ppt
... total mass of the reactants is equal to the total mass of the products. Atoms are not created nor destroyed. ...
... total mass of the reactants is equal to the total mass of the products. Atoms are not created nor destroyed. ...
CLASS NOTES- Balancing Chemical Equations.pptx
... balance the equation 4. Check your answer to see if: • The numbers of atoms on both sides of the equation are now balanced • The coefficients are in the lowest possible whole number ratios. (reduced) ...
... balance the equation 4. Check your answer to see if: • The numbers of atoms on both sides of the equation are now balanced • The coefficients are in the lowest possible whole number ratios. (reduced) ...
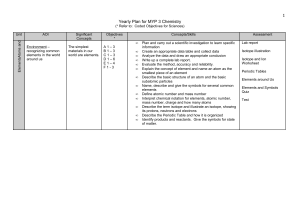
Yearly Plan for MYP 1 Science
... without them - recognizing common chemical reactions in our world - understanding what happens in a chemical change - noticing and identifying common chemicals we use in our everyday lives ...
... without them - recognizing common chemical reactions in our world - understanding what happens in a chemical change - noticing and identifying common chemicals we use in our everyday lives ...
File
... Physical or chemical change? The rain turned to snow… Marty broke a class on the bathroom floor… I burned my bagel! I fried eggs for breakfast… I mixed baking soda and vinegar for science ...
... Physical or chemical change? The rain turned to snow… Marty broke a class on the bathroom floor… I burned my bagel! I fried eggs for breakfast… I mixed baking soda and vinegar for science ...
Reactions Unit Plan
... ____3. Each substance from the reaction above should have the following states indicated as: a. (s), (l) (g), (aq) ...
... ____3. Each substance from the reaction above should have the following states indicated as: a. (s), (l) (g), (aq) ...
Elements, Compounds and Chemical Reactions
... have both PHYSICAL and CHEMICAL properties. A property that can be observed, measured or changed without changing the substance itself is called a physical property. ...
... have both PHYSICAL and CHEMICAL properties. A property that can be observed, measured or changed without changing the substance itself is called a physical property. ...
Document
... WHY ARE THERE CHEMICAL REACTIONS? CHEMICAL REACTIONS HAPPEN WHEN MOLECULES BUMP INTO EACH OTHER CAUSING THE STARTING BONDS TO BREAK APART, THE ATOMS REARRANGE, AND NEW BONDS ARE FORMED ...
... WHY ARE THERE CHEMICAL REACTIONS? CHEMICAL REACTIONS HAPPEN WHEN MOLECULES BUMP INTO EACH OTHER CAUSING THE STARTING BONDS TO BREAK APART, THE ATOMS REARRANGE, AND NEW BONDS ARE FORMED ...
Bacteria and Virus Research Jigsaw
... WHY ARE THERE CHEMICAL REACTIONS? CHEMICAL REACTIONS HAPPEN WHEN MOLECULES BUMP INTO EACH OTHER CAUSING THE STARTING BONDS TO BREAK APART, THE ATOMS REARRANGE, AND NEW BONDS ARE FORMED ...
... WHY ARE THERE CHEMICAL REACTIONS? CHEMICAL REACTIONS HAPPEN WHEN MOLECULES BUMP INTO EACH OTHER CAUSING THE STARTING BONDS TO BREAK APART, THE ATOMS REARRANGE, AND NEW BONDS ARE FORMED ...
Chapter 14 Chemical Reactions
... In chemical reactions, you start with reactants that are combined to make products. The reactants are the starting substances. The products are the new substances which result from the chemical reaction. ...
... In chemical reactions, you start with reactants that are combined to make products. The reactants are the starting substances. The products are the new substances which result from the chemical reaction. ...
Name___________________________________ Physical
... CHEMICAL REACTIONS - P. 582 12) The process in which the atoms of one or more substance are rearranged to form different substances is called a(n) ____________________. A) nuclear reaction B) substantiation C) chemical reaction D) physical reaction ...
