* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Dual atrioventricular nodal non-re
Management of acute coronary syndrome wikipedia , lookup
Cardiac contractility modulation wikipedia , lookup
Hypertrophic cardiomyopathy wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
Jatene procedure wikipedia , lookup
Ventricular fibrillation wikipedia , lookup
Electrocardiography wikipedia , lookup
Atrial fibrillation wikipedia , lookup
Arrhythmogenic right ventricular dysplasia wikipedia , lookup
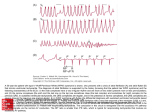
REVIEW Europace (2016) 18, 332–339 doi:10.1093/europace/euv056 Dual atrioventricular nodal non-re-entrant tachycardia Christiane Peiker1,2,3, Christian Pott 4, Lars Eckardt 4, Malte Kelm 3, Dong-In Shin 3, Stephan Willems 1,2, and Christian Meyer1,2,3* 1 Department of Cardiology – Electrophysiology, University Heart Centre, University Hospital Hamburg-Eppendorf, Martinistrasse 52, D-20246, Hamburg, Germany; 2DZHK (German Centre for Cardiovascular Research), Partner site Hamburg, Kiel, Lübeck, Germany; 3Department of Cardiology, Pulmonology and Vascular Medicine, Medical Faculty, University Hospital Duesseldorf, Germany; and 4Division of Electrophysiology, Department of Cardiovascular Medicine, University Hospital Muenster, Germany Received 23 December 2014; accepted after revision 19 February 2015; online publish-ahead-of-print 16 April 2015 Dual atrioventricular nodal non-re-entrant tachycardia (DAVNNT), also known as ‘double fire’, has recently received more attention since it was demonstrated to mimic more common arrhythmias such as atrial premature beats, atrial fibrillation, and ventricular tachycardia. This is important, since mistaken differential diagnoses and the resulting therapeutic decisions have severe consequences for affected patients. DAVNNT is characterized by conduction characteristics of the atrioventricular (AV) node that leads to a double antegrade conduction of one sinoatrial nodal activity via the slow and fast AV nodal pathways. As a result, the most significant hint from an electrocardiogram (ECG) is a P wave followed by two narrow QRS complexes. Although DAVNNT is rather a rare arrhythmia, it now appears to be more common than previously thought. To date, 68 cases including 3 small single-centre observational studies accumulated over the last 5 years have demonstrated the feasibility and safety of radiofrequency catheter ablation for DAVNNT. Catheter ablation treats this arrhythmia effectively by modifying or eliminating slow pathway function. Here, we review the current state of DAVNNT knowledge systematically and address current challenges presented by this ‘ECG chameleon from the AV node’. ----------------------------------------------------------------------------------------------------------------------------------------------------------Keywords Ablation † Atrial fibrillation † AVNRT † DAVNNT † Double fire † Slow pathway Introduction Understanding of the anatomical and physiological properties of the atrioventricular (AV) node as an integral part of the cardiac conduction system was important for the development of therapeutic concepts for bradycardia and tachycardia in the 20th century. It is now well known that the AV node area can be functionally dissociated; this may lead to pathways with different conduction characteristics, namely a fast and a slow pathway.1 This dual physiology is the prerequisite for the most common supraventricular tachycardia, AV nodal re-entrant tachycardia (AVNRT),2 where re-entry within these two AV nodal pathways is present.3 – 6 In contrast, the related but rare form of relatively benign tachycardia, dual AV nodal non-re-entrant tachycardia (DAVNNT), is characterized by a sinus beat that leads to consecutive double antegrade conduction via the fast and slow pathways, which results in two ventricular depolarizations.7 The phenomenon of dual fast and slow AV conduction of single atrial beats, also termed ‘AV nodal double firing’ or simply ‘double fire’, was first described in 1975.8 Since then the number of published case reports and small case series has increased in each decade (see references9 – 28 for those published in the last 5 years). It is now clear that this relatively rare arrhythmia, which is not addressed in the current guidelines of North American and European cardiology societies,29 may be more common than previously thought. We therefore review the current literature systematically and address the diagnostic and therapeutic challenges of this ‘electrocardiogram (ECG) chameleon from the AV node’. Methods The initial literature search was performed on 4 March 2014 and was repeated periodically until 1 June 2014. We searched PubMed/ MEDLINE (1 January 1995 until 1 June 2014), EMBASE (1950 until July 2014), and the Cochrane Central Register of Controlled Trials (CENTRAL—until June 2014) using the search terms ‘dual AV nodal nonreentrant tachycardia’ OR ‘DAVNNT’ OR ‘double fire’ OR ‘AV nodal double firing’ OR ‘non-reentrant AV nodal tachycardia’ OR ‘nonreentrant AV nodal tachycardia’. We excluded references not published in English unless an English version of the abstract was accessible. In an * Corresponding author. Tel: +49 40 7410 52438; fax: +49 40 7410 55862, E-mail address: [email protected], [email protected] Published on behalf of the European Society of Cardiology. All rights reserved. & The Author 2015. For permissions please email: [email protected]. 