* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Acid Base Balance (2)
Survey
Document related concepts
Transcript
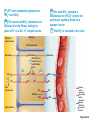
Acid Base Balance Mike Clark, M.D. • Acid - proton H+ donor • Base – proton H+ acceptor • Buffer – a chemical that resists a change in pH Acid-Base Balance • Normal pH of body fluids – Blood pH range 7.35 – 7.45 – Arterial blood is 7.4 – Venous blood and interstitial fluid is 7.35 – Intracellular fluid is 7.0 • Alkalosis or alkalemia – arterial blood pH rises above 7.45 • Acidosis or acidemia – arterial pH drops below 7.35 (physiological acidosis) pH Buffer A substance that resists a change in pH • Composition: A weak acid in equilibrium with its conjugate base Weak Acid Conjugate Base • [H3A] [H2A-] + [H+] • A weak acid does not completely dissociate -liberate its H+ whereas a strong acid completely or almost completely dissociates • Add outside acid to buffer it combines with the base H2A- to make more weak acid – add base it combines with the acid H+ to make more weak acid Chemical Buffer Systems • Three major chemical buffer systems 1. Bicarbonate buffer system – main extracellular buffer – Two non-bicarbonate buffer systems 2. Phosphate buffer system 3. Protein buffer system – most abundant – main intracellular buffer • Any drifts in pH are resisted by the entire chemical buffering system What Is the Problem with the wrong pH in the Human Body? • Improper pH denatures (bends out of shape) proteins. • When proteins bend too far out of shape they cease to function. • Functions of Proteins- Contractile, Regulatory, Enzymatic, Structural, Transport, Hormones • Most important function of all “Enzymes” • Why? They direct the pathway of all biochemical reactions. What are the mechanisms in the human body that regulate blood pH? • Concentration of hydrogen ions is regulated sequentially by: – Chemical buffer systems – act within seconds – The respiratory center in the brain stem – acts within 1-3 minutes – Renal mechanisms – require hours to days to effect pH changes Why is the regulation of blood pH so important? Don’t we have other fluids and tissues to protect also? • Since blood transports throughout the entire human body (except dead areas like the top of the skin) – it keeps the pH of the other body areas proper – if its pH is proper. pH Scale • Goes from 0 – 14 with 7 being neutral • Below seven is acidic • Greater than 7 is basic (alkaline) What is pH and how is it determined? • pH – stands for the powers of hydrogen • It is calculated using a mathematical formula pH = - Log [H+] • This is the universal formula used in all of chemistry to determine pH • However – the biochemical community uses another formula derived from the universal pH formula (Henderson-Hesselbach formula) Henderson-Hasselbach • pH = pKa + Log [Base] / [Acid] • The equation was derived from the universal pH equation. The equation uses the reaction H2CO3 HCO3- + H+ as its basis • Using this reaction the pKa is 6.1 • The Base is HCO3- The Acid is H2CO3 • • In an arterial blood gas – one does get the HCO3- (bicarbonate) value but not the H2CO3 (carbonic acid value). But the amount of Carbonic acid in the blood depends on Henry’s law – thus the partial pressure of the gas times the solubility coefficient. Thus .03 x PaCO2 is used. The arterial blood gas does give the value of PaCO2. • pH = pKa (6.1) + Log [HCO3- ] / .03 x [PaCO2 ] • The ideal arterial pH of the blood should be 7.4 • So if 7.4 = 6.1 + Log [HCO3- ] / .03 x [PaCO2 ] • The Log of Base of Acid needs to equal to 1.3 • The Log of 20 is 1.3 – thus the ratio of base to acid needs to be 20 (20 more times base than acid) [Total Acid] = [Volatile Acid] + [Fixed Acid] • The total [H+] (Acid) in the blood is measured when you calculate pH – it makes no difference where the H+ came from There are two acid types in the body • Fixed Acids and Volatile Acids • There is only one type of Volatile Acid – Carbonic acid – created from carbon dioxide mixing with water • All the other Acids in the body are termed “fixed acids” like lactic acid, hydrochloric acid and others • Homeostasis – if the fixed or volatile acid concentration goes up because of a problem the acid concentration without the problem should go down to compensate Normal Arterial Blood Gas Values • • • • pH – 7.35 – 7.45 PaO2 - 80 to 100 mm Hg. HCO3- - 22 to 26 mEq/liter PaCO2 - 35-45 mm Hg When Acid/Base Balance in the Blood Goes Wrong • Respiratory Acidosis – Lungs caused the acidosis • Metabolic Acidosis – there is blood acidosis, but the lungs did not cause – something else in the body caused it • Respiratory Alkalosis – Lungs caused the alkalosis • Metabolic Alkalosis - there is blood alkalosis, but the lungs did not cause – something else in the body caused it Respiratory Acidosis and Alkalosis • Result from failure of the respiratory system to balance pH • PCO2 is the single most important indicator of respiratory inadequacy • PCO2 levels – Normal PCO2 fluctuates between 35 and 45 mm Hg – Values above 45 mm Hg signal respiratory acidosis – Values below 35 mm Hg indicate respiratory alkalosis pH = 6.1 + Log [HCO3]/PaCO2 x .03 Must keep a ratio of 20 to 1 Base to Acid for pH to be 7.4. • Respiratory Acidosis If PaCO2 goes up then the ratio drops and the blood becomes acidic – unless the kidney holds on to more bicarbonate to compensate • Respiratory Alkalosis If PaCO2 goes down then the ratio increases and the blood becomes basic – unless the kidney removes (urinates out) more bicarbonate to compensate pH = 6.1 + Log [HCO3]/PaCO2 x .03 Must keep a ratio of 20 to 1 Base to Acid for pH to be 7.4. • Metabolic Acidosis If PaCO2 is normal or low and the blood is acidotic then the lungs are not the problem since they are not causing more carbonic acid to be made – thus the acidosis is due to something else in the body “metabolic” - the lungs maybe blowing off more CO2 than usual to help – thus compensate. Examples Lactic Acidosis or Diabetic Ketoacidosis • Metabolic Alkalosis If PaCO2 is normal or elevated and the blood is alkalotic then the lungs are not the problem since they are not causing less carbonic acid to be made – thus the alkalosis is due to something else in the body “metabolic” - the lungs maybe holding on to more CO2 than usual to help – thus compensate. Example Milk alkali sydrome Compensatory Actions • Complete compensation – though a metabolic or respiratory problem – the compensatory mechanism is so good it completely compensates – thus pH stays completely normal (this very, very rarely occurs – for the most part never) • Partial compensation- though a metabolic or respiratory problem – the compensatory mechanism tries to keep the pH normal – and does to some extent. • Respiratory Acidosis (completely or partially) compensated by a metabolic alkalosis • Metabolic Acidosis (completely or partially) compensated by a respiratory alkalosis • This also occurs for respiratory or metabolic alkalosis venport Curves pH Problems • Arrhythmias can result when the pH falls below 7.25, and seizures and vascular collapse can occur when pH rises above 7.55. Reabsorption of Bicarbonate • Carbonic acid formed in filtrate dissociates to release carbon dioxide and water • Carbon dioxide then diffuses into tubule cells, where it acts to trigger further hydrogen ion secretion PLAY InterActive Physiology ®: Acid/Base Homeostasis, page 34 Figure 26.12 Figure 26.13 New HCO3– is generated via buffering of secreted H+ by HPO42– (monohydrogen phosphate). Slide 1 3b For each H+ secreted, a HCO3– enters the 1 CO2 combines with water within the peritubular capillary blood via an antiport carrier in a HCO3–-CI– exchange process. type A intercalated cell, forming H2CO3. 2 H2CO3 is quickly split, forming H+ and bicarbonate ion (HCO3–). 4 Secreted H+ combines with HPO42– in H+ ATPase pump. 5 The H2PO4– is excreted in the urine. the tubular filtrate, forming H2PO4–. 3a H+ is secreted into the filtrate by a Nucleus Filtrate in tubule lumen Peritubular capillary H2O + CO2 1 H2CO3 HPO42– 2 3a H+ + HCO3– H+ 4 H2PO4– 3b HCO3– (new) ATPase Cl– Type A intercalated cell of collecting duct 5 out in urine Copyright © 2010 Pearson Education, Inc. Cl– Cl– Primary active transport Secondary active transport Simple diffusion Facilitated diffusion Transport protein Ion channel Carbonic anhydrase 1 PCT cells metabolize glutamine to NH4+ and HCO3–. 2a This weak acid NH4+ (ammonium) is secreted into the filtrate, taking the place of H+ on a Na+- H+ antiport carrier. 2b For each NH4+ secreted, a bicarbonate ion (HCO3–) enters the peritubular capillary blood via a symport carrier. 3 The NH + is excreted in the urine. 4 Nucleus Filtrate in tubule lumen Peritubular capillary PCT tubule cells Glutamine Glutamine Deamination, 1 oxidation, and acidification (+H+) 2a NH4+ 3 Na+ Glutamine 2b 2NH4+ 2HCO3– HCO3– Na+ Na+ Na+ NH4+ out in urine 2K+ HCO3– (new) 2K+ ATPase 3Na+ Tight junction 3Na+ Na+ Primary active transport Secondary active transport Simple diffusion Transport protein Figure 26.14