Electromagnetic Spectrum and Light
... The Electromagnetic Spectrum The electromagnetic spectrum consists of radio waves, infrared rays ,visible light, ultraviolet rays, x-rays, and gamma rays Each type of wave is characterized by specific wavelength and frequencies As wavelength shortens, frequency becomes higher ...
... The Electromagnetic Spectrum The electromagnetic spectrum consists of radio waves, infrared rays ,visible light, ultraviolet rays, x-rays, and gamma rays Each type of wave is characterized by specific wavelength and frequencies As wavelength shortens, frequency becomes higher ...
Chapter 7 Lect. 2
... III. Orbital Shapes and Energies A. Atomic orbital shapes are surfaces that surround 90% of the total probability of where its electrons are 1. Look at l = 0, the s-orbitals 2. Basic shape of an s-orbital is spherical centered on the nucleus 3. Basic shape is same for same l values 4. Nodes = area ...
... III. Orbital Shapes and Energies A. Atomic orbital shapes are surfaces that surround 90% of the total probability of where its electrons are 1. Look at l = 0, the s-orbitals 2. Basic shape of an s-orbital is spherical centered on the nucleus 3. Basic shape is same for same l values 4. Nodes = area ...
Chapter 13 Spectroscopy NMR, IR, MS, UV-Vis
... is deshielded from the applied field, shifted downfield, a smaller field is needed to bring it into resonance. Three kinds of carbon. Note how deshielded the carbonyl carbon is. Look at the chemical shift ranges. Very different for the different nuclei. TMS is tetramethylsilane and is chosen because ...
... is deshielded from the applied field, shifted downfield, a smaller field is needed to bring it into resonance. Three kinds of carbon. Note how deshielded the carbonyl carbon is. Look at the chemical shift ranges. Very different for the different nuclei. TMS is tetramethylsilane and is chosen because ...
PHOTONS AND PHOTON STATISTICS
... Classical Electrodynamics. Maxwell’s theory is perfectly adequate for understanding diffraction, interference, image formation, and even nonlinear phenomena such as frequency doubling or mixing. However, many fascinating quantum effects like correlations between photons are not captured, e.g., the p ...
... Classical Electrodynamics. Maxwell’s theory is perfectly adequate for understanding diffraction, interference, image formation, and even nonlinear phenomena such as frequency doubling or mixing. However, many fascinating quantum effects like correlations between photons are not captured, e.g., the p ...
High Resolution Laboratory UV Spectroscopy for Cassini UVIS at Titan
... Molecular Bands or NI,II lines per UVIS feature 16 UVIS features vs 800 Lab features (8001350 Å) ...
... Molecular Bands or NI,II lines per UVIS feature 16 UVIS features vs 800 Lab features (8001350 Å) ...
manual
... analysis to obtain approximate solutions instead of exact solutions. Since it takes too much time to get the solutions manually by numerical methods, ...
... analysis to obtain approximate solutions instead of exact solutions. Since it takes too much time to get the solutions manually by numerical methods, ...
Pretest 4.3 2008
... a. In Period 2, electronegativity increases as the atomic number increases. b. In Period 2, ionization energy decreases as the atomic number increases. c. In Period 2, atomic radius does not change as the atomic number increases. d. In group 1 (alkali metals), boiling points decrease and then increa ...
... a. In Period 2, electronegativity increases as the atomic number increases. b. In Period 2, ionization energy decreases as the atomic number increases. c. In Period 2, atomic radius does not change as the atomic number increases. d. In group 1 (alkali metals), boiling points decrease and then increa ...
ELECTRON I: Free electron model
... occupy one state with the same energy, a state labled by quantum number n can accommodate two electrons. The Fermi energy, defined as the energy of the topmost filled state (relative to the energy of the ground state), can be calculated by counting the number of electrons in the system at zero tempe ...
... occupy one state with the same energy, a state labled by quantum number n can accommodate two electrons. The Fermi energy, defined as the energy of the topmost filled state (relative to the energy of the ground state), can be calculated by counting the number of electrons in the system at zero tempe ...
Chemistry EOC Review
... 34. How are frequency and wavelength related? 35. Calculate the wavelength of a yellow light by a sodium lamp if the frequency of the radiation is 3.34 x 10 14 Hz. ...
... 34. How are frequency and wavelength related? 35. Calculate the wavelength of a yellow light by a sodium lamp if the frequency of the radiation is 3.34 x 10 14 Hz. ...
Covalent Bonds - WordPress.com
... The Energy Levels of Electrons • Electrons of atoms participate in reactions • Energy is the capacity to cause change • Potential energy is the energy that matter has because of its location or structure • The electrons of an atom differ in their amounts of potential energy • An electron’s state of ...
... The Energy Levels of Electrons • Electrons of atoms participate in reactions • Energy is the capacity to cause change • Potential energy is the energy that matter has because of its location or structure • The electrons of an atom differ in their amounts of potential energy • An electron’s state of ...
know thy reference tables!
... Litharge, PbO, is an ore that can be roasted (heated) in the presence of carbon monoxide, CO, to produce elemental lead. The reaction that takes place during this roasting process is represented by the balanced equation below. ...