333 Dual atrioventricular nodal non-re-entrant tachycardia attempt to access the grey literature, we searched the OpenGrey Database (http://www.opengrey.eu) and the online trial registry (www. clinicaltrials.gov). We attempted to contact authors to clarify any areas of uncertainty regarding study data. Potentially eligible studies were assessed independently by two investigators (C.P. and C.M.). We included all published case reports, case series, and studies that were found with the specified search terms, as only 68 cases have been published so far. Because of well-documented weaknesses in the methodology, accuracy, and completeness of conference abstracts, we excluded studies published only in abstract form. We supplemented our database searches with manual searches of the reference lists of published studies and major review articles. Results Clinical presentation The currently reported age range of adult patients is broad, and varies from 16 to 84 years (68 cases) with similar proportions of males and females (Table 1).7,9 – 22,24 – 28,30 – 63 So far, only one case of a child (a 5-year-old boy) has been reported.23 Dual atrioventricular nodal non-re-entrant tachycardia has been observed in patients with and without structural heart disease, with no evidence of risk factors or accumulating comorbidities.27,28 So far no relationship is evident Table 1 Differential diagnosis of DAVNNT, reported as initial mistaken diagnosis in 48 of 68 cases Differential diagnosis Author Sinus tachycardia Arena, 1999 Bhatt, 2010 Wang, 2010 Kim, 1987 Nakao, 2001 Trajkov, 2006 Takahahi, 2012 Wang, 2013 Patients (n) Age (range) Sex: M/F (%) 3 16–46 66/33 5 5 –84 60/40 ............................................................................................................................................................................... Supraventricular premature beats Non-specific supraventricular tachycardia Elizari, 1991 Li, 1994 Anselme, 1996 Maury, 1999 Neumann, 2000 Labadet, 2001 Germano, 2005 Otsuka, 2005 Mofrad, 2007 Haman, 2009 Zimmermann, 2009 Barbato, 2010 Evertz, 2012 Mehairi, 2012 Pott, 2014 15 31–79 33/66 Atrial fibrillation Sutton, 1983 Fraticelli, 1999 Sorbera, 1999 Mansour, 2003 Rostock, 2005 Dixit, 2006 Clementy, 2007 Silver, 2008 Jastrzebski, 2009 Dingh, 2010 Ozcan, 2013 Kirmanoglou, 2014 16 27–74 50/50 Junctional ectopic beats Ventricular premature beats Aasbo, 2009 Sutton, 1983 Madle, 1990 Maury, 2008 Li, 2011 Neuss, 1982 Karnik, 2014 Kirmanoglou, 2014 1 4 56 33–69 100/0 50/50 4 20–80 100/0 Ventricular tachycardia Note that DAVNNT occurs equally in all ages and sexes. The most common misdiagnosis is atrial fibrillation. F, female; M, male; n, number of patients. 334 C. Peiker et al. common erroneous differential diagnosis.22,27,55 Other differential diagnoses that mimic a DAVNNT are rare but might result in inappropriate implantation of a pacemaker or ICD in certain cases.17,24 – 26,47 In an ECG, DAVNNT may be misinterpreted as ventricular tachycardia26,27,32 or ventricular premature beats.17,33,37,46,60 Functional left15,23 or right22,27 bundle branch block during DAVNNT has been reported in single cases and needs to be taken into consideration. In these cases, differential diagnosis of ventricular tachycardia can be guided by AV dissociation. The presence of unconventional AV association, which is characteristic of DAVNNT, needs to be evaluated (Figure 3). Differential diagnosis can be especially challenging if only standard monitoring documentation of an arrhythmia is available. In cases with the unclear presence/morphology of P waves, additional Holter electrode positions facilitate diagnosis. Apart from the risks of misdiagnosis, the average delay of more than a year for correct diagnosis7 may lead to tachycardia-induced cardiomyopathy, especially in relatively asymptomatic patients.15,17,24,42,52,57 A reduced left ventricular ejection fraction has been diagnosed in some patients who were ultimately diagnosed with DAVNNT and had a long history of palpitations. Interestingly, the reduced ejection fraction often returned to normal after successful treatment of DAVNNT, suggesting that left ventricular dysfunction was mainly induced by tachycardia.15,17,24,42,52,57 with other cardiac pathologies (including those related to the cardiac conduction system) apart from AVNRT. Patients diagnosed with DAVNNT initially presented with various symptoms. The most commonly reported symptoms are palpitations.7 Less common unspecific symptoms include dyspnoea, syncope, dizziness, fatigue, and chest pain.7 In two cases, ‘double fire’ resulted in inappropriate shocks from an implantable cardioverter-defibrillator (ICD),26,27 since current ICD discrimination algorithms are of limited value in differentiating DAVNNT from ventricular tachycardia. One patient did not report any symptoms, but a characteristic ECG was observed during routine monitoring of his pacemaker.25 Diagnosis The 12-lead surface ECG can be regarded as the ‘gold standard’ for detection of a suspected DAVNNT (Figure 1, left panel). The