Lecture Details - Earth and Atmospheric Sciences
... Darwin was unaware of the mechanism of inheritance. The term genetics was not coined until the early 20 th century. Gregor Mendel (1822-1884), who was Darwin’s contemporary, worked out fundamentals of genetics, but Darwin was unaware of it. Molecular basis of inheritance, DNA, not discovered until 1 ...
... Darwin was unaware of the mechanism of inheritance. The term genetics was not coined until the early 20 th century. Gregor Mendel (1822-1884), who was Darwin’s contemporary, worked out fundamentals of genetics, but Darwin was unaware of it. Molecular basis of inheritance, DNA, not discovered until 1 ...
Chapter 2
... major groups of inorganic chemicals common in a cell and describe the various types of organic chemicals in a cell (a.k.a. Molecules of Life) ...
... major groups of inorganic chemicals common in a cell and describe the various types of organic chemicals in a cell (a.k.a. Molecules of Life) ...
The Chemical Basis of Life
... Chemical reactions involve making and breaking bonds. Making bonds requires the input of energy. Energy is stored in molecules in the chemical bonds. Breaking bonds usually releases energy. ...
... Chemical reactions involve making and breaking bonds. Making bonds requires the input of energy. Energy is stored in molecules in the chemical bonds. Breaking bonds usually releases energy. ...
Explain how the study of living materials requires understanding of
... 2. Describe the relationships among matter, atoms and molecules. • Matter –anything that occupies space, solid liquid, or gas. • Elements-all matter is composed of elements, simplest form of matter, chemical reactions cannot change, an element is made up of millions of identical atoms • Atoms-small ...
... 2. Describe the relationships among matter, atoms and molecules. • Matter –anything that occupies space, solid liquid, or gas. • Elements-all matter is composed of elements, simplest form of matter, chemical reactions cannot change, an element is made up of millions of identical atoms • Atoms-small ...
Carbohydrates
... When was the last time you saw a flowering field of disodium phosphate? Or how about a fresh crop of maltodextrin? Didn’t think so. These cheese puffs consist largely of corn, but they’ve been processed to the point that no 20th-century farmer would ever recognize them as food. ...
... When was the last time you saw a flowering field of disodium phosphate? Or how about a fresh crop of maltodextrin? Didn’t think so. These cheese puffs consist largely of corn, but they’ve been processed to the point that no 20th-century farmer would ever recognize them as food. ...
chemistry of life
... 1) Synthesis when two or more atoms or reactants bond to form a new, more complex structure. Synthesis requires energy and is important to the growth of body parts. 2) Decomposition the opposite of ...
... 1) Synthesis when two or more atoms or reactants bond to form a new, more complex structure. Synthesis requires energy and is important to the growth of body parts. 2) Decomposition the opposite of ...
Slide 1
... • What is pH and how is it important to living organisms? • What are the four macromolecules found in living organisms? • What are the structure (subunits) and function of these 4 macromolecules? • How are proteins organized and how is their shape important to their function? • How are DNA similar a ...
... • What is pH and how is it important to living organisms? • What are the four macromolecules found in living organisms? • What are the structure (subunits) and function of these 4 macromolecules? • How are proteins organized and how is their shape important to their function? • How are DNA similar a ...
Living Environment Regents Review
... • Misuse of Renewable and NonRenewable Resources- lack of oil has lead to a renewed interest in finding alternative methods of energy. ...
... • Misuse of Renewable and NonRenewable Resources- lack of oil has lead to a renewed interest in finding alternative methods of energy. ...
Anatomy and Physiology Chapter #2
... 1) Synthesis when two or more atoms or reactants bond to form a new, more complex structure. Synthesis requires energy and is important to the growth of body parts. 2) Decomposition the opposite of ...
... 1) Synthesis when two or more atoms or reactants bond to form a new, more complex structure. Synthesis requires energy and is important to the growth of body parts. 2) Decomposition the opposite of ...
Anatomy and Physiology Chapter #2
... 1) Synthesis when two or more atoms or reactants bond to form a new, more complex structure. Synthesis requires energy and is important to the growth of body parts. 2) Decomposition the opposite of ...