... Litharge, PbO, is an ore that can be roasted (heated) in the presence of carbon monoxide, CO, to produce elemental lead. The reaction that takes place during this roasting process is represented by the balanced equation below. ...
The Fundamentals of Infrared Spectroscopy
... e = molar extinction coefficient (or molar absorptivity) c = concentration L = sample pathlength An absorption band can be characterized by two parameters; the wavelength at which maximum absorption occurs, and the intensity of absorption at this wavelength. In an absorption spectrum, the intensity ...
... e = molar extinction coefficient (or molar absorptivity) c = concentration L = sample pathlength An absorption band can be characterized by two parameters; the wavelength at which maximum absorption occurs, and the intensity of absorption at this wavelength. In an absorption spectrum, the intensity ...
Atomic Emission Spectra, Electron Configuration, Periodicity
... are able to “jump” to higher energy levels in the atom. We call this an electron’s excited state. The excited state is, however, unstable and the electron soon falls back to its original, lower energy position in the atom, we call this its ground state. As it travels back towards its ground state it ...
... are able to “jump” to higher energy levels in the atom. We call this an electron’s excited state. The excited state is, however, unstable and the electron soon falls back to its original, lower energy position in the atom, we call this its ground state. As it travels back towards its ground state it ...
Chapter 5: Electrons in Atoms
... Hund’s rule: every orbital is singly occupied with one electron before any orbital is doubly occupied, all electrons in singly occupied orbitals have the same spin. ...
... Hund’s rule: every orbital is singly occupied with one electron before any orbital is doubly occupied, all electrons in singly occupied orbitals have the same spin. ...
Generation of highly entangled photon pairs for continuous variable
... significantly broadened. Specific materials meet this condition only at certain wavelengths, thus limiting the usefulness of such an approach. We will show, however, that through the use of crystal superlattices, the overall GVD term can be made to vanish provided a certain condition (see below) is me ...
... significantly broadened. Specific materials meet this condition only at certain wavelengths, thus limiting the usefulness of such an approach. We will show, however, that through the use of crystal superlattices, the overall GVD term can be made to vanish provided a certain condition (see below) is me ...
7. In CCl 4 carbon is the “central atom”. In NF3 nitrogen is the
... 24 electrons instead of 22. (Where did the extra two electrons come from?) ...
... 24 electrons instead of 22. (Where did the extra two electrons come from?) ...
This Week Final Exam Marks on the Web
... • The electrons suffer centripetal acceleration in their orbits. • Any accelerated charge should radiate electromagnetic energy. ⇒ The electrons should lose energy and spiral into the nucleus in very little time. ⇒ A planetary atom should not be stable! ⇒ Classical theory does not explain the st ...
... • The electrons suffer centripetal acceleration in their orbits. • Any accelerated charge should radiate electromagnetic energy. ⇒ The electrons should lose energy and spiral into the nucleus in very little time. ⇒ A planetary atom should not be stable! ⇒ Classical theory does not explain the st ...
Chapter 4
... – Matter is simply material, such as rocks, water, or air. – Mass is the amount of matter an object has. ...
... – Matter is simply material, such as rocks, water, or air. – Mass is the amount of matter an object has. ...
Optical Properties of Silica-Copper Oxide Thin Films Prepared by Spin Coating
... and the expressions for rs and rp in an interface between layers change, as effects from reflection in other interfaces must be accounted for, but the principle is still the same. Instead of expressing material properties through n and k, the dielectric constant can be used (eq. 8) where the constan ...
... and the expressions for rs and rp in an interface between layers change, as effects from reflection in other interfaces must be accounted for, but the principle is still the same. Instead of expressing material properties through n and k, the dielectric constant can be used (eq. 8) where the constan ...
Word - The Chemistry Book
... extensive research on atomic fission reaction. Manhattan Project Below the football field at the University of Chicago, the United States developed the very first working nuclear fission reactor. The Manhattan Project was in process. ...
... extensive research on atomic fission reaction. Manhattan Project Below the football field at the University of Chicago, the United States developed the very first working nuclear fission reactor. The Manhattan Project was in process. ...
C. - Biloxi Public Schools
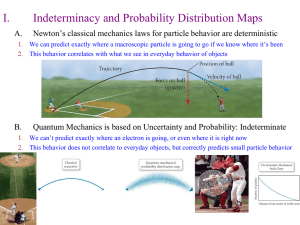
... • The Heisenberg uncertainty principle states that it is fundamentally impossible to know precisely both the velocity and position of a particle at the same time. • The only quantity that can be known is the probability for an electron to occupy a certain region around the nucleus. ...
... • The Heisenberg uncertainty principle states that it is fundamentally impossible to know precisely both the velocity and position of a particle at the same time. • The only quantity that can be known is the probability for an electron to occupy a certain region around the nucleus. ...
X-ray fluorescence

X-ray fluorescence (XRF) is the emission of characteristic ""secondary"" (or fluorescent) X-rays from a material that has been excited by bombarding with high-energy X-rays or gamma rays. The phenomenon is widely used for elemental analysis and chemical analysis, particularly in the investigation of metals, glass, ceramics and building materials, and for research in geochemistry, forensic science and archaeology.