most significant hint from the ECG for diagnosis of DAVNNT is a P wave followed by two narrow QRS complexes. There are several differential diagnoses for the characteristic (but not yet widely known) DAVNNT ECG pattern (Figure 1, middle panel). Dual AV nodal conduction may be intermittent, thus mimicking atrial premature beats or atrial fibrillation. This might additionally make the diagnosis of DAVNNT difficult.24 Atypical presentation of supraventricular tachycardias such as atrial fibrillation (Figure 2A and B)11,12,22,27,30,33,46,51,55,57,61,64 or atrial tachycardia10,14,15,20,21,28,38,42,48,53,54,58,60 is common erroneous differential diagnoses (Table 1). Atypical sinus tachycardia,16,19,45 supraventricular premature beats (Figure 2C and D),23,24,36,50,56 atrial bigeminy,11,61 and atypical AVNRT with retrograde 2:1 block21 also need to be taken into account. Furthermore, sporadic junctional extrasystoles9,21,23,45,46 or parasystoles21 can mimic a ‘double fire’. Atrial fibrillation is the most Therapy Since 1994, all patients diagnosed with ‘double fire’ have been treated with radiofrequency catheter ablation. As a consequence of the relevant AV nodal physiology, the therapeutic approach used for DAVNNT is closely related to that used in most cases for the Diagnosis I Anatomy Physiology V1 AF II Mechanism Differential 12-lead ECG V1 V2 SPB Atrium III V3 SVT AVR AV Node V4 Ventricle VPB AVL Conduction via fast pathway V5 Conduction via slow pathway AVF VT V6 Block in slow pathway 50 mm/s Figure 1 Anatomical and physiological background of DAVNNT with ventricular ‘double fire’ following a single sinus node activity. Note the regular PR intervals, with one P wave followed by two QRS complexes due to consecutive antegrade conduction via fast and slow pathways from the AV node. In this example, due to early RS transition and a slurred upstroke resulting in a relatively broad QRS complex, finding the correct diagnosis might be challenging. For details see text. Black arrows denote P waves. AF, atrial fibrillation; ECG, electrocardiogram; SPB, supraventricular premature beats; SVT, supraventricular tachycardia; VPB, ventricular premature beats; VT, ventricular tachycardia. 335 Dual atrioventricular nodal non-re-entrant tachycardia A V1 50 mm/s B V1 HRA HBE RV 50 mm/s C V1 25 mm/s D avL V1 V3 V6 CS 1–2 CS 3–4 CS 5–6 CS 7–8 25 mm/s Figure 2 Electrocardiographic differential diagnosis of DAVNNT. Examples of DAVNNTs (A, B) and the relevant differential diagnosis of supraventricular premature beats (C, D) are presented. (A) Typical DAVNNT ECG pattern with biphasic P wave morphology in lead V1 consistent with sinus rhythm followed by two QRS complexes. Despite the clear P wave, atrial fibrillation was initially misdiagnosed, which resulted in an oral anticoagulation regime. Searching for P waves in lead V1 might have minimized this misdiagnosis. (B) Atypical P waves in combination with ‘irregular’ RR intervals often lead to the misdiagnosis of atrial fibrillation. Bundle branch block morphology may also delay correct diagnosis and here resulted in the initial misdiagnosis of ventricular tachycardia. Note the ‘unconventional’ AV association characterizing DAVNNT. Also note the subtle QRS changes, which might be explained by variable infra-His conduction.15 (C ) Frequent supraventricular premature beats with post extrasystolic pauses mimicking a DAVNNT. In this case, P waves occurring before each QRS complex within slight changes of T-wave morphology were detectable in lead V1. Note the shorter PQ interval for supraventricular premature beats compared with sinus rhythm. (D) Dual atrioventricular nodal non-re-entrant tachycardia-like morphology induced by frequent supraventricular premature beats exhibiting a typical ‘P on T’ pattern in a patient undergoing pulmonary vein isolation. Atrial activity before each QRS complex is confirmed by the multipolar catheter located in the coronary sinus (CS). Note that these supraventricular premature beats induce a QRS morphology change in lead V1. Black arrows denote P waves. HBE, his bundle electrogram; HRA, high right atrium; RV, right ventricle. treatment of AVNRT.65,66 Key differences are the approaches for the final diagnosis as well as the procedural endpoint. The most widely accepted procedural endpoint is non-inducibility of DAVNNT.7,46,51 For slow pathway modulation, radiofrequency energy is applied to the well-described right atrial posteroseptal area until conduction differences of the remaining AV node occur.3,67 Ablation of the slow pathway 336 C. Peiker et al. 50 mm/s AS 765 AS 765 VS 383 PVC 378 AS 765 VS 383 PVC 378 AS 765 VS 383 PVC 378 VS 383 PVC 378 Figure 3 Intracardiac recordings illustrating DAVNNT in a patient with a dual chamber ICD. Double ventricular activity was initially misdiagnosed as ventricular tachycardia. Dual atrioventricular nodal non-re-entrant tachycardia was revealed by unconventional AV association and later confirmed in an electrophysiological study. Importantly, isorhythmic dissociation needs to be ruled out. In this case, this was possible due to repetitive induction of an ongoing tachycardia by programmed atrial stimulation. The upper signals denote atrial near-field intracardiac electrograms (IEGMs), the