STUDY GUIDE
... MAIN IDEA: Hydrocarbons with multiple bonds are more reactive than alkanes and participate in addition reactions in which atoms from one molecule are added to another molecule. Addition reactions include hydrogenation, halogenation, hydrohalogenation, and hydration. Markovnikov’s rule states that, ...
... MAIN IDEA: Hydrocarbons with multiple bonds are more reactive than alkanes and participate in addition reactions in which atoms from one molecule are added to another molecule. Addition reactions include hydrogenation, halogenation, hydrohalogenation, and hydration. Markovnikov’s rule states that, ...
13.0 Redox Reactions PowerPoint
... transferred between entities • The total number of electrons gained in the reduction equals the total number of electrons lost in the oxidation • Reduction is a process in which electrons are gained by an entity • Oxidation is a process in which electrons are lost by an entity • Both reduction and o ...
... transferred between entities • The total number of electrons gained in the reduction equals the total number of electrons lost in the oxidation • Reduction is a process in which electrons are gained by an entity • Oxidation is a process in which electrons are lost by an entity • Both reduction and o ...
Science 11th grade LEARNING OBJECT Why are carboxylic acids
... molecules, boiling point, weak and strong acids. Relative clauses (which, that) Bridge (noun): a structure that is built over a river, road, or railway to allow people and vehicles to cross from one side to the other. Chemical bond (noun): an electrical force that holds atoms together to form a mole ...
... molecules, boiling point, weak and strong acids. Relative clauses (which, that) Bridge (noun): a structure that is built over a river, road, or railway to allow people and vehicles to cross from one side to the other. Chemical bond (noun): an electrical force that holds atoms together to form a mole ...
Firefly Bioluminescence
... presence of luciferase to produce light emission identical to that obtained with the natural substrates D-luciferin and Mg-ATP. ...
... presence of luciferase to produce light emission identical to that obtained with the natural substrates D-luciferin and Mg-ATP. ...
NOBLE-GAS CHEMISTRY
... adducts of noble gases to a metal center, NgBe(II)O, where Ng = Ar, Kr, and Xe.31 The case of XeBeO is rather straightforward: a coordinatively unsaturated Be(II) cation exposes its empty (sp) hybrid, and is ready to bind whatever Lewis base you provide. Hence, it will bind a Xe atom with a surprisi ...
... adducts of noble gases to a metal center, NgBe(II)O, where Ng = Ar, Kr, and Xe.31 The case of XeBeO is rather straightforward: a coordinatively unsaturated Be(II) cation exposes its empty (sp) hybrid, and is ready to bind whatever Lewis base you provide. Hence, it will bind a Xe atom with a surprisi ...
The reaction pathways of hydrogen peroxide in
... by the reaction with H2O2 at various temperatures. The integrated data have been plotted vs. time which allowed to calculate the individual rate constants of the reaction steps at various temperatures. Based on the rate constants the activation parameters, the ...
... by the reaction with H2O2 at various temperatures. The integrated data have been plotted vs. time which allowed to calculate the individual rate constants of the reaction steps at various temperatures. Based on the rate constants the activation parameters, the ...
P_14 Enhancement of Bioactive Compounds of Roselle Vinegar by
... fermentation. Methods: Vinegar fermentationis a two-step process: First, the anaerobic conversion of sugars to ethanol by Saccharomyces cerevisiae TISTR5048 and then the aerobic oxidation of ethanol to acetic acid by mixed culture of Acetobacter aceti TISTR102 and Acetobacter cervisiae TN4497. Total ...
... fermentation. Methods: Vinegar fermentationis a two-step process: First, the anaerobic conversion of sugars to ethanol by Saccharomyces cerevisiae TISTR5048 and then the aerobic oxidation of ethanol to acetic acid by mixed culture of Acetobacter aceti TISTR102 and Acetobacter cervisiae TN4497. Total ...
The Process of Cellular Respiration
... Citric Acid Production The Krebs cycle begins when pyruvic acid produced by glycolysis passes through the two membranes of the mitochondrion and into the matrix. The matrix is the innermost compartment of the mitochondrion and the site of the Krebs cycle reactions. Once inside the matrix, 1 carbon a ...
... Citric Acid Production The Krebs cycle begins when pyruvic acid produced by glycolysis passes through the two membranes of the mitochondrion and into the matrix. The matrix is the innermost compartment of the mitochondrion and the site of the Krebs cycle reactions. Once inside the matrix, 1 carbon a ...
lecture5
... (FAD), hydration, oxidation by NAD+, and thiolysis by CoA (Figure 22.8). The fatty acyl chain is shortened by two carbon atoms as a result of these reactions, and FADH2, NADH, and acetyl CoA are generated. Because oxidation is on the b carbon, this series of reactions is called the b-oxidation pathw ...
... (FAD), hydration, oxidation by NAD+, and thiolysis by CoA (Figure 22.8). The fatty acyl chain is shortened by two carbon atoms as a result of these reactions, and FADH2, NADH, and acetyl CoA are generated. Because oxidation is on the b carbon, this series of reactions is called the b-oxidation pathw ...
