* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download 5 6. moles and rxn - New Hartford Central Schools
Survey
Document related concepts
Rigid rotor wikipedia , lookup
Rate equation wikipedia , lookup
Magnetorotational instability wikipedia , lookup
Debye–Hückel equation wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Gas chromatography–mass spectrometry wikipedia , lookup
Transcript
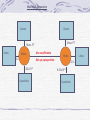
Mole Moles How a chemist measures the amount of matter in a substance counting unit - number of molecules ; atoms ; particles MOLE TRIANGLE Way to convert Road Map – map out where you start to where you end Conversions are along side the arrows How? Divide in () Multiply out (x) 22.4 L Volume Grams Formula wt. or gram-form. mass MOLES 6.02 x 1023 # molecules or # particles Moles How many moles of oxygen in 1 mole of Al2(SO4)3? 3 x 4 = 12 (the parantheses are Like a distribution in math) How many moles of oxygen in 1 mole of CuSO4 5H2O? 4 + (5 x 1) = 9 Gram atomic/formula mass (molecular mass) = mole of substance Al2(SO4)3 Al 2 2 x 27 = 54 S 3 3 x 32 = 96 O 12 12 x 16 = 192 Gram form mass is the sum of the total mass of each element in the formula Add all the masses up 54+96+192 = 342 g/mol Avogadro’s Number Volume of Gas at STP 6.02 x 1023 1 mole = 22.4 L Atoms / mole Percent Composition “ parts per hundred” divide the quantity under consideration by the total quantity involved Part x 100 whole Table T For any % composition problem--you must first find the formula mass of the compound What is the percent by mass of chlorine in CaCl2? Part x 100 whole 2Cl X 100 CaCl2 2(35.45) 110.98 X 100 % Cl = 63.89% What is the percent by mass of water in Na3PO4 •12H2O ? What is the percent by mass of water in hydrate: Mass of hydrate: 132.4g Mass of anhydrous 124.2g Find the formula mass first 3 x 23 = 69 1 x 31 = 31 4 x 16 = 64 + Mass of water To find mass of water Subtract the two #’s 12 x 18 = 216 Add them all up 69+31+64+216 = 380 g/mol Now find % 216 g/mol X 100 =56.8 % 380 g/mol 132.4 -124.2 = 8.2 g Now find % 8.2 g X 100 = 6.2 % 132.4 g Empirical Formula the simplest integer ratio in which atoms combine to form a compound 75 % C and 25% H What is the empirical formula? 1. Change % to grams 75g 1. Change % to grams 75g 2. Find number of moles 12g 2. Find number of moles 3. Divide by the smallest number 6.25 4. find ratio (becomes the subscripts) 3. Divide by the smallest number 6.25 5. Write the formula 6.25 4. find ratio 1 to 4 5.formula Molecular Formula the actual ratio of the atoms in a molecule 1. Write down your given 2. Find the molecular mass of the compound 3. Divide the mass given in the problem by the Molecular mass 4. Multiply subscripts by Answer to step 2 CH4 The molecular mass of a compound with an empirical formula of CH2 is 28g, what is the molecular formula? 1. Find formula 12 + 2(1) = 14 g/mol mass 2. 28g =2 14 g/mol 3. C2H4 25g 25g 1g 25 25 6.25 Density D= M V For a gas at STP Table T : D = Formula wt 22.4L CO2 = [12 + 2(16)] 22.4 L Reactions Chemical Reaction Chemical sentence that describes a reaction Reactants Products + = and Yields or produces Equilibrium- reaction forward and reverse (s) solid (l) (g) liquid gas (aq) Solution dissolved in water heat D Pt Heat was added catalyst Balancing Equations Law of Conservation of Mass-Energy Mass, energy, & charge are conserved (MEC) Balance equations by using coefficients 1. Determine if the reaction is combustion (CxHy +O2 or not If it is: CO2 + H2O) If it’s not: 2. Start with the carbons & make sure they are the same on both sides of the arrow. 3. Next balance the H’s 4. Finally balance the O’s- watch out they are separated on the product side! 2. Find the most complicated formula 3. By inspection use coefficient to balance atoms (each element!!) 4. Continue to add coefficients until you have same number on both sides Check your work!!!! Hydrogen plus oxygen yields water H2 + O2 = H2O OR H2 + O2 H2O How many H’s do I have on both sides of the equation? 2 Now, How many O’s do I have on both sides of the equation? 2 on left, 1 on right Put a 2 as a coefficient in front of the H2O H2 + O2 2H2O How does that affect your number of H’s? How do we fix it? 2H2 + O2 2H2O Place a 2 in front of The H on left side to balance Nitrogen plus hydrogen yields ammonia Look at both sides N2 + H2 NH3 N2 + 3H2 2NH3 Pick one element and balance firs Place 2 in front of NH3 Now go back and balance H’s Types of Chemical Reactions A compound is burned with oxygen and produces CO2 and H2O. The compound can contain C, H, & O. Burning Incomplete Combustion: CxHy + O2 Complete Combustion: CxHy + O2 Combination of two separate things to make ONE new thing. CREATE/Construct 2 1 A+B AB 4Fe+3O2 + (decompose) Break apart a compound into two or more things. Deconstruct 1 AB 2 A+B Examples Examples Sr + I2 Decomposition SrI2 2Fe2O3 2H2O2 Mg(Cl03)2 2H2O + O2 MgCl2 + 3O2 + Single Replacement An element (all alone by itself) takes the place of an element in a compound to form a new compound and a new element by itself. Stud kicks out wimp A + BC AC + B Stud meter = Table J Higher elements replace lower. Higher elements are more reactive. Examples Mg + H2SO4 F2 + 2NaBr + Synthesis (Combination) CO + H20 CO2 + H20 MgSO4 + H2 Br2 + 2NaF + Combustion Double Replacement The positive charge elements swap. Two new compounds are formed ONLY IF one of the new compounds is insoluble. Swap Meet Use TABLE F to figure out if swap meet happens. Innies vs Outies +- +- AB + CD +- +- AD+ CB(s) Example CdBr2 + Na2S CdS(s) +2NaBr + + Mole Mall Expansion Grams Grams Mass PT Mass PT Liters Moles 22.4 Use coefficients Moles Set up a proportion 6.02x1023 # particles Liters 22.4 6.02x1023 # particles