2A6
... Visible-light-induced molecular photodissociation of dimethyl disulfide ((CH3S)2) adsorbed on Ag(111) and Cu(111) surfaces was investigated by means of scanning tunneling microscopy (STM) combined with density functional theory (DFT) calculations. The visible-light-induced photodissociation on metal ...
... Visible-light-induced molecular photodissociation of dimethyl disulfide ((CH3S)2) adsorbed on Ag(111) and Cu(111) surfaces was investigated by means of scanning tunneling microscopy (STM) combined with density functional theory (DFT) calculations. The visible-light-induced photodissociation on metal ...
Unit 9 Chemical Equations and Reactions Balancing Equations Notes
... Synthesis- _____________________ elements or compounds combine to form one compound. Decomposition- a _________________ compound decomposes into two or more elements or smaller compounds. Single Replacement- a metal will _________________ a less active metal in an ionic compound OR a nonmetal will r ...
... Synthesis- _____________________ elements or compounds combine to form one compound. Decomposition- a _________________ compound decomposes into two or more elements or smaller compounds. Single Replacement- a metal will _________________ a less active metal in an ionic compound OR a nonmetal will r ...
Chapter 8
... designates an aqueous solution, one that is dissolved in water, placed after the formula. 8. __________ indicates that heat is supplied to the reaction 9. __________ a formula written above or below the yield sign indicates it is used as a catalyst. In this case, platinum. G. What is a skeleton equa ...
... designates an aqueous solution, one that is dissolved in water, placed after the formula. 8. __________ indicates that heat is supplied to the reaction 9. __________ a formula written above or below the yield sign indicates it is used as a catalyst. In this case, platinum. G. What is a skeleton equa ...
Burning to Learn: An Introduction to Flame Retardants
... acid (H3BO3) or laundry borax (Na2B4O7ⴢ10H2O), 3 small containers (to hold about 20 mL of solution), spoon or stirring rod, and paper towels. An oven or hair dryer is optional. A fire extinguisher should be available at all times. __1. Cut filter paper or coffee filters into 6 strips about 1 cm wide ...
... acid (H3BO3) or laundry borax (Na2B4O7ⴢ10H2O), 3 small containers (to hold about 20 mL of solution), spoon or stirring rod, and paper towels. An oven or hair dryer is optional. A fire extinguisher should be available at all times. __1. Cut filter paper or coffee filters into 6 strips about 1 cm wide ...
cbse class – x science solutions
... called charcoal. Charcoal burns without smoke and has higher calorific value. In an experiment with a rectangular glass slab, a student observed that a ray of light incident at an angle of 55o with the normal on one face of the slab, after refraction strikes the opposite face of the slab before emer ...
... called charcoal. Charcoal burns without smoke and has higher calorific value. In an experiment with a rectangular glass slab, a student observed that a ray of light incident at an angle of 55o with the normal on one face of the slab, after refraction strikes the opposite face of the slab before emer ...
Chapter 1: Aqueous Processing Systems
... various physical and chemical steps that connect ores to refined metals and to engineered materials. And in Aqueous Processing, in particular, our focus is on water-based chemical process technologies. Water is the most abundant solvent, and it has some unique properties, which facilitate the dissol ...
... various physical and chemical steps that connect ores to refined metals and to engineered materials. And in Aqueous Processing, in particular, our focus is on water-based chemical process technologies. Water is the most abundant solvent, and it has some unique properties, which facilitate the dissol ...
Preparation of Supported Catalysts
... stirring (equilibrium reached within several hours) separation (eventually multiple exchange ⇒ higher loadings) calcination, reduction, activation Example: ...
... stirring (equilibrium reached within several hours) separation (eventually multiple exchange ⇒ higher loadings) calcination, reduction, activation Example: ...
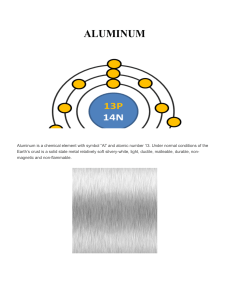
ALUMINUM
... Aluminum is highly reactive and it is very rare to find natively on the crust, so it is a very versatile, which can constitute about 300 alloys and a lot of variants. Despite this, aluminum does not ignite instantly, so it has many applications in handling flammable or explosive materials. Aluminum ...
... Aluminum is highly reactive and it is very rare to find natively on the crust, so it is a very versatile, which can constitute about 300 alloys and a lot of variants. Despite this, aluminum does not ignite instantly, so it has many applications in handling flammable or explosive materials. Aluminum ...
Belarus, National Final, 2001 (PDF 149K).
... fertilizer. Name this fertilizer and write the chemical equation for its production. Problem 9-5. A 1.92 L sample (at STP) of a gaseous mixture of hydrocarbons that is 12.6% elemental hydrogen (by mass) was burned in an excess of oxygen. The volatile combustion products were dried and bubbled throug ...
... fertilizer. Name this fertilizer and write the chemical equation for its production. Problem 9-5. A 1.92 L sample (at STP) of a gaseous mixture of hydrocarbons that is 12.6% elemental hydrogen (by mass) was burned in an excess of oxygen. The volatile combustion products were dried and bubbled throug ...