middle signals are ventricular near-field IEGMs while the lower signals reflect far-field IEGMs. Coloured arrows denote antegrade conduction via fast and slow pathways. Marker channel: AS, atrial sensing; PVC, premature ventricular contraction; VS, ventricular sensing. Cycle lengths are annotated in milliseconds. eliminates dual conduction.68 Cryoablation is possible, but has only been reported in one patient.24 In this case, a second procedure using radiofrequency ablation was necessary due to recurrent DAVNNT. Recurrence of reduced symptoms has only been reported in three patients treated with modulation/ablation of the slow pathway.24,28 In one of these patients, DAVNNT was not inducible during an electrophysiological studyand slow pathway modulation was undertaken empirically, so that in retrospect it is not clear that the diagnosis of DAVNNT was appropriate in this case.28 Another patient was successfully treated by adjusting his ICD settings. The lower rate limit was increased from 40 to 70 b.p.m. to suppress slow pathway conduction, and the AV delay was increased to 300 ms to ensure intrinsic conduction.24 To date, the success rate of slow pathway modulation/ablation for DAVNNT treatment seems to be comparable but slightly lower than for AVNRT. This might be explained by the small number of reported cases. In our experience with .10 cases from three centres27,28 (n ¼ 8 published of 68 reported cases worldwide), no AV block or other complications were observed. Treatment with antiarrhythmic medications was only successful in the long term in one of the 68 patients, who was successfully treated with flecainide in 1987.36 Short-term successful treatment with medication has been described in two other patients. One of these did not give consent for slow pathway ablation and was treated with propafenone,25 which was successful at the 1-day follow-up but could not be evaluated later. The remaining patient was initially treated successfully with amiodarone,37 but medication had to be discontinued due to side effects. Electrophysiological characteristics of dual atrioventricular nodal non-re-entrant tachycardia Well-established dual AV node physiology is verified if programmed decremental atrial stimulation induces a ‘jump’, defined as a conduction delay of 50 ms.2,3,27,69 This ‘jump’ is not reported for all patients with DAVNNT. This might be explained in part by the difficulty of performing diagnostic stimulation manoeuvres during frequent ventricular ‘double firing’ where this ‘jump’ is already clinically manifest.19,22,31,41,46,50 Thus, dual AV nodal antegrade conduction in DAVNNT can also be characterized by discontinuous AV conduction, or it may be implied by a single atrial complex followed by two His and two related ventricular complexes (Figure 1, right panel).7,37 A DAVNNT is further defined by a fixed interval between His and ventricular activation,7,22,59 although slight variations in the R1R2 interval (interval between the first and second R wave) may occur due to fluctuations in AV nodal conduction velocity.70 In addition, slight changes in QRS morphology may be manifest (Figure 2B). Variability in slow and fast pathway conductions influences infra-His conduction as a form of gap phenomenon, leading to these changes in QRS morphology.17 Diagnosis is confirmed if modulation/ablation of the slow pathway terminates this ‘double firing’.7,46,51 The temporal delay between fast and slow pathway conductions is measured as the interval between His bundle activities induced by conduction in fast and slow pathways, both of which follow the same atrial complex. Conduction delay between the fast and the slow pathways needs to be relatively long and ranges from 265 to 520 ms with a mean + standard deviation of 359 + 46 ms for all reported cases.7,28,36,48 For a DAVNNT to occur, it is crucial that the effective refractory period of the His-Purkinje system is shorter than the interval between fast and slow pathway conductions.42,51,58,60 Retrograde conduction from the ventricle to the atrium is usually absent or weak.7,42,51,55,58,60 Moreover, His bundle ectopies followed by ventricular depolarization, which may also cause double ventricular responses, can be differentiated from DAVNNT by their irregular interval between both His complexes.7,28 Dual atrioventricular nodal non-re-entrant tachycardia 337 Dual atrioventricular nodal non-re-entrant tachycardia and junctional extrasystoles can be differentiated by administering atropine, which should suppress DAVNNT and intensify junctional activity.36 Discussion The major findings of this systematic review are: (i) DAVNNT seems to be more common than previously thought; (ii) since misdiagnosis can have severe consequences for patients, DAVNNT needs to be considered as a differential diagnosis for tachycardias involving suspected but atypical ECG patterns of atrial fibrillation and ventricular tachycardia; and (iii) ECG analysis allows instant visual diagnosis, which can be verified and successfully treated in an electrophysiological study with subsequent radiofrequency catheter ablation. The presence of slow and fast pathways within the AV node is the prerequisite for both the re-entry tachycardias such as the three commonly seen forms