ALA - Alpha Lipoic Acid Lipolate; Thiotic acid.
... Reactive oxygen species (ROS) and reactive nitrogen species (RNS) are highly reactive compounds with the potential to damage DNA, www.healthoracle.org ...
... Reactive oxygen species (ROS) and reactive nitrogen species (RNS) are highly reactive compounds with the potential to damage DNA, www.healthoracle.org ...
1 Cellular Respiration: Harvesting Chemical Energy Introduction
... glycolysis can continue in absence of oxygen • Glycolysis can produce ATP with or without oxygen, in aerobic or anaerobic conditions – couples with fermentation to produce ATP ...
... glycolysis can continue in absence of oxygen • Glycolysis can produce ATP with or without oxygen, in aerobic or anaerobic conditions – couples with fermentation to produce ATP ...
Hyaluronic Acid in Dermatology and Dermocosmetics
... FREE RADICALS An important role known as “oxidation stress” is played by the increased production of free radicals in the ageing process. When the cells in our body produce energy, unstable oxygenated molecules are formed as a by-product of normal cell function. These substances, known as free radi ...
... FREE RADICALS An important role known as “oxidation stress” is played by the increased production of free radicals in the ageing process. When the cells in our body produce energy, unstable oxygenated molecules are formed as a by-product of normal cell function. These substances, known as free radi ...
Answer Key
... Be able to give examples where each of these is used. Alcohol – beer, wine, bread Lactic acid – muscles, kimchi, saurerkraut What molecule is burned to provide quick energy during exercise? ATP Why do cells use fermentation? (Hint: It’s NOT to make alcohol or lactic acid) To produce produce NAD+ for ...
... Be able to give examples where each of these is used. Alcohol – beer, wine, bread Lactic acid – muscles, kimchi, saurerkraut What molecule is burned to provide quick energy during exercise? ATP Why do cells use fermentation? (Hint: It’s NOT to make alcohol or lactic acid) To produce produce NAD+ for ...
ATP Molecules
... • Per glucose molecule, ten NADH and two FADH2 take electrons to the electron transport system; three ATP are formed per NADH and two ATP per FADH2. • Electrons carried by NADH produced during glycolysis are shuttled to the electron transport chain by an organic molecule. ...
... • Per glucose molecule, ten NADH and two FADH2 take electrons to the electron transport system; three ATP are formed per NADH and two ATP per FADH2. • Electrons carried by NADH produced during glycolysis are shuttled to the electron transport chain by an organic molecule. ...
FLEMINGIA STROBILIFERA R.B Research Article
... autoimmune disorders. Most of the hepatotoxic chemicals damage liver cells mainly by inducing lipid peroxidation and other oxidative damages2. Hepatotoxicity is one of very common aliment resulting into serious debilities ranging from severe metabolic disorders to even mo ...
... autoimmune disorders. Most of the hepatotoxic chemicals damage liver cells mainly by inducing lipid peroxidation and other oxidative damages2. Hepatotoxicity is one of very common aliment resulting into serious debilities ranging from severe metabolic disorders to even mo ...
basic chemistry of atoms and molecules
... Carbohydrate polymers are built from monomers of simple sugars such as glucose. Plants use glucose molecules to build the polymer starch. The figure below illustrates the long spiral chain of glucose molecules that form starch (each green hexagon is an individual glucose molecule). Only plants produ ...
... Carbohydrate polymers are built from monomers of simple sugars such as glucose. Plants use glucose molecules to build the polymer starch. The figure below illustrates the long spiral chain of glucose molecules that form starch (each green hexagon is an individual glucose molecule). Only plants produ ...
chemical reactions
... This is an introduction to chemical reactions. The goal is to demonstrate chemical reactions, reinforce formula writing, introduce students to writing and balancing chemical equations, and to present the reasons why chemical reactions go to completion. This can be reinforced by microscale or small s ...