... 1) Synthesis when two or more atoms or reactants bond to form a new, more complex structure. Synthesis requires energy and is important to the growth of body parts. 2) Decomposition the opposite of ...
Standard B-2
... Organic molecules: contain carbon atoms; made of smaller units that bond to form larger molecules; energy is stored in these bonds Proteins: composed of chains of amino acids; same caloric value as carbohydrates ○ Amino acids are molecules that are composed of carbon, hydrogen, oxygen, nitrogen, and ...
... Organic molecules: contain carbon atoms; made of smaller units that bond to form larger molecules; energy is stored in these bonds Proteins: composed of chains of amino acids; same caloric value as carbohydrates ○ Amino acids are molecules that are composed of carbon, hydrogen, oxygen, nitrogen, and ...
Unit 2: The Chemistry of Life
... Using one 8.5 x 11 sheet of paper answer all questions and define all vocabulary. Take the time to look up and study those questions/concepts not immediately familiar. Any questions you still have mark with a star, and we will address them in class. The Nature of Matter Name and describe the three s ...
... Using one 8.5 x 11 sheet of paper answer all questions and define all vocabulary. Take the time to look up and study those questions/concepts not immediately familiar. Any questions you still have mark with a star, and we will address them in class. The Nature of Matter Name and describe the three s ...
C1 Topic 7 the earth revision Earth`s Structure (and rocks) 1. List the
... 27. What is the process called of how we can separate air and why is this method possible? ...
... 27. What is the process called of how we can separate air and why is this method possible? ...
Beginnings - Big Picture
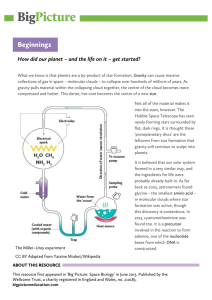
... the first organisms we would classify as alive? The most famous experiment into the origin of life on our planet was performed by Stanley Miller and Harold Urey in 1953. They tried to replicate the conditions of the early Earth in the lab, mixing water vapour with the gases that were thought to have ...
... the first organisms we would classify as alive? The most famous experiment into the origin of life on our planet was performed by Stanley Miller and Harold Urey in 1953. They tried to replicate the conditions of the early Earth in the lab, mixing water vapour with the gases that were thought to have ...
SBI 4U biochem 1
... • A polymer of nucleotides is a strand that joins adjacent nucleotides with a phosphodiester bond (between the phosphate and the hydroxyl groups) • DNA is two strands twisted in a double helix • The two strands are held together by hydrogen bonds • Thymine always bonds with adenine (with 2 H-bonds) ...
... • A polymer of nucleotides is a strand that joins adjacent nucleotides with a phosphodiester bond (between the phosphate and the hydroxyl groups) • DNA is two strands twisted in a double helix • The two strands are held together by hydrogen bonds • Thymine always bonds with adenine (with 2 H-bonds) ...
unit-review-key
... iii. A polymer consists of repeated, linked units, which can also bind forming large polymers called Macromolecules. (macro = large ) b. Monomers link to form polymers through a chemical reaction called condensation reaction or dehydration synthesis. During the formation of polymers, Water (H2O), is ...
... iii. A polymer consists of repeated, linked units, which can also bind forming large polymers called Macromolecules. (macro = large ) b. Monomers link to form polymers through a chemical reaction called condensation reaction or dehydration synthesis. During the formation of polymers, Water (H2O), is ...
Living Environment Regents Review
... of cells Cells are the basic unit of structure and function of all organisms. Cells come from pre-existing cells. All cells are surrounded by a thin membrane, that controls the movement of materials in and out of the cell (cell membrane). ...
... of cells Cells are the basic unit of structure and function of all organisms. Cells come from pre-existing cells. All cells are surrounded by a thin membrane, that controls the movement of materials in and out of the cell (cell membrane). ...
Chemical reaction
... • Contain carbon atoms that are covalently bonded to other carbon atoms and to other elements : Typically hydrogen (H), nitrogen (N) and oxygen (O) ...
... • Contain carbon atoms that are covalently bonded to other carbon atoms and to other elements : Typically hydrogen (H), nitrogen (N) and oxygen (O) ...