of slow–fast, fast –slow, and slow–slow AVNRT, as well as the non-re-entry tachycardia DAVNNT.27,69 Dual atrioventricular nodal non-re-entrant tachycardia can also initiate or convert into AVNRT.20 The true co-incidence of these arrhythmias is currently unknown. An international multicentre registry might be useful to determine the prevalence of DAVNNT. This might result in first recommendations regarding this rare arrhythmia in future guidelines of North American and European cardiology societies. Patient characteristics and clinical presentation are not helpful in discriminating between DAVNNT and more common arrhythmias. As outlined, DAVNNT may mimic clinical signs of AF or ventricular tachycardia, while ECG documentation is key for differential diagnosis. The general concept of diagnosing a DAVNNT based on the occurrence of a P wave followed by two narrow QRS complexes may be complicated by bundle branch block or intraventricular conduction disturbances (Figure 2B). It is worth noting that although the interval between the two QRS complexes is fixed in most episodes, marginal changes in the R1R2 interval may occur. Furthermore, the fibrosis that accompanies ageing and cardiovascular disease increases the extent and heterogeneity of structural discontinuity, which can affect QRS complex morphology.71 This might explain the minimal differences between conducted QRS complexes in surface ECG recordings that have been described in a significant number of patients (Figure 2B).17,63 Asynchronous activation spread via ventricular myocardium and heterogeneous repolarization might also affect QRS morphology in DAVNNT. The underlying mechanisms of AV nodal and consecutive ventricular activation are not fully understood. Early findings indicated that slow conduction involves impulse transmission to the compact AV node from regions inferior to the coronary sinus through transitional cells (posterior input), whereas fast regions involve direct input to the AV node from septal and left-sided inputs.63,65 In 2013, this concept was extended by the observation that two distinct patterns of pathway conduction may be present in patients with DAVNNT.64 Using novel mathematical techniques, the authors found that functional interaction between the slow and fast pathways may be present in some patients, whereas others might display largely independent activation of the ventricle. Unification of both individual pathways into one common pathway (equivalent to a lower common pathway in AVNRT) before activation of the His bundle and downstream structures has been suggested.64 Characterization of the 3D geometry in combination with computerized mapping models of the cardiac conduction system or analysis of its components at the cellular level might also improve our understanding of AV nodal function.71 This is supported by a model that incorporates atrial myocardium, inferior nodal extension, penetrating bundle, His bundle, and ventricular myocardium based on connexin 43 and neurofilament expression.72 This 3D reconstruction has been used to investigate the structural and functional bases of re-entry in the AV node.72 The related electrophysiological properties of myocardial and conduction system tissue are partly but not entirely influenced by autonomic control on a beat-to-beat basis.73 It is therefore not surprising that pharmacological beta-adrenergic and muscarinergic modulation has proved to be useful for diagnosis but not for therapy in patients with DAVNNT. Optical mapping techniques including optical coherence tomography (with a spatial resolution down to 1–2 mm) or microelectrodes addressing cells with different morphologic and molecular characteristics might be useful in analysing the neuro-myocardial interplay in AV nodal physiology in more detail. Slow pathway ablation for AVNRT is reported to have an immediate success rate of .95% with a risk of permanent AV block of ,1%.65 Whether these results can be extrapolated to DAVNNT is yet not known but indicated by published cases and our experience. Modulation or ablation of the slow pathway should be considered as a long-term remedy for this ‘ECG chameleon from the AV node’. Funding This work was supported by a research grant from the Forschungskommission of the University of Duesseldorf. C.M. is also supported by a grant from the DZHK (German Centre for Cardiovascular Research). Conflict of interest: none declared. References 1. Moe G, Preston J, Burlington H. Physiologic evidence for a dual A-V transmission system. Circ Res 1956;4:357 –75. 2. Denes P, Wu D, Dhingra R, Chuquimia R, Rosen K. Demonstration of dual A-V nodal pathways in patients with paroxysmal supraventricular tachycardia. Circulation 1973; 48:549–55. 3. Jackman W, Beckman K, McClelland J, Wang X, Friday K, Roman C et al. Treatment of supraventricular tachycardia due to atrioventricular nodal reentry, by radiofrequency catheter ablation of slow-pathway conduction. N Engl J Med 1992;327:313–8. 4. Link M. Clinical practice. Evaluation and initial treatment of supraventricular tachycardia. N Engl J Med 2012;367:1438 –48. 5. Katritsis DG, Sepahpour A, Marine JE, Katritsis GD, Tanawuttiwat T, Calkins H et al. Atypical atrioventricular nodal reentrant tachycardia: prevalence, electrophysiologic characteristics, and tachycardia circuit. Europace 2015;17:1099 –106. 