... This is an introduction to chemical reactions. The goal is to demonstrate chemical reactions, reinforce formula writing, introduce students to writing and balancing chemical equations, and to present the reasons why chemical reactions go to completion. This can be reinforced by microscale or small s ...
how cells obtain energy from food
... energy, part of which must be released from the cells to their environment as heat that disorders the surroundings. The only chemical reactions possible are those that increase the total amount of disorder in the universe. The free-energy change for a reaction, ∆G, measures this disorder, and it mus ...
... energy, part of which must be released from the cells to their environment as heat that disorders the surroundings. The only chemical reactions possible are those that increase the total amount of disorder in the universe. The free-energy change for a reaction, ∆G, measures this disorder, and it mus ...
Unit 3: Bonding and Nomenclature Content Outline: Chemical
... A. The natural tendency is to achieve the lowest possible Potential Energy state and thus behave “like” a Noble gas element. B. Energy is released in bond formation between atoms. C. Energy is required in the breaking of a bond between atoms. 1. The energy to make or break a bond is referred to as b ...
... A. The natural tendency is to achieve the lowest possible Potential Energy state and thus behave “like” a Noble gas element. B. Energy is released in bond formation between atoms. C. Energy is required in the breaking of a bond between atoms. 1. The energy to make or break a bond is referred to as b ...
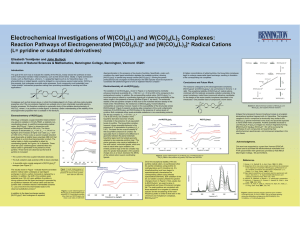
Electrochemical Investigations of W(CO) (L) and W(CO) (L) Complexes:
... tetracarbonyl pyridine fragment with 4,4'-bipyridine. The tungsten complex in such a compound is structurally very similar to the W(CO)4(L)2 species examined in this study and would be expected to have similar redox properties. Another possibility would be to use W(CO)4(4,4'-bpy)2 to bridges two rhe ...
... tetracarbonyl pyridine fragment with 4,4'-bipyridine. The tungsten complex in such a compound is structurally very similar to the W(CO)4(L)2 species examined in this study and would be expected to have similar redox properties. Another possibility would be to use W(CO)4(4,4'-bpy)2 to bridges two rhe ...
Focus on Metabolism
... Glycolysis: Anaerobic Metabolism The first stage of cellular respiration takes place in the cytosol of the cell and is called glycolysis, meaning “glucose breakdown.” Because oxygen isn’t needed for this reaction, glycolysis is also called anaerobic metabolism. In glycolysis, the 6-carbon sugar gluc ...
... Glycolysis: Anaerobic Metabolism The first stage of cellular respiration takes place in the cytosol of the cell and is called glycolysis, meaning “glucose breakdown.” Because oxygen isn’t needed for this reaction, glycolysis is also called anaerobic metabolism. In glycolysis, the 6-carbon sugar gluc ...
lecture_22 - WordPress.com
... Acyl-CoA dehydrogenase: an enzyme similar to succinate dehydrogenase ...
... Acyl-CoA dehydrogenase: an enzyme similar to succinate dehydrogenase ...
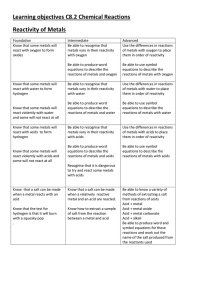
Learning objectives C8.2 Chemical Reactions Reactivity of Metals
... methods of extracting a salt from reactions of acids Acid + metal Acid + metal oxide Acid + metal carbonate Acid + alkali Be able to produce word and symbol equations for these reactions and work out the name of the salt produced from the reactants used ...
... methods of extracting a salt from reactions of acids Acid + metal Acid + metal oxide Acid + metal carbonate Acid + alkali Be able to produce word and symbol equations for these reactions and work out the name of the salt produced from the reactants used ...
Lecture 7. Fundamentals of atmospheric chemistry: Part 2 1
... electron. Keep in mind it's the charge that's being reduced in this case. Oxidation receives its name because almost all reactions with oxygen involve some other element losing electrons to the oxygen. Only fluorine would cause oxygen to formally lose electrons. When the term "oxidation" came into u ...