Chapter 26
... Earth formed about 4.6 billion years ago Conditions on early Earth were very different from today “Primitive soup” experiment of Miller and Urey ...
... Earth formed about 4.6 billion years ago Conditions on early Earth were very different from today “Primitive soup” experiment of Miller and Urey ...
Molecules of Life
... • Polymer – Poly = many, meros = parts – A large molecule that contains many molecules – A large molecule made of smaller, molecules of the same type (monomers) linked together. • A protein (the polymer) is made of many amino acids (monomers) ...
... • Polymer – Poly = many, meros = parts – A large molecule that contains many molecules – A large molecule made of smaller, molecules of the same type (monomers) linked together. • A protein (the polymer) is made of many amino acids (monomers) ...
Some Bio 230 Exam I Topics
... 2. The experiments of Urey and Miller provided evidence that: a. it is likely that the building blocks of life came from meterorites that hit the earth b. the reducing atmosphere of the early earth was conducive to the formation of abiotically-produced organic molecules. c. today¹s atmosphere could ...
... 2. The experiments of Urey and Miller provided evidence that: a. it is likely that the building blocks of life came from meterorites that hit the earth b. the reducing atmosphere of the early earth was conducive to the formation of abiotically-produced organic molecules. c. today¹s atmosphere could ...
Organic Chemistry and Biochemistry Essential Concepts
... Carbon is unique among elements in that it can bond to other carbon atoms to form long chains and rings. Millions of organic compounds exist. Among them are hydrocarbons (which contain only hydrogen and carbon), and polymers (large molecules made up of many smaller, repeating units), including plast ...
... Carbon is unique among elements in that it can bond to other carbon atoms to form long chains and rings. Millions of organic compounds exist. Among them are hydrocarbons (which contain only hydrogen and carbon), and polymers (large molecules made up of many smaller, repeating units), including plast ...
Abiogenesis

Abiogenesis (Brit.: /ˌeɪbaɪ.ɵˈdʒɛnɨsɪs/ AY-by-oh-JEN-ə-siss U.S. English pronunciation: /ˌeɪˌbaɪoʊˈdʒɛnᵻsɪs/), or biopoiesis, is the natural process of life arising from non-living matter, such as simple organic compounds. It is thought to have occurred on Earth between 3.8 and 4 billion years ago, and is studied through a combination of laboratory experiments and extrapolation from the genetic information of modern organisms in order to make reasonable conjectures about what pre-life chemical reactions may have given rise to a living system.The study of abiogenesis involves three main types of considerations: the geophysical, the chemical, and the biological, with more recent approaches attempting a synthesis of all three. Many approaches investigate how self-replicating molecules, or their components, came into existence. It is generally accepted that current life on Earth descended from an RNA world, although RNA-based life may not have been the first life to have existed. The Miller–Urey experiment and similar experiments demonstrated that most amino acids, basic chemicals of life, can be synthesized from inorganic compounds in conditions intended to be similar to early Earth. Several mechanisms have been investigated, including lightning and radiation. Other approaches (""metabolism first"" hypotheses) focus on understanding how catalysis in chemical systems in the early Earth might have provided the precursor molecules necessary for self-replication. Complex organic molecules have been found in the Solar System and in interstellar space, and these molecules may have provided starting material for the development of life on Earth.According to the panspermia hypothesis, microscopic life—distributed by meteoroids, asteroids and other small Solar System bodies—may exist throughout the Universe. It is speculated that the biochemistry of life may have begun shortly after the Big Bang, 13.8 billion years ago, during a habitable epoch when the age of the universe was only 10–17 million years.Nonetheless, Earth is the only place in the Universe known to harbor life. The age of the Earth is about 4.54 billion years. The earliest undisputed evidence of life on Earth dates at least from 3.5 billion years ago, during the Eoarchean Era after a geological crust started to solidify following the earlier molten Hadean Eon. There are microbial mat fossils found in 3.48 billion-year-old sandstone discovered in Western Australia. Other early physical evidence of a biogenic substance is graphite in 3.7 billion-year-old metasedimentary rocks discovered in southwestern Greenland.