rod Thermometer Ice (+ ice cooler to store ice) Hot plate (but hot tap water will work fine) Materials for Station Three -----------------------------------------------------------------------------------------------------------Experiment: 1. Which rod do you think will float/sink in the beaker with the cold water? What is the reasoning for your prediction? 2. Place the rods in the beaker with cold water. Make sure that no air bubbles are stuck to the rods. Record your initial observation in your journal. Does your observation meet your prediction? 3. Observe the rods for a few minutes. What is happening here? 4. Repeat this experiment, this time using the beaker with the hot water. SMS491/EDW742 – Density Lab Last Updated: 01/22/2007 Page 7 © Karp-Boss, Weller and Boss, 2007 5. How would you explain the different behaviors of the rods in cold and warm water? With your group discuss possible explanations for what you have observed and record them in your journal. EXPLANATION: In this activity, the rods are used to demonstrate the effect of temperature on density of liquids and solids and the activity can be used as a discrepant event. One rod is made out of aluminum and the other is made out of PVC. When you place the rods in cold water both rods initially float because their density is lower than that of the cold water. Over time, as the PVC rod gets colder it contracts and as a result its density changes (volume shrinks but its mass remains the same). When the density of the rod exceeds that of the water the PVC rod begins to sink. Aluminum has a much lower temperature expansion coefficient and hence its density is less affected by temperature and it remains floating. When you place the rods in hot water, the density of the water is now lower than that of the aluminum rod ands it will sink. The PVC rod is initially denser than the water and sinks but as it warms up it begins to expand. As a results its density changes (again, the mass remains constant) and when its density becomes lower than that of the water in begins to float. Station 4: Coca-Cola (Note: we used Coke but the success of the activity depends on the temperature of the water in the container. We later found out that Mountain Dew works better because the difference in mass between regular and diet mountain Dew is larger compared to that of Coke and diet Coke) Materials: Cans of Coke and diet Coke A fish tank or a large container with water. Paper towels SMS491/EDW742 – Density Lab Last Updated: 01/22/2007 Page 8 © Karp-Boss, Weller and Boss, 2007 Materials for Station Four -----------------------------------------------------------------------------------------------------------Experiment: 1. Examine the two cans. List as many similarities and differences between the 2 cans. 2. Now place the two cans in the tank. How would you explain this observation? 3. Wipe each can well and obtain the density of each cans (how can you determine it?). Is the density of coke smaller/greater than that of tap water? Is the density of diet coke smaller/greater than that of tap water? SMS491/EDW742 – Density Lab Last Updated: 01/22/2007 Page 9 © Karp-Boss, Weller and Boss, 2007 Morgan and Michelle discover the difference between adding sugar or NutraSweet to a beverage EXPLANATION: Density of Coke- 1.01 g/cm3 (volume=376 ml (1 ml= 1 cm3), mass=380.8 g.) Density of Diet Coke- 0.97 g/cm3 (volume=376 ml (1 ml= 1 cm3), mass=366.6 g.) The two cans have the same volume but they differ in their sugar content and hence in their mass. There are 39 grams of sugar in a 12 oz Coke can (see label) vs. approximately 100 mg of Nutra sweet in a 12 oz diet Coke can. SMS491/EDW742 – Density Lab Last Updated: 01/22/2007 Page 10 © Karp-Boss, Weller and Boss, 2007 Station 5: Density of rocks (This lab is based on an activity designed by Donald F. Collins, Warren-Wilson College http://www.warrenwilson.edu/~physics/EarthLightSky/Activities/Density/Density.html) Materials: Rock samples of Basalt (typical of oceanic crust) and Granite (typical of continental crust) A graduate cylinder or a graduated beaker A container with a spout Scale Materials for Station Five -----------------------------------------------------------------------------------------------------------Experiment: 1. Given what you know about density (ρ = M/V), obtain the densities of the rocks you have (to your aid, you have a graduate cylinder, a scale and a container with a spout). How do the granite and basalt compare to each other? How would you use it to explain the topography and bathymetry of the earth? 