... electron. Keep in mind it's the charge that's being reduced in this case. Oxidation receives its name because almost all reactions with oxygen involve some other element losing electrons to the oxygen. Only fluorine would cause oxygen to formally lose electrons. When the term "oxidation" came into u ...
Cell Respiration - Biology Junction
... 3) turns twice because two acetyl-CoA molecules enter the cycle per glucose molecule; 4) produces two immediate ATP molecules per glucose molecule. d. The electron transport chain: 1) is a series of carriers in the inner mitochondrial membrane that accept electrons from glucose--electrons are passed ...
... 3) turns twice because two acetyl-CoA molecules enter the cycle per glucose molecule; 4) produces two immediate ATP molecules per glucose molecule. d. The electron transport chain: 1) is a series of carriers in the inner mitochondrial membrane that accept electrons from glucose--electrons are passed ...
Chapter 7 Lecture
... Writing Chemical Equations for Reactions in Solution: Molecular and Complete Ionic Equations • A molecular equation is an equation showing the complete neutral formulas for every compound in the reaction. • Complete ionic equations show aqueous ionic compounds that normally dissociate in solution a ...
... Writing Chemical Equations for Reactions in Solution: Molecular and Complete Ionic Equations • A molecular equation is an equation showing the complete neutral formulas for every compound in the reaction. • Complete ionic equations show aqueous ionic compounds that normally dissociate in solution a ...
CH 4: Chemical Reactions
... usually has the same oxidation number it would have if it were a monatomic ion. A. Hydrogen can be either +1 or –1. B. Oxygen usually has an oxidation number of –2. In peroxides, oxygen is –1. C. Halogens usually have an oxidation number of –1. • When bonded to oxygen, chlorine, bromine, and iodine ...
... usually has the same oxidation number it would have if it were a monatomic ion. A. Hydrogen can be either +1 or –1. B. Oxygen usually has an oxidation number of –2. In peroxides, oxygen is –1. C. Halogens usually have an oxidation number of –1. • When bonded to oxygen, chlorine, bromine, and iodine ...
Chapter 8 - South Sevier High School
... 3) turns twice because two acetyl-CoA molecules enter the cycle per glucose molecule; 4) produces two immediate ATP molecules per glucose molecule. d. The electron transport chain: 1) is a series of carriers in the inner mitochondrial membrane that accept electrons from glucose--electrons are passed ...
... 3) turns twice because two acetyl-CoA molecules enter the cycle per glucose molecule; 4) produces two immediate ATP molecules per glucose molecule. d. The electron transport chain: 1) is a series of carriers in the inner mitochondrial membrane that accept electrons from glucose--electrons are passed ...
handout 4
... 3.6 Determining the Formulas of Compounds A. Determining formulas from data: 1. Determining formulas from mass % (we did this already) Try this example on your own to review: During physical activity, lactic acid (molar mass = 90.08 g/mol) forms in muscles and is responsible for muscle soreness. Ele ...
... 3.6 Determining the Formulas of Compounds A. Determining formulas from data: 1. Determining formulas from mass % (we did this already) Try this example on your own to review: During physical activity, lactic acid (molar mass = 90.08 g/mol) forms in muscles and is responsible for muscle soreness. Ele ...
CHEMICAL REACTIONS
... • e.g. Br2 + CH2=CH2 Æ CH2Br-CH2Br • e.g. H2 + CH2=CH2 Æ CH3-CH3 2. Substitution reactions Occur when an atom attached to carbon is replaced by something else. • e.g. Cl2 + CH4 Æ CH3Cl + HCl 3. Combustion Reactions When an organic compound is oxidized. Complete combustion results in carbon dioxide a ...
... • e.g. Br2 + CH2=CH2 Æ CH2Br-CH2Br • e.g. H2 + CH2=CH2 Æ CH3-CH3 2. Substitution reactions Occur when an atom attached to carbon is replaced by something else. • e.g. Cl2 + CH4 Æ CH3Cl + HCl 3. Combustion Reactions When an organic compound is oxidized. Complete combustion results in carbon dioxide a ...