New Materials from Metal Vapour Chemistry
... In this particular application, a commercially available liquid poly( phenylmethyl-co-dimethyl ) siloxane in a rotary reactor is allowed to interact with resistively generated Fe atoms at around -40 to -50° C. to form polymer attached bis ( arene ) iron ( 0 ) ( Figure 1, also included for the purpos ...
... In this particular application, a commercially available liquid poly( phenylmethyl-co-dimethyl ) siloxane in a rotary reactor is allowed to interact with resistively generated Fe atoms at around -40 to -50° C. to form polymer attached bis ( arene ) iron ( 0 ) ( Figure 1, also included for the purpos ...
ST. PAUL`S CONVENT SCHOOL METALLIC RAINBOW 金屬彩虹
... In part I, NH3, en, citrate and glycine were chosen for Cu2+ for the conduction of colorimetry; EDTA, oxalate and tartrate for Ni2+; conc. HCl, en and tartrate for Co2+; NaBr, EDTA and leucine for Cr3+; KSCN, rust indicator, en, citrate and glycine for Fe3+. In part II, en and glycine were selec ...
... In part I, NH3, en, citrate and glycine were chosen for Cu2+ for the conduction of colorimetry; EDTA, oxalate and tartrate for Ni2+; conc. HCl, en and tartrate for Co2+; NaBr, EDTA and leucine for Cr3+; KSCN, rust indicator, en, citrate and glycine for Fe3+. In part II, en and glycine were selec ...
Biologically Important Inorganic Elements Occurrence and Availability
... Although Mo is rare in the earth’s crust, Mo is the most abundant transition metal in sea water as MoO4 has fairly high solubility in water. Better correlation exists between the abundance of elements in in human body and in sea water than between the human body and the earth's crust. Taken as evid ...
... Although Mo is rare in the earth’s crust, Mo is the most abundant transition metal in sea water as MoO4 has fairly high solubility in water. Better correlation exists between the abundance of elements in in human body and in sea water than between the human body and the earth's crust. Taken as evid ...
Reactions Flowchart
... 2NaCl 2Na + Cl2 • Metal Hydroxide Metal oxide + H2O Ca(OH)2 CaO + H2O • Metal Carbonate Metal oxide + CO2 Li2CO3 LiO + CO2 • Metal bicarbonate Metal carbonate+ CO2 + H2O NaHCO3 Na2CO3 + CO2 + H2O •Metal Nitrate Metal Oxide + NO2 + O2 2Mg(NO3)2 MgO + 4NO2 + O2 • Metal Chlorate Meta ...
... 2NaCl 2Na + Cl2 • Metal Hydroxide Metal oxide + H2O Ca(OH)2 CaO + H2O • Metal Carbonate Metal oxide + CO2 Li2CO3 LiO + CO2 • Metal bicarbonate Metal carbonate+ CO2 + H2O NaHCO3 Na2CO3 + CO2 + H2O •Metal Nitrate Metal Oxide + NO2 + O2 2Mg(NO3)2 MgO + 4NO2 + O2 • Metal Chlorate Meta ...
Chapter 9 Atomic Absorption and Atomic Fluorescence Spectrometry
... Types of Flames: Several common fuels and oxidants can be employed in flame spectroscopy depending on temperature needed. Temperatures of 1700oC to 2400oC are obtained with the various fuels when air serves as the oxidant. At these temperature, only easily decomposed samples are atomized. For more ...
... Types of Flames: Several common fuels and oxidants can be employed in flame spectroscopy depending on temperature needed. Temperatures of 1700oC to 2400oC are obtained with the various fuels when air serves as the oxidant. At these temperature, only easily decomposed samples are atomized. For more ...
12.26MB - Stanford University
... Variation of particle concentration (i.e., number, surface area, volume, or mass of particles per unit volume of air) with size. Size distribution usually divided into modes ...
... Variation of particle concentration (i.e., number, surface area, volume, or mass of particles per unit volume of air) with size. Size distribution usually divided into modes ...
terpconnect.umd.edu
... • Goal: Want a process in which particles having controlled characteristics such as size, morphology and composition can be produced, at the lowest cost and highest yield. ...
... • Goal: Want a process in which particles having controlled characteristics such as size, morphology and composition can be produced, at the lowest cost and highest yield. ...
8492_Chemichal Weapons Production Indicators
... The vapors of chlorinated solvents exposed to high temperatures have been known to produce phosgene. Chlorinated solvents are chlorine-containing chemicals that are typically used in industrial processes to dissolve or clean other materials, such as in paint stripping, metal cleaning, and dry cleani ...
... The vapors of chlorinated solvents exposed to high temperatures have been known to produce phosgene. Chlorinated solvents are chlorine-containing chemicals that are typically used in industrial processes to dissolve or clean other materials, such as in paint stripping, metal cleaning, and dry cleani ...