2. Based on the information you have, can you predict the average density of earth? SMS491/EDW742 – Density Lab Last Updated: 01/22/2007 Page 11 © Karp-Boss, Weller and Boss, 2007 3. Given that earth’s mass is 5.9742 × 1024 kilograms (a brain teaser: how this was determined?) and that earth’s radius is 6,378 km, calculate the average density of earth. How does the average earth’s density compare to the density of the rocks? What does it tells you about the structure of earth? Students examine the volume of water displaced by a sample of granite EXPLANATION: Density of basalt sample- 2.8 g/cm3 (volume=45 ml (1 ml= 1 cm3), mass=125.5 g.) Density of granite sample- 2.6 g/cm3 (volume=53 ml (1 ml= 1 cm3), mass=136.9 g.) The Earth crust is made of two types of crusts: continental and oceanic. Continental crust is composed mostly of granite while oceanic crust is mostly composed of basalt. Oceanic crust is thinner and denser than continental crust (textbook values: 2.9-3.0 vs. 2.7-2.8 g/cm3 , respectively; remember that you have measured the density of only one sample of each type of rock). Both overlie the earth’s mantle which is denser (3.3 g/cm3), with the continental crust “floating “ higher. Understanding the differences between oceanic and continental crust is crucial for understanding plate tectonics and the formation of ocean basins. We will discuss this in more details on February 14. SMS491/EDW742 – Density Lab Last Updated: 01/22/2007 Page 12 © Karp-Boss, Weller and Boss, 2007 On average, earth’s density is 5.5 g/cm3. That implies that the earth must be composed of additional layer/s of higher density then the rocks within its crust. In fact, the crust is the least dense layer. The estimated density of the core mantle is 5.5 g/cm3, the estimated density of the outer core is 10-12 g/cm3 and the estimated density of the core is 12-13 g/cm3. The density of the Earth can be computed using Newton’s laws: (1) Two bodies attract each other with a force (F) that is directly proportional to the product of their masses (m1,m2) and inversely proportional to the square of the distance between them (r) F=Gm1m2/r2, where G is the gravitational constant. G=6.7x10-11N m2 kg-2 (2) The force attracting a body to earth if its weight (not to confuse with mass) - mg, where g is the gravitational acceleration (g=980 cm/s2): F=mg Thus, Gme/r2 = g Æme =g*r2/G~6x1024Kg. Dividing by the volume of the Earth (4/3πR3) we obtain the Earth’s density (5461Kg/m3= 5.5 g/cm3) Station 6: Density of fluids Materials: A rectangular tank with a divider A salt solution or salt Food coloring Ice 2 beakers SMS491/EDW742 – Density Lab Last Updated: 01/22/2007 Page 13 © Karp-Boss, Weller and Boss, 2007 Materials for Station Six -----------------------------------------------------------------------------------------------------------Experiment: 1. In one beaker prepare a salt solution. Fill the other beaker with cold tap water and add ice so the water remains cold. 2. Place tap water in one compartment of the tank and salt water in the other. Add food coloring to one compartment. 3. What do you predict will happen when you remove the divider between the compartments? Explain in your journal. 4. Test your prediction. 5. Measure the densities of the tap water and the salt solution. Do the densities you obtained support your observations? 6. Now repeat the experiments with hot and cold water. SMS491/EDW742 – Density Lab Last Updated: 01/22/2007 Page 14 © Karp-Boss, Weller and Boss, 2007 What happens when I combine two fluids of differing densities? Amazing!! One floats on top of the other! Station 6: Density of fluids (EXPLANATION) Fluids arrange into layers according to their density. When the divider is removed the denser water (salt water or cold water) sink to the bottom of the container and the less dense water (fresh water or warm water) float above. See reading material. SMS491/EDW742 – Density Lab Last Updated: 01/22/2007 Page 15 © Karp-Boss, Weller and Boss, 2007