Cellular Respiration
... Sometimes energy is required to transport NADH + H+ formed by glycolysis from the cytoplasm through the inner mitochondrial membrane. Some H+ in chemiosmosis may leak through the membrane. • Aerobic Respiration is generally 19 times more efficient than anaerobic respiration. • The ATP produced d ...
... Sometimes energy is required to transport NADH + H+ formed by glycolysis from the cytoplasm through the inner mitochondrial membrane. Some H+ in chemiosmosis may leak through the membrane. • Aerobic Respiration is generally 19 times more efficient than anaerobic respiration. • The ATP produced d ...
C9 Cellular Respiration (Video)
... 3. Coenzyme A is attached to the acetate by unstable bond that makes the acetyl group very reactive. (Now acetyl CoA). Krebs cycle – Hans Krebs, 1930’s. 8 steps, each catalyzed by a specific enzyme in the mitochondrial matrix. CO2 given off in steps 3 and 4. Most energy conserved as NADH. For each a ...
... 3. Coenzyme A is attached to the acetate by unstable bond that makes the acetyl group very reactive. (Now acetyl CoA). Krebs cycle – Hans Krebs, 1930’s. 8 steps, each catalyzed by a specific enzyme in the mitochondrial matrix. CO2 given off in steps 3 and 4. Most energy conserved as NADH. For each a ...
Old Photo Respiration test
... a. It was released as CO2 and H2O. b. Chemical energy was converted to heat and then released. c. It was converted to ATP, which weighs much less than fat. d. It was broken down to amino acids and eliminated from the body. e. It was converted to urine and eliminated from the body. ____ 28. Organisms ...
... a. It was released as CO2 and H2O. b. Chemical energy was converted to heat and then released. c. It was converted to ATP, which weighs much less than fat. d. It was broken down to amino acids and eliminated from the body. e. It was converted to urine and eliminated from the body. ____ 28. Organisms ...
Radical (chemistry)

In chemistry, a radical (more precisely, a free radical) is an atom, molecule, or ion that has unpaired valency electrons.With some exceptions, these unpaired electrons make free radicals highly chemically reactive towards other substances, or even towards themselves: their molecules will often spontaneously dimerize or polymerize if they come in contact with each other. Most radicals are reasonably stable only at very low concentrations in inert media or in a vacuum.A notable example of a free radical is the hydroxyl radical (HO•), a molecule that has one unpaired electron on the oxygen atom. Two other examples are triplet oxygen and triplet carbene (:CH2) which have two unpaired electrons. In contrast, the hydroxyl anion (HO−) is not a radical, since the unpaired electron is resolved by the addition of an electron; singlet oxygen and singlet carbene are not radicals as the two electrons are paired.Free radicals may be created in a number of ways, including synthesis with very dilute or rarefied reagents, reactions at very low temperatures, or breakup of larger molecules. The latter can be affected by any process that puts enough energy into the parent molecule, such as ionizing radiation, heat, electrical discharges, electrolysis, and chemical reactions. Indeed, radicals are intermediate stages in many chemical reactions.Free radicals play an important role in combustion, atmospheric chemistry, polymerization, plasma chemistry, biochemistry, and many other chemical processes. In living organisms, the free radicals superoxide and nitric oxide and their reaction products regulate many processes, such as control of vascular tone and thus blood pressure. They also play a key role in the intermediary metabolism of various biological compounds. Such radicals can even be messengers in a process dubbed redox signaling. A radical may be trapped within a solvent cage or be otherwise bound.Until late in the 20th century the word ""radical"" was used in chemistry to indicate any connected group of atoms, such as a methyl group or a carboxyl, whether it was part of a larger molecule or a molecule on its own. The qualifier ""free"" was then needed to specify the unbound case. Following recent nomenclature revisions, a part of a larger molecule is now called a functional group or substituent, and ""radical"" now implies ""free"". However, the old nomenclature may still occur in the literature.