Modeling Physical and Chemical Properties of Aerosols: A
... -nitrate formation still a problem (size dependence, role of particles such as dust, sea-salt, etc) -role of organics for aging (oxidation processes, oligomerization processes, etc.) Aqueous chemistry: -clear evidence of change in chemical composition due to sulfate oxidation, etc Both these process ...
... -nitrate formation still a problem (size dependence, role of particles such as dust, sea-salt, etc) -role of organics for aging (oxidation processes, oligomerization processes, etc.) Aqueous chemistry: -clear evidence of change in chemical composition due to sulfate oxidation, etc Both these process ...
7-1 Avogadro`s Number and Molar Conversions Objectives: • Identify
... 2. Amount in Moles Can Be Converted to Number of Particles - Board 3. Choose the Conversion Factor That Cancels the Given Units - Board 4. Number of Particles Can Be Converted to Amount in Moles - Board B Molar Mass Relates Moles to Grams 1. Amount in Moles Can Be Converted to Mass a.) The molar mas ...
... 2. Amount in Moles Can Be Converted to Number of Particles - Board 3. Choose the Conversion Factor That Cancels the Given Units - Board 4. Number of Particles Can Be Converted to Amount in Moles - Board B Molar Mass Relates Moles to Grams 1. Amount in Moles Can Be Converted to Mass a.) The molar mas ...
Matter-Wave Metrology as a Complementary Tool for Mass
... Quantum coherence of very large molecules may, however, also be explored in a near-field interference process, such as the Talbot–Lau interferometer.[5, 6] A recent extension of this idea, the Kapitza–Dirac–Talbot–Lau (KDTL) interferometer, even promises to be scalable to particles up to 1 000 000 a ...
... Quantum coherence of very large molecules may, however, also be explored in a near-field interference process, such as the Talbot–Lau interferometer.[5, 6] A recent extension of this idea, the Kapitza–Dirac–Talbot–Lau (KDTL) interferometer, even promises to be scalable to particles up to 1 000 000 a ...
12.26MB - Stanford University
... Last update: March 23, 2005 The photographs shown here appear in the textbook and are provided to facilitate their display during course instruction. Permissions for publication of photographs must be requested from individual copyright holders. The source of each photograph is given below the figur ...
... Last update: March 23, 2005 The photographs shown here appear in the textbook and are provided to facilitate their display during course instruction. Permissions for publication of photographs must be requested from individual copyright holders. The source of each photograph is given below the figur ...
Ch. 07 Notes ch7notes
... Chemical Formulas and Moles • Chemical formulas give a ratio of elemental components. • Ionic formulas show the simplest ratio of cations and anions. • Covalent formulas (including polyatomic ions) show both elements and the number of each element. Formulas can be used to calculate Molar Masses • Fr ...
... Chemical Formulas and Moles • Chemical formulas give a ratio of elemental components. • Ionic formulas show the simplest ratio of cations and anions. • Covalent formulas (including polyatomic ions) show both elements and the number of each element. Formulas can be used to calculate Molar Masses • Fr ...
Tuesday, Define the law of definite proportions
... The mole (mol) is a unit used to count particles indirectly. A mole, also called Avogadro’s number may be expressed as 6.02 x 10 23, gram molecular mass and molar mass of the representative particles of that substance (atoms, ions, molecules or formula units). One mole of a gas occupies a volume of ...
... The mole (mol) is a unit used to count particles indirectly. A mole, also called Avogadro’s number may be expressed as 6.02 x 10 23, gram molecular mass and molar mass of the representative particles of that substance (atoms, ions, molecules or formula units). One mole of a gas occupies a volume of ...
mass Spectrometry (mS)
... NOTE: Because the positive ion formed has an unpaired electron it is sometimes shown with a dot indicating that it is a free radical, e.g. CH3+• The positive ions are then ACCELERATED by an electric field and focused into a fine beam by passing through a series of slits with increasing negative pote ...
... NOTE: Because the positive ion formed has an unpaired electron it is sometimes shown with a dot indicating that it is a free radical, e.g. CH3+• The positive ions are then ACCELERATED by an electric field and focused into a fine beam by passing through a series of slits with increasing negative pote ...
Chemistry Midterm Review
... - ______________ which are chemical combinations of two or more of the building blocks and have properties much different than the building blocks from which they are formed. The smallest particle with all the properties of this material is called a _______________. EXAMPLE:_____________ Mixtures ar ...
... - ______________ which are chemical combinations of two or more of the building blocks and have properties much different than the building blocks from which they are formed. The smallest particle with all the properties of this material is called a _______________. EXAMPLE:_____________ Mixtures ar ...
03 nanoparticles part 7 File - e-learning
... aerosol. After this, the aerosol is converted to a gas suspension of solid nanoparticles. The techniques are: aerosol pyrolysis and aerosol flame pyrolysis The techniques are also called “spray pyrolysis” since the formation of the aerosol is given by a spray nozzle which generates very small liquid ...
... aerosol. After this, the aerosol is converted to a gas suspension of solid nanoparticles. The techniques are: aerosol pyrolysis and aerosol flame pyrolysis The techniques are also called “spray pyrolysis” since the formation of the aerosol is given by a spray nozzle which generates very small liquid ...