CHAP NUM="9" ID="CH
... tapped to make ATP when the electrons complete their “fall” down an energy gradient from NADH to oxygen. How do electrons that are extracted from glucose and stored as potential energy in NADH finally reach oxygen? It will help to compare the redox chemistry of cellular respiration to a much simpler ...
... tapped to make ATP when the electrons complete their “fall” down an energy gradient from NADH to oxygen. How do electrons that are extracted from glucose and stored as potential energy in NADH finally reach oxygen? It will help to compare the redox chemistry of cellular respiration to a much simpler ...
Human Anatomy and Physiology Preparatory Course
... particular function (for example, organelles called mitochondrion provides energy to a cell). Different types of cells contain different amounts and types of organelles, depending on their function, (for example muscle cells use a lot of energy and therefore have many mitochondria while skin cells d ...
... particular function (for example, organelles called mitochondrion provides energy to a cell). Different types of cells contain different amounts and types of organelles, depending on their function, (for example muscle cells use a lot of energy and therefore have many mitochondria while skin cells d ...
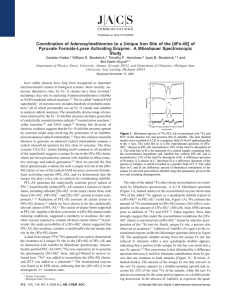
Coordination of Adenosylmethionine to a Unique Iron Site of the
... Iron-sulfur clusters have long been recognized as important electron-transfer centers in biological systems. More recently, numerous alternative roles for Fe-S clusters have been revealed,1 including a key role in catalyzing S-adenosylmethionine (AdoMet or SAM)-mediated radical reactions.2,3 The so- ...
... Iron-sulfur clusters have long been recognized as important electron-transfer centers in biological systems. More recently, numerous alternative roles for Fe-S clusters have been revealed,1 including a key role in catalyzing S-adenosylmethionine (AdoMet or SAM)-mediated radical reactions.2,3 The so- ...
Chemical Equations
... Note, there appear to be more oxygen atoms, fewer hydrogen atoms at the end that at the beginning! ...
... Note, there appear to be more oxygen atoms, fewer hydrogen atoms at the end that at the beginning! ...
Chem 2A Final Review
... 9. The answer closest to the number of grams of NaH2PO4 needed to react with 38.74 mL of 0.275 M NaOH, according to the following balanced equation is: (NaH2PO4 = 119.98 g/mol) NaH2PO4 (s) + 2NaOH (aq) Na3POH4 (aq) + 3H2O ...
... 9. The answer closest to the number of grams of NaH2PO4 needed to react with 38.74 mL of 0.275 M NaOH, according to the following balanced equation is: (NaH2PO4 = 119.98 g/mol) NaH2PO4 (s) + 2NaOH (aq) Na3POH4 (aq) + 3H2O ...
Carbohydrate Metabolism
... When oxygen is limited or absent, pyruvate enters an anaerobic pathway. In these reactions, pyruvate can be converted into lactic acid. ...
... When oxygen is limited or absent, pyruvate enters an anaerobic pathway. In these reactions, pyruvate can be converted into lactic acid. ...
Introduction to Lipid Metabolism Roles of Lipids - Rose
... Lipids have a wide variety of roles in biological systems. These roles are a consequence of their chemical and physical properties. Fatty acids and their derivatives (especially triacylglycerols) can act as highly concentrated energy storage molecules. The high energy density (i.e. the relatively la ...
... Lipids have a wide variety of roles in biological systems. These roles are a consequence of their chemical and physical properties. Fatty acids and their derivatives (especially triacylglycerols) can act as highly concentrated energy storage molecules. The high energy density (i.e. the relatively la ...
Carbohydrate Metabolism
... When oxygen is limited or absent, pyruvate enters an anaerobic pathway. In these reactions, pyruvate can be converted into lactic acid. ...
... When oxygen is limited or absent, pyruvate enters an anaerobic pathway. In these reactions, pyruvate can be converted into lactic acid. ...
chemical bonding and molecular structure
... the inner electrons) and the outer shell that could accommodate a maximum of eight electrons. He, further assumed that these eight electrons occupy the corners of a cube which surround the ‘Kernel’. Thus the single outer shell electron of sodium would occupy one corner of the cube, while in the case ...
... the inner electrons) and the outer shell that could accommodate a maximum of eight electrons. He, further assumed that these eight electrons occupy the corners of a cube which surround the ‘Kernel’. Thus the single outer shell electron of sodium would occupy one corner of the cube, while in the case ...
The Wizard Test Maker
... What is the internal energy change of the gas? (A) –1955. J (D) +1045. J (B) –1803. J (E) +1955. J (C) –1045. J 10. Pressure cookers are used at high altitudes to cook food faster, which of the following statements pertaining to this fact is true? (A) The cooker holds water at a constant pressure at ...