6. Nakatani Y, Mizumaki K, Nishida K, Sakamoto T, Yamaguchi Y, Kataoka N et al. Electrophysiological and anatomical differences of the slow pathway between the fast-slow form and slow-slow form of atrioventricular nodal reentrant tachycardia. Europace 2014;16:551 –7. 7. Wang N. Dual atrioventricular nodal nonreentrant tachycardia: a systematic review. Pacing Clin Electrophysiol 2011;34:1671 – 81. 8. Wu D, Denes P, Dhingra R, Pietras R, Rosen K. New manifestations of dual A-V nodal pathways. Eur J Cardiol 1975;2:459 – 66. 9. Aasbo J, Prasertwitayakij N, Morady F, Jongnarangsin K. Nonreentrant dual atrioventricular nodal tachycardia in a patient with atrioventricular nodal conduction abnormality. Heart Rhythm 2009;6:1504 –6. 10. Haman L, Praus R, Parizek P. Non-re-entrant atrioventricular nodal tachycardia. Mil Med 2009;174:866 –8. 11. Jastrzebski M, Kukla P. Tachycardia caused by a double fire – simultaneous double atrioventricular nodal conduction: a rare or underdiagnosed arrhythmia? Spectrum of electrocardiographic pictures in three patients. Kardiol Pol 2009;67: 77 – 86. 338 12. Singh S, Barrett C, Das S. Pseudo-atrial fibrillation due to non-reentrant AV nodal tachycardia. Europace 2009;12:36. 13. Zhong-qun Z, Chong-quan W, Shu-yi D. Double ventricular response via dual atrioventricular nodal pathways resulting in sustained supraventricular nonreentrant tachycardia. J Electrocardiol 2009;42:232. 14. Zimmermann M, Testuz A, Schmutz M, Burri H. Narrow-complex tachycardia with cycle length alternans: What is the mechanism? Heart Rhythm 2009;6:1238 – 9. 15. Barbato G, Carinci V, Badhwar N. Tachycardia-induced cardiomyopathy. Card Electrophysiol Clin 2010;2:209 –12. 16. Bhatt AG, Monahan K. Nonreentrant supraventricular tachycardia misdiagnosed as inappropriate sinus tachycardia. Pacing Clin Electrophysiol 2011;34:e70. 17. Li V, Mallick A, Concannon C, Li V. Wide complex tachycardia causing congestive heart failure. Pacing Clin Electrophysiol 2011;34:1154 –7. 18. Burri H, Hoffmann J, Zimmermann M. Double fire tachycardia. Heart 2012;98:958. 19. Wang N, Razak E, Jain S, Saba S. Isoproterenol facilitation of slow pathway ablation in incessant dual atrioventricular nodal nonreentrant tachycardia. Pacing Clin Electrophysiol 2012;35:e31. 20. Al Mehairi M, Al Ghamdi S, Dagriri K, Al Fagih A. Simultaneous antegrade dual AV node conduction initiates AV nodal re-entrant tachycardia (a rare initiation mechanism). J Saudi Heart Assoc 2013;25:35 –7. 21. Evertz R, Merschón F, Berruezo A, Mont L. Dual ventricular response: another road to supraventricular tachycardia in dual atrioventricular nodal physiology. Rev Esp Cardiol 2013;66:145 – 6. 22. Ozcan E, Szeplaki G, Merkely B, Geller L. Non-reentrant atrioventricular nodal tachycardia. Clin Res Cardiol 2013;102:383–6. 23. Takahashi K, Nakayashiro M, Ganaha H. Dual atrioventricular nodal non-reentrant tachycardia in a child undergoing repair of ventricular septal defect. Pediatr Cardiol 2013;34:715 –8. 24. Wang N, Shah H, Jain S, Saba S. Dual atrioventricular nodal nonreentrant tachycardia with alternating 1:1 and 1:2 AV conduction: mechanistic hypotheses and total suppression using right atrial pacing. Ann Noninvasive Electrocardiol 2013;18:199 – 203. 25. Kaczmarek K, Ruta J, Wranicz J, Ptaszynski P. A new type of dual atrioventricular nodal nonreentrant tachycardia. Ann Noninvasive Electrocardiol 2014;19:501 –3. 26. Karnik A, Hematpour K, Bhatt A, Mazzini M. Dual AV nodal nonreentrant tachycardia resulting in inappropriate ICD therapy in a patient with cardiac sarcoidosis. Indian Pacing Electrophysiol J 2014;14:44– 8. 27. Kirmanoglou K, Peiker C, Clasen L, Shin D, Kelm M, Meyer C. Duale AV-nodale nicht-reentry-tachykardie. Herzschrittmacherther Elektrophysiol 2014;25:109–15. 28. Pott C, Wegner F, Bögeholz N, Frommeyer G, Dechering D, Zellerhoff S et al. A patient series of dual atrioventricular nodal nonreentrant tachycardia (DAVNNT) — an often overlooked diagnosis? Int J Cardiol 2014;172:e9. 29. Blomström-Lundqvist C, Scheinman M, Aliot E, Alpert J, Calkins H, Camm A et al. ACC/AHA/ESC guidelines for the management of patients with supraventricular arrhythmias--executive summary. a report of the American college of cardiology/ American heart association task force on practice guidelines and the European society of cardiology committee for practice guidelines (writing committee to develop guidelines for the management of patients with supraventricular arrhythmias) developed in collaboration with NASPE-Heart Rhythm Society. J Am Coll Cardiol 2003;42:1493 –531. 30. Rostock T, Sanders P, Rotter M, Haı̈ssaguerre M. Sinus rhythm mimicking atrial fibrillation recurrence. Heart Rhythm 2005;2:1158. 31. Csapo G. Paroxysmal nonreentrant tachycardias due to simultaneous conduction in dual atrioventricular nodal pathways. Am J Cardiol 1979;43:1033 –45. 32. Neuss H, Buss J, Schlepper M, Mitrović V. Double ventricular response in dual AV nodal pathways mimicking supraventricular as well as ventricular tachycardia. Eur Heart J 1982;3:146 –54. 33. Sutton F, Lee Y. Supraventricular nonreentrant tachycardia due to simultaneous conduction through dual atrioventricular nodal pathways. Am J Cardiol 1983;51: 897 –900. 