EXAM # 1
... F = 96485.31 C mol-1 e = -1.602177 x 10-19C SECTION I (Electrochemistry, MS and NMR) 1) (a) Calculate the theoretical potential of the following cell: Pt,H2(0.100 atm)|HCl(4.50x10-4 M), AgCl(saturated)|Ag AgCl(s) + e- Ag (s) + ClEo = +0.222 Ecathode = +0.222 – (0.0592/1) log ((4.50x10-4)(1)/1) = 0 ...
... F = 96485.31 C mol-1 e = -1.602177 x 10-19C SECTION I (Electrochemistry, MS and NMR) 1) (a) Calculate the theoretical potential of the following cell: Pt,H2(0.100 atm)|HCl(4.50x10-4 M), AgCl(saturated)|Ag AgCl(s) + e- Ag (s) + ClEo = +0.222 Ecathode = +0.222 – (0.0592/1) log ((4.50x10-4)(1)/1) = 0 ...
Mass Spectroscopy
... field. • Amount of deflection depends on m/z. • The detector signal is proportional to the number of ions hitting it. • By varying the magnetic field, ions of all masses are collected and counted. => ...
... field. • Amount of deflection depends on m/z. • The detector signal is proportional to the number of ions hitting it. • By varying the magnetic field, ions of all masses are collected and counted. => ...
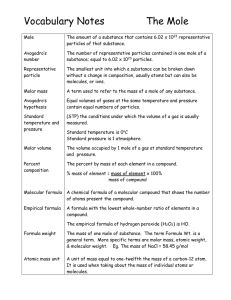
Vocabulary Notes
... The mass of one mole of substance. The term Formula Wt. is a general term. More specific terms are molar mass, atomic weight, & molecular weight. Eg. The mass of NaCl = 58.45 g/mol ...
... The mass of one mole of substance. The term Formula Wt. is a general term. More specific terms are molar mass, atomic weight, & molecular weight. Eg. The mass of NaCl = 58.45 g/mol ...
Mass Spectrometry
... • Sign up for turnitin.com • Formal reports due electronically up to time of lab • One minute24 hours late = 20pt penalty • Give copy of lab notebook to AI in lab ...
... • Sign up for turnitin.com • Formal reports due electronically up to time of lab • One minute24 hours late = 20pt penalty • Give copy of lab notebook to AI in lab ...
Sup Table 3. List of centres identified as performing mass
... Sup Table 3. List of centres identified as performing mass spectrometry BA analysis in urine to diagnose 3β-hydroxy-Δ5-C27-steroid oxidoreductase and Δ4-3-oxosteroid 5β-reductase deficiencies. BA, bile acids; MS, mass spectrometry. Centres for MS analysis of BA# ...
... Sup Table 3. List of centres identified as performing mass spectrometry BA analysis in urine to diagnose 3β-hydroxy-Δ5-C27-steroid oxidoreductase and Δ4-3-oxosteroid 5β-reductase deficiencies. BA, bile acids; MS, mass spectrometry. Centres for MS analysis of BA# ...
Mass spectrometry-led catalyst discovery
... capable of studying complex mixtures in solution, capabilities that make it, at first glance, the ideal technique for the direct analysis of homogeneous catalytic reactions. However, a variety of challenges have prevented ESI-MS from being more widely applied in this context, including the invisibil ...
... capable of studying complex mixtures in solution, capabilities that make it, at first glance, the ideal technique for the direct analysis of homogeneous catalytic reactions. However, a variety of challenges have prevented ESI-MS from being more widely applied in this context, including the invisibil ...
View PDF
... and microstructure of aerosol particles is now wellestablished for inferring key properties of the aerosol such as hygroscopicity, the activity of cloud condensation, the reactivity, the optical properties, etc. Aerosol particles consist of complex mixture of inorganic salts with hydrophilic and/or ...
... and microstructure of aerosol particles is now wellestablished for inferring key properties of the aerosol such as hygroscopicity, the activity of cloud condensation, the reactivity, the optical properties, etc. Aerosol particles consist of complex mixture of inorganic salts with hydrophilic and/or ...
Aerosol mass spectrometry

Aerosol mass spectrometry is the application of mass spectrometry to aerosol particles. Aerosol particles are defined as suspended solid and liquid particles with size range of 0.1 nm to 1000 μm in diameter. Aerosol particles are produce from natural and anthropogenic sources, through a variety of different process that include; wind-blown suspension, and combustion of fossil fuels and biomass. Analysis of aerosol particles is important because of their major impacts on the global climate change, visibility, regional air pollution and human health. Aerosol particles are very complex in structure and can contain thousand of different chemical compounds within a single particle. Due to this complexity the instrumentation used to analysis these particles must have the ability to separate based on size and in real-time provide information on their chemical composition. To meet these requirements for analysis, mass spectrometry instrumentation is used and they provide high sensitivity and the ability to detect a wide molecular mass range. Aerosol mass spectrometry can be divided into two categorizes; off-line and on-line. Off-line mass spectrometry is performed on collected particles. On-line mass spectrometry is performed on particles introduced in real time.