... CHEMICAL REACTIONS - P. 582 12) The process in which the atoms of one or more substance are rearranged to form different substances is called a(n) ____________________. A) nuclear reaction B) substantiation C) chemical reaction D) physical reaction ...
Notes for Types of Reactions:
... chemical reaction = the _________ by which one or more substances are __________ into one or more _________ substances. in any chemical reaction, the _________ substances are known as the reactants and the __________ substances are known as the products. total mass of reactants = according to ...
... chemical reaction = the _________ by which one or more substances are __________ into one or more _________ substances. in any chemical reaction, the _________ substances are known as the reactants and the __________ substances are known as the products. total mass of reactants = according to ...
Matter and Energy
... -determines the number of molecules (groups) of the formula -This number will be DISTRIBUTED just like in math. It applies to each element and is multiplied by each subscript to find the total number of atoms of each element and a total number of atoms in the molecule. ...
... -determines the number of molecules (groups) of the formula -This number will be DISTRIBUTED just like in math. It applies to each element and is multiplied by each subscript to find the total number of atoms of each element and a total number of atoms in the molecule. ...
Chapter 14, Section 1, pages 494-501
... Burn sulfur in oxygen as an example of a completion reaction. Input Completion Reactions and Reversible Reactions What does reversible mean? Completion Reactions are reactions that use up all or almost all of the reactants to form products S8 + 8O2 ----------->8 SO2 Reversible Reactions are those in ...
... Burn sulfur in oxygen as an example of a completion reaction. Input Completion Reactions and Reversible Reactions What does reversible mean? Completion Reactions are reactions that use up all or almost all of the reactants to form products S8 + 8O2 ----------->8 SO2 Reversible Reactions are those in ...
Chemical Reactions
... Chemical Reactions Everything that happens in an organism is based on chemical reactions. A chemical reaction is a process that changes one set of chemicals into another set of chemicals. The elements or compounds that enter into the reaction are the reactants. The elements or compounds produced by ...
... Chemical Reactions Everything that happens in an organism is based on chemical reactions. A chemical reaction is a process that changes one set of chemicals into another set of chemicals. The elements or compounds that enter into the reaction are the reactants. The elements or compounds produced by ...
Faith groups call for chemical policy reform for Father`s
... the foundation of sustainable economies, and that “dharma” – often translated “duty” - can be interpreted to support respect for Earth. Gandhi emphasized the Hindu teaching of “ahimsa,” or nonviolence towards the web of life. The ancient Indic tradition of Jainism declares non-violence as its suprem ...
... the foundation of sustainable economies, and that “dharma” – often translated “duty” - can be interpreted to support respect for Earth. Gandhi emphasized the Hindu teaching of “ahimsa,” or nonviolence towards the web of life. The ancient Indic tradition of Jainism declares non-violence as its suprem ...
3_2: More Chemical Changes
... Investigate: Chemical Reactions • In today’s lab, you will be looking at chemical reactions that occur between 8 different solid materials. The solids have been dissolved in water to make solutions. ...
... Investigate: Chemical Reactions • In today’s lab, you will be looking at chemical reactions that occur between 8 different solid materials. The solids have been dissolved in water to make solutions. ...
Chemical Equations and Tests for anions
... Law of Conservation of Matter In any chemical reaction matter is neither created nor destroyed but merely changes from one form to another If there is a particular number of atoms at the start of a reaction then there must be the same number of atoms at the end of the reaction ...
... Law of Conservation of Matter In any chemical reaction matter is neither created nor destroyed but merely changes from one form to another If there is a particular number of atoms at the start of a reaction then there must be the same number of atoms at the end of the reaction ...
Chemical Protection: In and Out of the Lab
... gloves and sleeve protectors may provide reasonable protection. The safety professional might even conservatively recommend a chemical apron but not require a full coverall in this particular application. In fact, two different lab safety professionals may make different recommendations on the same ...
