* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download presentation ()
Survey
Document related concepts
Transcript
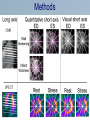
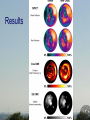
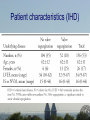
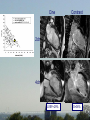
Dysfunctional but viable myocardium Ischemic heart disease assessed by MRI and SPECT Martin Ugander, MD Department of Clinical Sciences, Lund Department of Clinical Physiology Lund University Supervisor: Co-supervisor: Håkan Arheden, MD, PhD Clinical Physiology, Lund Peter Cain, MBBS, PhD Wesley Heart Clinic, Brisbane, AU Funding: • Swedish Research Council • Swedish Heart Lung Foundation • Faculty of Medicine at Lund University • Region of Scania Aim • To further elucidate the pathophysiology of dysfunctional but viable myocardium in patients with ischemic heart disease. Outline of Studies • Study I - Method for quantitative MRI & SPECT • Study II - Wall thickening vs. Infarct transmurality • Study III - LVEF vs. Infarct size • Study IV - Time course of perfusion & function after revascularization Study I Quantitative polar representation of left ventricular myocardial perfusion, function and viability using SPECT and cardiac magnetic resonance: initial results Cain PA, Ugander M, Palmer J, Carlsson M, Heiberg E, Arheden H. Clin Physiol Funct Imag 2005 (25) 215-222 Background • Clinical management of CAD involves complex assessment of the extent and severity of changes in function, perfusion and viability. • No adequate research tools for quantitative assessment exist. Aims • To explore the feasibility of integrative quantitative representation of LV perfusion, function and viability in polar plots. • To determine agreement between visual scoring and quantitative measures. Methods • 10 patients scheduled for CABG – rest/stress SPECT – Cine and delayed enhancment CMR • Quantification with in-house software • Comparison with visual scoring using Kendall’s coefficient of concordance (W) Methods Results Results Kendall’s W: 1.0 (p<0.001) 0.85 (p<0.001) Conclusions • Side-by-side quantitative polar representation of LV perfusion, function and viability is feasible and may aid in the complex assessment of these parameters. • The agreement between quantitative measurement and visual scoring was very good. Study II Infarct transmurality and adjacent segmental function as determinants of wall thickening in revascularized chronic ischemic heart disease Ugander M, Cain PA, Perron A, Hedström E, Arheden H. Clin Physiol Funct Imag 2005 (25) 209-214 Background • Regional LV function in patients with IHD may be influenced by many factors. Aims • To explore how regional wall thickening in patients with chronic IHD is affected by both infarct transmurality and the function of adjacent segments. • To compare with results from healthy subjects. Methods • 20 patients – 6 months after revascularization – Cine CMR – Delayed enhancement CMR • 20 matched controls – Cine CMR Multivariate analysis of parameters contributing to wall thickening t Infarct transmurality -4.5 Number dysf. adjacent seg. -22.9 p <0.001 <0.001 Conclusion • The number of dysfunctional adjacent segments is a greater determinant than infarct transmurality on regional wall thickening. • Infarction is difficult to assess by resting function alone. • DE CMR is an important tool in this setting. Study III A maximum predicted left ventricular ejection fraction in relation to infarct size in patients with ischemic heart disease Ugander M, Ekmehag B, Arheden H. Submitted Background • An understanding of the relationship between LVEF and infarct size is important when assessing the potential benefit of revascularization in patients with IHD. Aims • To explore the relationship between LVEF and IS. • To determine a maximum predicted LVEF for a given IS. Methods • 297 patients clinically referred for viability assessment by CMR • LVEF • Infarct size (% LVM) Methods LVEF (%) A C θ B Infarct size (%LVM) Patient characteristics (IHD) Distribution of infarctions Distribution of number of coronary artery vessel territories Results Cine Contrast 2ch 4ch LVEF=29% IS=36% Cine Contrast 2ch 4ch LVEF=25% IS=6% Conclusions • LVEF cannot be used to estimate IS. • IS cannot be used to estimate LVEF. • LVEF can be used to estimate a maximum predicted IS. • IS can be used to estimate a maximum predicted LVEF. Study IV Influence of the presence of chronic nontransmural myocardial infarction on the time course of perfusion and functional recovery after revascularization. Ugander M, Cain PA, Johnsson P, Palmer J, Arheden H. Manuscript Background • The time course of recovery of LV function and perfusion after revascularization is not fully understood. Aims • To study the effect of presence of infarction on the time course of recovery of perfusion and function after elective revascularization. Methods • 15 patients (inclusion ongoing) – first time elective CABG (n=13) or PCI (n=2) • Imaging – rest/stress SPECT – cine and delayed enhancement CMR – Before revasc., 1 & 6 months after revasc. Patient characteristics • • • • • • 14 men, 1 woman mean age 68 years (range 52-84) 3VD n=6 2VD n=6 1VD n=3 LVEF = 49 10% Distribution of infarct transmuralities Conclusions • Dysfunctional segments without infarction improved both perfusion and function at 1 month. • Segments with infarction showed improved perfusion at 1 month and improved function at 6 months. • This may reflect more severe ischemic burden in segments with infarction. Summary • Study I - Method for quantitative MRI & SPECT • Study II - Wall thickening vs Infarct transmurality • Study III - LVEF vs Infarct size • Study IV - Time course of perfusion & function after CABG Conclusion • It is important to perform quantitative assessment of function,perfusion and viability in combination when studying the pathophysiology of dysfunctional but viable myocardium in IHD. www.med.lu.se/cmr Bo Hedén, MD PhD Ann-Helen Arvidsson, tech Henrik Engblom, MD, PhD-student Håkan Arheden, MD PhD Christel Carlander, tech Karin Markenroth, PhD Marcus Carlsson, MD, PhD-student Martin Ugander, MD, PhD-student Erik Hedström, PhD Einar Heiberg ,PhD Henrik Mosén, MD, PhD Erik Bergvall, MSc, PhD-student The ischemic cascade Mahrholdt et al 2005 Eur Heart J




























































![Provider Bulletin: [Subject]](http://s1.studyres.com/store/data/000975616_1-3f817b14a0d66ce9d7f5c5c63cd4030c-150x150.png)