Unit 10
... Extracting metal – getting metal from ores. What are the metal compounds from mineral ores? Are they soluble in water? Insoluble metal oxides, carbonates and sulphides – found in ores Which metals are found free in nature? Unreactive metals such as gold and platinum found free (as elements) in natur ...
... Extracting metal – getting metal from ores. What are the metal compounds from mineral ores? Are they soluble in water? Insoluble metal oxides, carbonates and sulphides – found in ores Which metals are found free in nature? Unreactive metals such as gold and platinum found free (as elements) in natur ...
Specific Reactions Quiz.wpd
... a) various carbon products created due to lack of oxygen including solid carbon (black component) b) as air contacts the random carbon products (smaller hydrocarbons) created, they may further combust c) since energy is still tied up in carbon product bonds, energy is not released all at once d) the ...
... a) various carbon products created due to lack of oxygen including solid carbon (black component) b) as air contacts the random carbon products (smaller hydrocarbons) created, they may further combust c) since energy is still tied up in carbon product bonds, energy is not released all at once d) the ...
The s-Block Elements - GCG-42
... BeCl2 is essentially covalent, with comparatively low m.pt. The lower members in group II form essentially ionic chlorides, with Mg having intermediate properties. ...
... BeCl2 is essentially covalent, with comparatively low m.pt. The lower members in group II form essentially ionic chlorides, with Mg having intermediate properties. ...
v Plasma Particle Technology
... is possible, for example, to optimize wettability by liquids and thus produce stable aqueous dispersions. Furthermore, a coating can improve the flowability and separability of the particles by minimizing not only the mutual cohesion of the individual particles but also their unwanted adhesion to su ...
... is possible, for example, to optimize wettability by liquids and thus produce stable aqueous dispersions. Furthermore, a coating can improve the flowability and separability of the particles by minimizing not only the mutual cohesion of the individual particles but also their unwanted adhesion to su ...
Pollution Control - No Brain Too Small
... These dust-laden gases pass through an electrostatic precipitator that collects most of the dust. Cleaned gas then passes out of the precipitator and through a stack to the atmosphere. Precipitators typically collect 99.9% or more of the dust from the gas stream. Precipitators function by electrosta ...
... These dust-laden gases pass through an electrostatic precipitator that collects most of the dust. Cleaned gas then passes out of the precipitator and through a stack to the atmosphere. Precipitators typically collect 99.9% or more of the dust from the gas stream. Precipitators function by electrosta ...
File
... Lesson 2.4: Combustion Reactions In some areas of the Arctic, large amounts of methane, CH4 (g) are entering the atmosphere. Where is it coming from? As the climate becomes warmer and the ground thaws bacteria produce methane from the remains of dead plants and animals. How do scientists find the me ...
... Lesson 2.4: Combustion Reactions In some areas of the Arctic, large amounts of methane, CH4 (g) are entering the atmosphere. Where is it coming from? As the climate becomes warmer and the ground thaws bacteria produce methane from the remains of dead plants and animals. How do scientists find the me ...
Synthesis of Copper (II)-Containing Nickel (II) Hydroxide Particles as
... different hydrolytic and complexation chemistries of Cu(II) and Ni(II) would restrict the synthesis of the mixed hydroxides via coprecipitation to very narrow conditions, it has also important implications in the realm of the performance of materials. Indeed, both Ni1-xCuxO and its precursor, copper ...
... different hydrolytic and complexation chemistries of Cu(II) and Ni(II) would restrict the synthesis of the mixed hydroxides via coprecipitation to very narrow conditions, it has also important implications in the realm of the performance of materials. Indeed, both Ni1-xCuxO and its precursor, copper ...
Liquid-feed flame spray pyrolysis

Liquid–feed flame spray pyrolysis (LF-FSP) is one of the most recent iterations in flame spray pyrolysis (FSP) powder production technology. FSP produces metal oxide powders from highly volatile gaseous metal chlorides that are decomposed/oxidized in hydrogen-oxygen flames to form nano-oxide powders. However, products made from FSP's vapor-phase process are limited to Al-, Ti-, Zr-, and Si-based oxides from their metal chlorides. Thus, interest in producing more complex materials required a new methodology, LF-FSP.LF-FSP, as invented at the University of Michigan, uses metalloorganic precursors such as metal carboxylates or alkoxides, not metal chlorides. Briefly, alcohol (typically ethanol) solutions containing 1–10 wt % loading of the target ceramic components as precursors are aerosolized with O2 into a quartz chamber and ignited with methane pilot torches. Initial combustion temperatures run 1500–2000 °C, depending on the processing conditions, generating nanopowder ""soot"". Temperatures drop to 300–500 °C over 1.5 m, equivalent to a 1000 °C quench in 100 ms leading to kinetic products and nanopowders that are unaggregated. Production rates can be 200 g/h when using wire-in-tube electrostatic precipitators operating at 10 kV. Typical powders have 15–100 nm average particle sizes (APS) with specific surface areas of 30–100 m2/g. LF-FSP technology can be used to produce mixed and single metal oxides easily from low cost starting materials in a single step without forming harmful byproducts like HCl, which forms when metal chlorides are used as precursors.