... gloves and sleeve protectors may provide reasonable protection. The safety professional might even conservatively recommend a chemical apron but not require a full coverall in this particular application. In fact, two different lab safety professionals may make different recommendations on the same ...
CHEMISTry is life - World of Teaching
... -Too often kids get to high school chemistry and they are scared before they even begin. -My goal is to shape a positive image in their minds about chemistry so that they can be more prepared mentally for high school. -I will do this by showing them how applicable chemistry is to every day life. It ...
... -Too often kids get to high school chemistry and they are scared before they even begin. -My goal is to shape a positive image in their minds about chemistry so that they can be more prepared mentally for high school. -I will do this by showing them how applicable chemistry is to every day life. It ...
Biotechnology and the manufacturing industry
... Most microbes employed are usually isolated from nature and then modified using classical mutation and/or selection procedures. Analysis of the microbial cell composition have revealed that over 90% of the cells dry weight is made up of both micro and macro elements which includes C, O, N, H, S, K, ...
... Most microbes employed are usually isolated from nature and then modified using classical mutation and/or selection procedures. Analysis of the microbial cell composition have revealed that over 90% of the cells dry weight is made up of both micro and macro elements which includes C, O, N, H, S, K, ...
Introduction to Chemistry
... attract together (+ is attracted to - after an electron is transferred) ...
... attract together (+ is attracted to - after an electron is transferred) ...
Balancing chemical equations notes
... Chemical equations can be viewed as recipes for chemical reactions. They give a description of what chemicals are combined together and what chemicals are made when a reaction occurs. The law of conservation of mass says that matter can neither be created nor destroyed, and this requires that all ch ...
... Chemical equations can be viewed as recipes for chemical reactions. They give a description of what chemicals are combined together and what chemicals are made when a reaction occurs. The law of conservation of mass says that matter can neither be created nor destroyed, and this requires that all ch ...
Fine chemical

Fine chemicals are complex, single, pure chemical substances, produced in limited quantities in multipurpose plants by multistep batch chemical or biotechnological processes. They are described by exacting specifications, used for further processing within the chemical industry and sold for more than $10/kg (see the comparison of fine chemicals, commodities and specialties). The class of fine chemicals is subdivided either on the basis of the added value (building blocks, advanced intermediates or active ingredients), or the type of business transaction, namely standard or exclusive products.Fine chemicals are produced in limited volumes (< 1000 tons/year) and at relatively high prices (> $10/kg) according to exacting specifications, mainly by traditional organic synthesis in multipurpose chemical plants. Biotechnical processes are gaining ground. The global production value is about $85 billion. Fine chemicals are used as starting materials for specialty chemicals, particularly pharmaceuticals, biopharmaceuticals and agrochemicals. Custom manufacturing for the life science industry plays a big role; however, a significant portion of the fine chemicals total production volume is manufactured in house by large users. The industry is fragmented and extends from small, privately owned companies to divisions of big, diversified chemical enterprises. The term ""fine chemicals"" is used in distinction to ""heavy chemicals"", which are produced and handled in large lots and are often in a crude state.Since their inception in the late 1970s, fine chemicals have become an important part of the chemical industry. The total production value of $85 billion is split about 60 / 40 among in-house production by the main consumers, the life science industry, on the one hand, and the fine chemicals industry on the other hand. The latter pursues both a “supply push” strategy, whereby standard products are developed in-house and offered ubiquitously, and a “demand pull” strategy, whereby products or services determined by the customer are provided exclusively on a “one customer / one supplier” basis. The products are mainly used as building blocks for proprietary products. The hardware of the top tier fine chemical companies has become almost identical. The design, lay-out and equipment of the plants and laboratories has become practically the same all over the world. Most chemical reactions performed go back to the days of the dyestuff industry. Numerous regulations determine the way labs and plants have to be operated, thereby contributing to the uniformity.