34. Buss J, Kraatz J, Stegaru B, Neuss H, Heene D. Unusual mechanism of PR interval variation and nonreentrant supraventricular tachycardia as manifestation of simultaneous anterograde fast and slow conduction through dual atrioventricular nodal pathways. Pacing Clin Electrophysiol 1985;8:235 – 41. 35. Sutton F, Lee Y. Paroxysmal nonreentrant tachycardia due to simultaneous conduction via dual atrioventricular nodal pathways. Am Heart J 1985;109:157 –9. 36. Kim S, Lal R, Ruffy R. Paroxysmal nonreentrant supraventricular tachycardia due to simultaneous fast and slow pathway conduction in dual atrioventricular node pathways. J Am Coll Cardiol 1987;10:456–61. 37. Mádle A. A nonreentrant arrhythmia due to a dual atrioventricular nodal pathway. Int J Cardiol 1990;26:217 –9. 38. Elizari M, Sanchez R, Chiale P. Manifest fast and slow pathway conduction patterns and reentry in a patient with dual AV nodal physiology. J Cardiovasc Electrophysiol 1991;2:98 –102. C. Peiker et al. 39. Li H, Klein G, Natale A, Thakur R, Yee R. Nonreentrant supraventricular tachycardia due to simultaneous conduction over fast and slow AV node pathways: successful treatment with radiofrequency ablation. Pacing Clin Electrophysiol 1994;17:1186 –93. 40. Goutner C, Dechandol A, Bourguet J, Donzeau J. Tachycardie supraventriculaire due à une conduction simultanée dans la voie nodale rapide et lente. Traitement par ablation de la voie lente. Arch Mal Coeur Vaiss 1995;88:1651 – 5. 41. Ajiki K, Murakawa Y, Yamashita T, Oikawa N, Sezaki K, Kotsuka Y et al. Nonreentrant supraventricular tachycardia due to double ventricular response via dual atrioventricular nodal pathways. J Electrocardiol 1996;29:155–60. 42. Anselme F, Frederiks J, Boyle N, Papageorgiou P, Josephson M. An unusual cause of tachycardia-induced myopathy. Pacing Clin Electrophysiol 1996;19:115 –9. 43. Verdino R, Iuliano S, Tracy C. Successful ablation of a nonreentrant dual atrioventricular nodal tachycardia. J Interv Card Electrophysiol 1997;1:159 –61. 44. Glotz de Lima G, Roy D, Talajic M, Dubuc M. One-to-two atrioventricular conduction causing nonreentrant tachycardia: successful treatment with radiofrequency ablation. Pacing Clin Electrophysiol 1998;21:1152 – 4. 45. Arena G, Bongiorni M, Soldati E, Gherarducci G, Mariani M. Incessant nonreentrant atrioventricular nodal tachycardia due to multiple nodal pathways treated by radiofrequency ablation of the slow pathways. J Cardiovasc Electrophysiol 1999;10:1636 – 42. 46. Fraticelli A, Saccomanno G, Pappone C, Oreto G. Paroxysmal supraventricular tachycardia caused by 1:2 atrioventricular conduction in the presence of dual atrioventricular nodal pathways. J Electrocardiol 1999;32:347–54. 47. Sorbera C, Cohen M, Dhakam S, Fazio J. Symptomatic atrioventricular dual pathway double responses: a role for slow pathway ablation. Pacing Clin Electrophysiol 1999;22: 958 –61. 48. Neumann T, Schulte B, Pitschner H, Neuss H, Hamm C, Neuzner J. Doppelte Ventrikelerregung bei dualer AV-Knoten-Leitungsphysiologie: Katheterablation des langsamen Leitungsweges des dualen AV-Knotens. Z Kardiol 2000;89:1014 –8. 49. Labadet C, Di Villamil A, Tommaso F, Maurice MF, Prieto N. Double atrioventricular conduction during sinus rhythm. Rev Argent Cardiol 2001;69:281 –3. 50. Nakao K, Hayano M, Iliev I, Doi Y, Fukae S, Matsuo K et al. Double ventricular response via dual atrioventricular nodal pathways resulting with nonreentrant supraventricular tachycardia and successfully treated with radiofrequency catheter ablation. J Electrocardiol 2001;34:59–63. 51. Mansour M, Marrouche N, Ruskin J, Natale A, Keane D. Incessant nonreentrant tachycardia due to simultaneous conduction over dual atrioventricular nodal pathways mimicking atrial fibrillation in patients referred for pulmonary vein isolation. J Cardiovasc Electrophysiol 2003;14:752–5. 52. Gaba D, Pavri B, Greenspon A, Ho R. Dual antegrade response tachycardia induced cardiomyopathy. Pacing Clin Electrophysiol 2004;27:533 – 6. 53. Germano J, Essebag V, Papageorgiou P, Josephson M. Concealed and manifest 1:2 tachycardia and atrioventricular nodal reentrant tachycardia: manifestations of dual atrioventricular nodal physiology. Heart Rhythm 2005;2:536 –9. 54. Otsuka T, Igarashi M, Fujino N, Nakanishi R, Takamura K, Kobayashi K et al. Successful radiofrequency catheter ablation of nonreentrant supraventricular tachycardia due to simultaneous conduction through dual atrioventricular nodal pathways. Europace 2005;7:37. 55. Dixit S, Callans D, Gerstenfeld E, Marchlinski E. Reentrant and nonreentrant forms of atrio-ventricular nodal tachycardia mimicking atrial fibrillation. J Cardiovasc Electrophysiol 2006;17:312 –6. 56. Trajkov I, Kovacevic D, Boskov V, Poposka L, Gjorgov N. Induction of atrioventricular node reentry by simultaneous anterograde conduction over the fast and slow pathways. Prilozi 2006;27:113 –20. 57. Clementy N, Casset-Senon D, Giraudeau C, Cosnay P. Tachycardiomyopathy secondary to nonreentrant atrioventricular nodal tachycardia: recovery after slow pathway ablation. Pacing Clin Electrophysiol 2007;30:925 – 8. 58. Mofrad P, Hsia H. An unusual cause of incessant tachycardia. Pacing Clin Electrophysiol 2007;30:418 –20. 