Chemical Reactions - hrsbstaff.ednet.ns.ca
... What is a chemical reaction? • A chemical reaction is a chemical change where chemical substances (called reactants) react to give new chemical substances (called products). • Example – The combustion of hydrogen in oxygen is a chemical reaction which gives water. • Hydrogen and Oxygen are the reac ...
... What is a chemical reaction? • A chemical reaction is a chemical change where chemical substances (called reactants) react to give new chemical substances (called products). • Example – The combustion of hydrogen in oxygen is a chemical reaction which gives water. • Hydrogen and Oxygen are the reac ...
CHEM 101 Final (Term 151)
... the produced gas, if the total pressure is 1.00 atm. (Vapor pressure of water at 25 °C is 0.0313 atm). A) 3.0 x102 mL B) 2.8 x103 mL C) 9.1 x103 mL D) 5.6 x102 mL E) 1.4 x 102 mL ...
... the produced gas, if the total pressure is 1.00 atm. (Vapor pressure of water at 25 °C is 0.0313 atm). A) 3.0 x102 mL B) 2.8 x103 mL C) 9.1 x103 mL D) 5.6 x102 mL E) 1.4 x 102 mL ...
Stuff Matters Handout
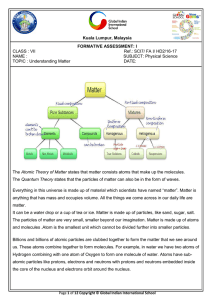
... Matter is everything around you. Matter is anything made of atoms and molecules. Matter is anything that has mass and takes up space. If you are new to the idea of mass, it is the amount of stuff in an object. Matter is sometimes related to light and electromagnetic radiation. Even though matter can ...
... Matter is everything around you. Matter is anything made of atoms and molecules. Matter is anything that has mass and takes up space. If you are new to the idea of mass, it is the amount of stuff in an object. Matter is sometimes related to light and electromagnetic radiation. Even though matter can ...
The chemical building blocks of life Carbon
... The comparison with other molecules generally favours water as an optimal medium for life For instance other polar solvents, such as HF, are interesting in principle, but are by far less abundant than water in the cosmos. Here the index in the row (g) represents the product of the cosmic abundances ...
... The comparison with other molecules generally favours water as an optimal medium for life For instance other polar solvents, such as HF, are interesting in principle, but are by far less abundant than water in the cosmos. Here the index in the row (g) represents the product of the cosmic abundances ...
water - Portal UniMAP
... Water is a common chemical substance that is essential for the survival of all known forms of life. (In typical usage, water refers only to its liquid form or state, but the substance also has a solid state, ice, and a gaseous state, water vapor. ...
... Water is a common chemical substance that is essential for the survival of all known forms of life. (In typical usage, water refers only to its liquid form or state, but the substance also has a solid state, ice, and a gaseous state, water vapor. ...
water - Portal UniMAP
... Water is a common chemical substance that is essential for the survival of all known forms of life. (In typical usage, water refers only to its liquid form or state, but the substance also has a solid state, ice, and a gaseous state, water vapor. ...
... Water is a common chemical substance that is essential for the survival of all known forms of life. (In typical usage, water refers only to its liquid form or state, but the substance also has a solid state, ice, and a gaseous state, water vapor. ...
Solutions, Acids, and Bases
... temperature is said to be saturated. Unsaturated = contains less solute than it can possibly hold Supersaturated = a solution that holds more solute than it should at a given temperature. ...
... temperature is said to be saturated. Unsaturated = contains less solute than it can possibly hold Supersaturated = a solution that holds more solute than it should at a given temperature. ...
Soquids Answers M/C 58. C 68. C 27. E 54. A 21. A 49. C 50. B 51
... solvent because the solution has a lower vapor pressure than the water (Raoult’s Law) . the temperature of the solution has be higher to produce enough vapor pressure to equal the atmospheric pressure (i.e., boiling) (ii) the amount of boiling point elevation depends on the number of non-volatile pa ...
... solvent because the solution has a lower vapor pressure than the water (Raoult’s Law) . the temperature of the solution has be higher to produce enough vapor pressure to equal the atmospheric pressure (i.e., boiling) (ii) the amount of boiling point elevation depends on the number of non-volatile pa ...
worksheer format 11-12
... they can flow and are described as being fluid, while solids do not possess fluidity; instead they are rigid in nature. The rigidity of solids is due to the presence of strong intermolecular force between their constituent particles. Hence particles are not free to move and they oscillate at their f ...
... they can flow and are described as being fluid, while solids do not possess fluidity; instead they are rigid in nature. The rigidity of solids is due to the presence of strong intermolecular force between their constituent particles. Hence particles are not free to move and they oscillate at their f ...
