9F Reactivity - Parrs Wood High School
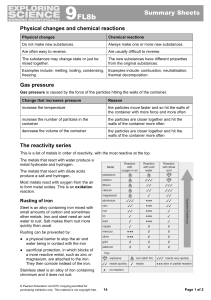
... These substances contain hydrogen and carbon only. They burn in a plentiful supply of air to form carbon dioxide and water: hydrocarbon + oxygen → carbon dioxide + water The test for oxygen is that it relights a glowing splint. An input of energy from a flame or spark is needed to start the combusti ...
... These substances contain hydrogen and carbon only. They burn in a plentiful supply of air to form carbon dioxide and water: hydrocarbon + oxygen → carbon dioxide + water The test for oxygen is that it relights a glowing splint. An input of energy from a flame or spark is needed to start the combusti ...
lecture 13
... BALANCING EQUATIONS: The same number of each type of element must occur on the left (BEFORE the reaction) and on the right (AFTER the reaction) ...
... BALANCING EQUATIONS: The same number of each type of element must occur on the left (BEFORE the reaction) and on the right (AFTER the reaction) ...
Critical Point Dryer
... First precursor gas (A Source) is introduced into the process chamber and produces a monolayer on the wafer surface. Then a second precursor gas (B Source) is introduced into the chamber, which reacts with the first precursor to produce a monolayer of film on the wafer surface. Separation of the pre ...
... First precursor gas (A Source) is introduced into the process chamber and produces a monolayer on the wafer surface. Then a second precursor gas (B Source) is introduced into the chamber, which reacts with the first precursor to produce a monolayer of film on the wafer surface. Separation of the pre ...
Balancing Single Replacement Reactions - Kossmann
... Name: __________________________ Class: ____________________ Date: _____________ ...
... Name: __________________________ Class: ____________________ Date: _____________ ...
HOMEWORK : CHAPTER 20
... 20.52 Explain the change in bonding when Al2Cl6 dissociates to form AlCl3 in the gas phase. 20.54 When 1.164 g of a certain metal sulfide was roasted in air, 0.972 g of the metal oxide was formed. If the oxidation number of the metal is +2, calculate the molar mass of the metal. 20.56 Referring to F ...
... 20.52 Explain the change in bonding when Al2Cl6 dissociates to form AlCl3 in the gas phase. 20.54 When 1.164 g of a certain metal sulfide was roasted in air, 0.972 g of the metal oxide was formed. If the oxidation number of the metal is +2, calculate the molar mass of the metal. 20.56 Referring to F ...
03 nanoparticles part 7 File - e-learning
... compounds in gas phase and the successive deposition of the produced solid material. The technique results costly for those materials exhibiting low vapor pressure values (ceramics). In the other cases, the technique offers all advantages working in gas phase; moreover, it is possible to address the ...
... compounds in gas phase and the successive deposition of the produced solid material. The technique results costly for those materials exhibiting low vapor pressure values (ceramics). In the other cases, the technique offers all advantages working in gas phase; moreover, it is possible to address the ...
Liquid-feed flame spray pyrolysis

Liquid–feed flame spray pyrolysis (LF-FSP) is one of the most recent iterations in flame spray pyrolysis (FSP) powder production technology. FSP produces metal oxide powders from highly volatile gaseous metal chlorides that are decomposed/oxidized in hydrogen-oxygen flames to form nano-oxide powders. However, products made from FSP's vapor-phase process are limited to Al-, Ti-, Zr-, and Si-based oxides from their metal chlorides. Thus, interest in producing more complex materials required a new methodology, LF-FSP.LF-FSP, as invented at the University of Michigan, uses metalloorganic precursors such as metal carboxylates or alkoxides, not metal chlorides. Briefly, alcohol (typically ethanol) solutions containing 1–10 wt % loading of the target ceramic components as precursors are aerosolized with O2 into a quartz chamber and ignited with methane pilot torches. Initial combustion temperatures run 1500–2000 °C, depending on the processing conditions, generating nanopowder ""soot"". Temperatures drop to 300–500 °C over 1.5 m, equivalent to a 1000 °C quench in 100 ms leading to kinetic products and nanopowders that are unaggregated. Production rates can be 200 g/h when using wire-in-tube electrostatic precipitators operating at 10 kV. Typical powders have 15–100 nm average particle sizes (APS) with specific surface areas of 30–100 m2/g. LF-FSP technology can be used to produce mixed and single metal oxides easily from low cost starting materials in a single step without forming harmful byproducts like HCl, which forms when metal chlorides are used as precursors.