59. Laszlo R, Weig H, Weretka S, Schreieck J. Narrow complex tachycardia with alternating R-R intervals during physical stress: double ventricular excitation. Indian Pacing Electrophysiol J 2008;8:129 –32. 60. Maury P, Hebrard A, Duparc A, Fontan A, Adegnon K, Delay M. Incessant nonreentrant supraventricular tachycardia interrupted by apparent simultaneous wenckebach sequences along both nodal pathways. Pacing Clin Electrophysiol 2008; 31:757 – 60. 61. Silver J, Smith B, John R. Regularly irregular narrow complex tachycardia: what is the mechanism? Heart Rhythm 2008;5:487–8. 62. Katritsis D, Josephson M. Classification of electrophysiological types of atrioventricular nodal re-entrant tachycardia: a reappraisal. Europace 2013;15:1231 – 40. 63. Mendenhall GS, Voigt A, Saba S. Insights into atrioventricular nodal function from patients displaying dual conduction properties: interactive and orthogonal pathways. Circ Arrhythm Electrophysiol 2013;6:364 –70. 64. Meyer C, Pürerfellner H. Continuous heart rhythm monitoring to detect and quantify atrial fibrillation. Expert Rev Cardiovasc Ther 2010;8:889 –90. 339 Dual atrioventricular nodal non-re-entrant tachycardia 65. Nakagawa H, Jackman W. Catheter ablation of paroxysmal supraventricular tachycardia. Circulation 2007;116:2465 – 78. 66. Insulander P, Bastani H, Braunschweig F, Drca N, Gudmundsson K, Kennebäck G et al. Cryoablation of substrates adjacent to the atrioventricular node: acute and long-term safety of 1303 ablation procedures. Europace 2014;16:271–6. 67. Haissaguerre M, Gaita F, Fischer B, Commenges D, Montserrat P, d’Ivernois C et al. Elimination of atrioventricular nodal reentrant tachycardia using discrete slow potentials to guide application of radiofrequency energy. Circulation 1992;85: 2162– 75. 68. Posan E, Gula L, Skanes A, Krahn A, Yee R, Petrellis B et al. Characteristics of slow pathway conduction after successful AVNRT ablation. J Cardiovasc Electrophysiol 2006;17:847 –51. 69. Eickholt C, Boring Y, Kelm M, Shin D, Meyer C. Nonfluoroscopic catheter ablation of a double-loop re-entry tachycardia guided by real-time contact force information. Can J Cardiol 2013;29:254.e9– 254.e11. 70. Amasyali B, Kilic A. Dual atrioventricular nodal nonreentrant tachycardia: The rarest aspect of the structure causing the most frequent PSVT. Int J Cardiol 2014;173:550. 71. Smaill BH, Zhao J, Trew ML. Three-dimensional impulse propagation in myocardium: arrhythmogenic mechanisms at the tissue level. Circ Res 2013;112:834–48. 72. Li J, Greener ID, Inada S, Nikolski VP, Yamamoto M, Hancox JC et al. Computer three-dimensional reconstruction of the atrioventricular node. Circ Res 2008;102: 975 –85. 73. Appel ML, Berger RD, Saul JP, Smith JM, Cohen RJ. Beat to beat variability in cardiovascular variables: noise or music? J Am Coll Cardiol 1989;14:1139 –48. EP CASE EXPRESS doi:10.1093/europace/euv428 Online publish-ahead-of-print 6 February 2016 ............................................................................................................................................................................. Catheter ablation of multiple accessory pathways in Ebstein anomaly guided by intracardiac echocardiography Mihailo Vukmirović, Petr Peichl*, and Josef Kautzner Department of cardiology, Institute for Clinical and Experimental Medicine, Vı́den̆ská 1958/9, Prague 140 21, Czech Republic * Corresponding author. E-mail address: [email protected] A 24-year-old-female with Ebstein anomaly and preexcitation (ECG#1) was referred for catheter ablation. The procedure was guided by fluoroscopy, electroanatomical mapping system (CARTO 3TM , Biosense-Webster), and intracardiac echocardiography (ICE) (Acunav, Siemens). At first, ICE (upper Panel) was used to define the true location of tricuspid annulus (TA) by course of the right coronary artery (RCA). Both anatomical and functional insertions of the tricuspid valve were then annotated (lower Panel shows right atrium in left anterior oblique and caudal view). Subsequently, the earliest ventricular activity during sinus rhythm was mapped to the posterolateral portion of TA. After delivery of radiofrequency (RF) current at that site, a small change in pre-excitation pattern (ECG#2) was noted and the earliest ventricular activation shifted more septally. Further RF lesions (dark red points in lower Panel) were deployed along the posterior and subsequently towards posteroseptal part of the TA, leading to gradual elimination of conduction (ECG#3) via the wide/multiple accessory pathways (APs). It has been recognized that catheter ablation in Ebstein anomaly should optimally target anatomical and not functional TA. In this case, ICE enabled easy identification of the true TA by displaying the course of RCA and guided successfully ablation despite complex anatomy. Funding This work was supported by MH CZ - DRO (Institute for Clinical and Experimental Medicine – IKEM, IN 00023001) Conflict of interest: none declared. The full-length version of this report can be viewed at: http://www.escardio.org/Guidelines-&-Education/E-learning/Clinical-cases/ Electrophysiology/EP-Case-Reports. Published on behalf of the European Society of Cardiology. All rights reserved. & The Author 2016. For permissions please email: [email protected].