SampleTest3withAnswers
... B) steam condenses to liquid C) water evaporates D) more than one response is correct _D___11. Which of the following is an exothermic process? A) Sublimation [(s) to (g)] B) melting C) evaporation D) condensation _C___12. The vapor pressure of a liquid A) decreases with increasing temperature B) is ...
... B) steam condenses to liquid C) water evaporates D) more than one response is correct _D___11. Which of the following is an exothermic process? A) Sublimation [(s) to (g)] B) melting C) evaporation D) condensation _C___12. The vapor pressure of a liquid A) decreases with increasing temperature B) is ...
CHEM 150
... ____ 23. Which of the following molecules can have only London dispersion forces? a. CH4 b. CO2 c. both (a) and (b) d. neither (a) nor (b) ____ 24. Which of the following molecules cannot engage in hydrogen bonding? a. CH4 b. NH3 c. H2O d. all of them ____ 25. When comparing a liquid with a gas at t ...
... ____ 23. Which of the following molecules can have only London dispersion forces? a. CH4 b. CO2 c. both (a) and (b) d. neither (a) nor (b) ____ 24. Which of the following molecules cannot engage in hydrogen bonding? a. CH4 b. NH3 c. H2O d. all of them ____ 25. When comparing a liquid with a gas at t ...
Summer Assignment Ch. 2-5
... concentrations carefully, and show the calculations here for preparing a 1-molar solution of sucrose. Steps to help you do this follow. The first step is done for you. Fill in the rest. Steps to prepare a solution: a. Write the molecular formula. C12H22O11 b. Use your periodic table to calculate the ...
... concentrations carefully, and show the calculations here for preparing a 1-molar solution of sucrose. Steps to help you do this follow. The first step is done for you. Fill in the rest. Steps to prepare a solution: a. Write the molecular formula. C12H22O11 b. Use your periodic table to calculate the ...
Name ………………………………………………… Unit 7: States of
... (1) Propanone has a higher vapor pressure and stronger intermolecular forces than water. (2) Propanone has a higher vapor pressure and weaker intermolecular forces than water. (3) Propanone has a lower vapor pressure and stronger intermolecular forces than water. (4) Propanone has a lower vapor pres ...
... (1) Propanone has a higher vapor pressure and stronger intermolecular forces than water. (2) Propanone has a higher vapor pressure and weaker intermolecular forces than water. (3) Propanone has a lower vapor pressure and stronger intermolecular forces than water. (4) Propanone has a lower vapor pres ...
Nature of Molecules and Water
... • Single most outstanding chemical property of water is its ability to form hydrogen bonds – Weak chemical associations that form between the partially negative O atoms and the partially positive H atoms of two water molecules • Each individual bond is weak • Cumulative effects are enormous • Respon ...
... • Single most outstanding chemical property of water is its ability to form hydrogen bonds – Weak chemical associations that form between the partially negative O atoms and the partially positive H atoms of two water molecules • Each individual bond is weak • Cumulative effects are enormous • Respon ...
Document
... tendency to ionize into corresponding hydrogen (H+) and hydroxide (OH-) ions: HOH <=> H+ + OH- The hydrogen (H+) ion is more commonly referred to as a “proton” - In essence, the proton (H+) largely exists as an hydronium ion (H3O+) in solution—ie the H+ is associated with another H2O molecule rather ...
... tendency to ionize into corresponding hydrogen (H+) and hydroxide (OH-) ions: HOH <=> H+ + OH- The hydrogen (H+) ion is more commonly referred to as a “proton” - In essence, the proton (H+) largely exists as an hydronium ion (H3O+) in solution—ie the H+ is associated with another H2O molecule rather ...
SampleTest3
... B) steam condenses to liquid C) water evaporates D) more than one response is correct ____11. Which of the following is an exothermic process? A) Sublimation [(s) to (g)] B) melting C) evaporation D) condensation ____12. The vapor pressure of a liquid A) decreases with increasing temperature B) is i ...
... B) steam condenses to liquid C) water evaporates D) more than one response is correct ____11. Which of the following is an exothermic process? A) Sublimation [(s) to (g)] B) melting C) evaporation D) condensation ____12. The vapor pressure of a liquid A) decreases with increasing temperature B) is i ...
Bio 102 Lecture - chapter 2 The Chemical Basis of Life
... Heat of Vaporization is the quantity of heat a liquid must absorb to be converted from the liquid to the gaseous state. A substance which has a high heat of vaporization requires more heat to turn from liquid to vapor. ...
... Heat of Vaporization is the quantity of heat a liquid must absorb to be converted from the liquid to the gaseous state. A substance which has a high heat of vaporization requires more heat to turn from liquid to vapor. ...
