* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Slides from Lecture09
Survey
Document related concepts
Transcript
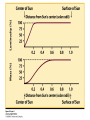
ASTR 1102-002 2008 Fall Semester Joel E. Tohline, Alumni Professor Office: 247 Nicholson Hall [Slides from Lecture09] Chapter 16: Our Star, the Sun A Problem with Time Scales! • Kelvin-Helmholtz contraction explains how the Sun’s interior could stay hot (even as it radiates copious amounts of heat from its surface) for hundreds of thousands of years. • But geological and fossil records show that the Earth is far older than this (the Earth-Moon system is about 4.6 billion years old!). A Problem with Time Scales! • Perhaps the Sun’s interior remains hot because the Sun is burning fuel in a manner similar to the way we humans burn fuel (for example, wood or coal) to generate heat/energy. – This type of “burning” involves building or breaking chemical bonds. • This won’t work because, in practice, relatively little energy is released through chemical burning processes. – To generate the Sun’s luminosity via chemical burning, the entire Sun would be consumed in about 10,000 years! Is there a Solution? • Is there some other source of energy that can be called upon to explain how the Sun’s structure can remain virtually unchanged for billions of years? • The answer is…the Sun “burns” its fuel (hydrogen) via nuclear reactions, rather than via chemical reactions. • Energy is released when the nuclei of hydrogen atoms are fused together to produce helium. Nuclear Reactions • Two basic types of nuclear reactions: – Fusion = the nuclei of two or more elements “fuse” together to create the nucleus of a heavier element – Fission = the nucleus of one element breaks apart to form nuclei of two or more lighter elements • Some reactions generate heat/energy (exothermic); other reactions absorb heat/energy (endothermic) • Rule of thumb: The dividing line falls within the Fe-Ni (iron-nickel) group of elements – Energy/heat generated via fusion when elements lighter than the Fe-Ni group are involved in the reaction – Energy/heat generated via fission when elements heavier than the Fe-Ni group are involved in the reaction Chemical Elements & Their Isotopes Courtesy of: http://atom.kaeri.re.kr/ Chemical Elements & Their Isotopes Hydrogen Chemical Elements & Their Isotopes Helium Chemical Elements & Their Isotopes Carbon Chart of Nuclides Chart of Nuclides 14 6 + 8 = 14 C How is Energy Generated? (and how much energy?) • Mass is converted into energy! • The total mass of the nuclei (or nucleus) produced by a reaction, Moutput, is less than the total mass of the nuclei (or nucleus) that start(s) the reaction, Minput. • E = (DM)c2 – Here, the quantity, DM = (Minput – Moutput) • Example: When 4 hydrogen nuclei fuse to form 1 helium nucleus, 0.7% of Minput is converted into energy/heat Is there a Solution? • Via nuclear fusion reactions (so-called, nuclear burning), the Sun can “live” for approximately 10 billion years and only use up approximately 10% of its total fuel supply! • We can express this mathematically: – tage = fMc2/L – For M = Msun, L = Lsun, and f = 0.7% x 10%, tage = 10 billion years Figure 16-4 Sun’s Internal Structure Chapter 19: Stellar Evolution: On & after the Main Sequence Apply the “Age” Concept to Other Stars • How long can other stars live? – tage = fMc2/L – (tage /1010 years) = (M/Msun)/(L/Lsun) Apply the “Age” Concept to Other Stars Checking Accuracy of Solar Model • Solar Oscillation measurements • Solar Neutrino measurements Solar Oscillation Measurements Solar Neutrino Measurements