... What is the internal energy change of the gas? (A) –1955. J (D) +1045. J (B) –1803. J (E) +1955. J (C) –1045. J 10. Pressure cookers are used at high altitudes to cook food faster, which of the following statements pertaining to this fact is true? (A) The cooker holds water at a constant pressure at ...
GCE Getting Started - Edexcel
... Understand the interactions in molecules, such as H2O, liquid NH3 and liquid HF, which give rise to hydrogen bonding. Understand the following anomalous properties of water resulting from hydrogen bonding: i. its relatively high melting temperature and boiling temperature ii. the density of ice comp ...
... Understand the interactions in molecules, such as H2O, liquid NH3 and liquid HF, which give rise to hydrogen bonding. Understand the following anomalous properties of water resulting from hydrogen bonding: i. its relatively high melting temperature and boiling temperature ii. the density of ice comp ...
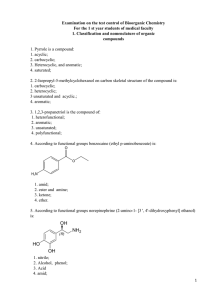
2. 2-Isopropyl-5-methylcyclohexanol on carbon skeletal
... 1. OH- group consisting of carboxylic acid functional groups; 2. hydroxyl group with sp 3-hybrid oxygen; 3. N-H acidic center; 4. OH group bonded directly to the heterocycle; 39. The most strong acidic properties of the compound shown in: 1. acetic acid; 2. propanoic acid; 3. 2-methylpropanoic acid; ...
... 1. OH- group consisting of carboxylic acid functional groups; 2. hydroxyl group with sp 3-hybrid oxygen; 3. N-H acidic center; 4. OH group bonded directly to the heterocycle; 39. The most strong acidic properties of the compound shown in: 1. acetic acid; 2. propanoic acid; 3. 2-methylpropanoic acid; ...
KHOA: HÓA HỌC - CCS - Trường Đại học Sư phạm Hà Nội
... throughout a given sample and from one sample to another. A chemical element is a substance comprised of a single type of atom. The elements are the building blocks of our nature. An element is either discovered in nature or synthesized in the laboratory in pure form that cannot be separated into s ...
... throughout a given sample and from one sample to another. A chemical element is a substance comprised of a single type of atom. The elements are the building blocks of our nature. An element is either discovered in nature or synthesized in the laboratory in pure form that cannot be separated into s ...
Chemistry - Tumkur University
... ionic size, ionization enthalpy, electronegativity (Pauling, Mulliken, and Alfred-Rochow scales).Allotropy in C, S, and P. Inert pair effect, diagonal relationship and anomalous behaviour of first member of each group. (3 Lectures) Atomic Structure: Review of: Bohr’s theory and its limitations, dual ...
... ionic size, ionization enthalpy, electronegativity (Pauling, Mulliken, and Alfred-Rochow scales).Allotropy in C, S, and P. Inert pair effect, diagonal relationship and anomalous behaviour of first member of each group. (3 Lectures) Atomic Structure: Review of: Bohr’s theory and its limitations, dual ...
Radical (chemistry)

In chemistry, a radical (more precisely, a free radical) is an atom, molecule, or ion that has unpaired valency electrons.With some exceptions, these unpaired electrons make free radicals highly chemically reactive towards other substances, or even towards themselves: their molecules will often spontaneously dimerize or polymerize if they come in contact with each other. Most radicals are reasonably stable only at very low concentrations in inert media or in a vacuum.A notable example of a free radical is the hydroxyl radical (HO•), a molecule that has one unpaired electron on the oxygen atom. Two other examples are triplet oxygen and triplet carbene (:CH2) which have two unpaired electrons. In contrast, the hydroxyl anion (HO−) is not a radical, since the unpaired electron is resolved by the addition of an electron; singlet oxygen and singlet carbene are not radicals as the two electrons are paired.Free radicals may be created in a number of ways, including synthesis with very dilute or rarefied reagents, reactions at very low temperatures, or breakup of larger molecules. The latter can be affected by any process that puts enough energy into the parent molecule, such as ionizing radiation, heat, electrical discharges, electrolysis, and chemical reactions. Indeed, radicals are intermediate stages in many chemical reactions.Free radicals play an important role in combustion, atmospheric chemistry, polymerization, plasma chemistry, biochemistry, and many other chemical processes. In living organisms, the free radicals superoxide and nitric oxide and their reaction products regulate many processes, such as control of vascular tone and thus blood pressure. They also play a key role in the intermediary metabolism of various biological compounds. Such radicals can even be messengers in a process dubbed redox signaling. A radical may be trapped within a solvent cage or be otherwise bound.Until late in the 20th century the word ""radical"" was used in chemistry to indicate any connected group of atoms, such as a methyl group or a carboxyl, whether it was part of a larger molecule or a molecule on its own. The qualifier ""free"" was then needed to specify the unbound case. Following recent nomenclature revisions, a part of a larger molecule is now called a functional group or substituent, and ""radical"" now implies ""free"". However, the old nomenclature may still occur in the literature.