Hydrogen Chemistry of Basalt Aquifers -- Treiman et
... hydrogen gas should be produced. However, as we reported, when basalt and ground water are actually mixed together, hydrogen is not produced to any appreciable extent. Furthermore, their model predicts a pH of 11.9, whereas the pH in the real ground water (8.0) is much lower. We believe that the rea ...
... hydrogen gas should be produced. However, as we reported, when basalt and ground water are actually mixed together, hydrogen is not produced to any appreciable extent. Furthermore, their model predicts a pH of 11.9, whereas the pH in the real ground water (8.0) is much lower. We believe that the rea ...
L1 – CHEMISTRY FINAL REVIEW
... MP/BP depends on what type of compounds you are comparing them with. With other molecular comps. High, but compared to other types of cmpds. low. Non-Polar Mc’s- Cl2; CH4; nonconductors and low MP/BP. Will dissolve in non-polar Solvents only. Network Covalent- Ex; graphite, diamond and quartz; they ...
... MP/BP depends on what type of compounds you are comparing them with. With other molecular comps. High, but compared to other types of cmpds. low. Non-Polar Mc’s- Cl2; CH4; nonconductors and low MP/BP. Will dissolve in non-polar Solvents only. Network Covalent- Ex; graphite, diamond and quartz; they ...
02-Atoms-Molecules
... Nonpolar molecules are termed hydrophobic Water-fearing These do not form hydrogen bonds and are therefore not water soluble ...
... Nonpolar molecules are termed hydrophobic Water-fearing These do not form hydrogen bonds and are therefore not water soluble ...
Chemistry I Exams and Keys 2014 Season
... rises over several minutes. Eventually, bubbles appear at the bottom of the beaker and rise to the top of the liquid. More bubbles begin to appear all over the volume of the water as it starts to boil. After boiling continues for several minutes, which statement or statements below is/are completely ...
... rises over several minutes. Eventually, bubbles appear at the bottom of the beaker and rise to the top of the liquid. More bubbles begin to appear all over the volume of the water as it starts to boil. After boiling continues for several minutes, which statement or statements below is/are completely ...
water properties - What is Chemistry
... Water is a powerful solvent that solubilizes a large variety of solid, liquid and gaseous chemicals, like carbohydrates, amines, acids and alkalis, and inorganic salts. Many substances separate as electrically charged particles, called ions, when dissolved in water. Such substances are called electr ...
... Water is a powerful solvent that solubilizes a large variety of solid, liquid and gaseous chemicals, like carbohydrates, amines, acids and alkalis, and inorganic salts. Many substances separate as electrically charged particles, called ions, when dissolved in water. Such substances are called electr ...
Chemistry: the study of composition, structure, and properties of
... An element is a pure substance made of only one kind of atom. – Carbon is an element made of only carbon atoms. Atoms of two or more elements bond together to make compounds. – CO2 (___ carbon atom and ___ oxygen atoms) – H2O2 (___ hydrogen atoms & ___ oxygen atoms) ...
... An element is a pure substance made of only one kind of atom. – Carbon is an element made of only carbon atoms. Atoms of two or more elements bond together to make compounds. – CO2 (___ carbon atom and ___ oxygen atoms) – H2O2 (___ hydrogen atoms & ___ oxygen atoms) ...
Physical Properties
... • Hydrogen bonds occur between two polar molecules, or between different polar regions of one large macromolecule. • One “relatively” negative region is attracted to a second “relatively” positive region. ...
... • Hydrogen bonds occur between two polar molecules, or between different polar regions of one large macromolecule. • One “relatively” negative region is attracted to a second “relatively” positive region. ...
CHAPTER 2 THE CHEMISTRY OF LIFE 2.1 Chemical Elements
... charge. A hydrogen bond is a weak attraction between a slightly positive hydrogen atom and a slightly negative oxygen or nitrogen atom within the same or a different molecule. Many hydrogen bonds taken together are relatively strong and help maintain the structure and function of cellular molecules ...
... charge. A hydrogen bond is a weak attraction between a slightly positive hydrogen atom and a slightly negative oxygen or nitrogen atom within the same or a different molecule. Many hydrogen bonds taken together are relatively strong and help maintain the structure and function of cellular molecules ...
Properties of water
Water (H2O) is the most abundant compound on Earth's surface, covering 70 percent of the planet. In nature, water exists in liquid, solid, and gaseous states. It is in dynamic equilibrium between the liquid and gas states at standard temperature and pressure. At room temperature, it is a tasteless and odorless liquid, nearly colorless with a hint of blue. Many substances dissolve in water and it is commonly referred to as the universal solvent. Because of this, water in nature and in use is rarely pure and some properties may vary from those of the pure substance. However, there are also many compounds that are essentially, if not completely, insoluble in water. Water is the only common substance found naturally in all three common states of matter and it is essential for all life on Earth. Water makes up 55% to 78% of the human body.