Critical Point Dryer
... based on the sequential self terminating gas-solid reactions to give thin films of nanometer range. ...
... based on the sequential self terminating gas-solid reactions to give thin films of nanometer range. ...
Unit 5 Student Packet
... change from 25.246C to 26.386C. If the heat capacity of the calorimeter was 7.15 kJ/C, calculate H comb for octane. 3. Calcium oxide (lime) reacts with water to give calcium hydroxide. A 5.40 g sample of calcium oxide was added to 500. mL of water in a calorimeter of heat capacity 350. J/C. The ...
... change from 25.246C to 26.386C. If the heat capacity of the calorimeter was 7.15 kJ/C, calculate H comb for octane. 3. Calcium oxide (lime) reacts with water to give calcium hydroxide. A 5.40 g sample of calcium oxide was added to 500. mL of water in a calorimeter of heat capacity 350. J/C. The ...
The Mole - Rothschild Science
... One formula unit of calcium carbonate reacts to form one formula unit of calcium oxide and one molecule of carbon dioxide. One mole of calcium carbonate reacts to form one mole of calcium oxide and one mole of carbon dioxide. ...
... One formula unit of calcium carbonate reacts to form one formula unit of calcium oxide and one molecule of carbon dioxide. One mole of calcium carbonate reacts to form one mole of calcium oxide and one mole of carbon dioxide. ...
Honors Chemistry Unit 4 Student Packet: Honors Chemistry Problem
... 1. The coefficients in a balanced chemical equation can be used to form mole ratios relating reactants to products. 2. To convert molecules to grams you can either use molar mass or Avogadro’s number. 3. One mole of any gas occupies a volume of 22.4L. 4. Mass and the number of moles are conserved in ...
... 1. The coefficients in a balanced chemical equation can be used to form mole ratios relating reactants to products. 2. To convert molecules to grams you can either use molar mass or Avogadro’s number. 3. One mole of any gas occupies a volume of 22.4L. 4. Mass and the number of moles are conserved in ...
full text pdf
... flue gas from the coal combustion without the desulfurization16. The temperature in the reaction chamber was maintained at the level of 300°C during whole experiment. The gas flow was controlled by dual regulation system: the controller coupled with flow meter and volumetric flow controller. The SO2 ...
... flue gas from the coal combustion without the desulfurization16. The temperature in the reaction chamber was maintained at the level of 300°C during whole experiment. The gas flow was controlled by dual regulation system: the controller coupled with flow meter and volumetric flow controller. The SO2 ...
Lecture-3
... The CH4 is at 25°C and 1 atm, and thus its absolute entropy is s– for CH4 =186.16 kJ/kmol K (Table A– 26). The entropy values listed in the ideal-gas tables are for 1 atm pressure. Both the air and the product gases are at a total pressure of 1 atm, but the entropies are to be calculated at the part ...
... The CH4 is at 25°C and 1 atm, and thus its absolute entropy is s– for CH4 =186.16 kJ/kmol K (Table A– 26). The entropy values listed in the ideal-gas tables are for 1 atm pressure. Both the air and the product gases are at a total pressure of 1 atm, but the entropies are to be calculated at the part ...
the pdf of this lesson!
... Because calcium can hold 2 of the alginate chains, or 2 parts of one long chain, it is less soluble in water than sodium alginate. That is why it comes out of solution. It starts to clump. As it does this, it traps water from the solution. The Calcium Connection You may have noticed that the wormy t ...
... Because calcium can hold 2 of the alginate chains, or 2 parts of one long chain, it is less soluble in water than sodium alginate. That is why it comes out of solution. It starts to clump. As it does this, it traps water from the solution. The Calcium Connection You may have noticed that the wormy t ...
Part I - American Chemical Society
... There is only one correct answer to each question. Any questions for which more than one response has been blackened will not be counted. Your score is based solely on the number of questions you answer correctly. It is to your advantage to answer every question. 1. Which absorbs gaseous carbon ...
... There is only one correct answer to each question. Any questions for which more than one response has been blackened will not be counted. Your score is based solely on the number of questions you answer correctly. It is to your advantage to answer every question. 1. Which absorbs gaseous carbon ...
2006 Practice Final Exam - Department of Chemistry | Oregon State
... calculator, and your University ID Card. If you have notes with you, place them in a sealed backpack and place the backpack OUT OF SIGHT or place the notes directly on the table at the front of the room. Fill in the front page of the Scantron answer sheet with your test form number (listed above), l ...
... calculator, and your University ID Card. If you have notes with you, place them in a sealed backpack and place the backpack OUT OF SIGHT or place the notes directly on the table at the front of the room. Fill in the front page of the Scantron answer sheet with your test form number (listed above), l ...
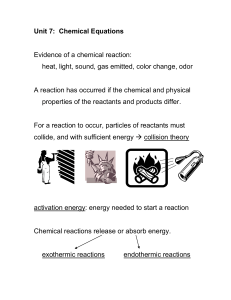
Unit 9 Chemical Equations and Reactions Balancing Equations Notes
... Synthesis- _____________________ elements or compounds combine to form one compound. Decomposition- a _________________ compound decomposes into two or more elements or smaller compounds. Single Replacement- a metal will _________________ a less active metal in an ionic compound OR a nonmetal will r ...
... Synthesis- _____________________ elements or compounds combine to form one compound. Decomposition- a _________________ compound decomposes into two or more elements or smaller compounds. Single Replacement- a metal will _________________ a less active metal in an ionic compound OR a nonmetal will r ...
Answers to Homework Problem Sheet 8
... After the reactants mix the solution has a volume of 200 mL. Assuming the solution the same density as pure water, this corresponds to a mass of: mass (g) = density (g mL-1) × volume (mL) = (0.997 g mL-1) × (200 mL) = 199 g The temperature change, ΔT = (31.1 °C – 24.6 °C) = 6.7 K. The heat capacity ...
... After the reactants mix the solution has a volume of 200 mL. Assuming the solution the same density as pure water, this corresponds to a mass of: mass (g) = density (g mL-1) × volume (mL) = (0.997 g mL-1) × (200 mL) = 199 g The temperature change, ΔT = (31.1 °C – 24.6 °C) = 6.7 K. The heat capacity ...
Name: Per: Date: Unit 1. Materials: Formulating Matter B. Periodic
... More than 100 chemical elements have been discovered and explored. Of those, a small number are considered highly prized and used for coins, jewelry, and art. Why are some more valuable than others? How do scientists classify and organize the characteristics of the elements? In this section, you wil ...
... More than 100 chemical elements have been discovered and explored. Of those, a small number are considered highly prized and used for coins, jewelry, and art. Why are some more valuable than others? How do scientists classify and organize the characteristics of the elements? In this section, you wil ...
Chapter 8 - Inorganic carbon chemistry
... using calcium carbonate, sodium chloride, carbon dioxide and ammonia in a continuous process, with the Solvay tower the centre of reactions. Sodium carbonate is made as economically as possible because both the carbon dioxide and the ammonia components are recycled continuously. ...
... using calcium carbonate, sodium chloride, carbon dioxide and ammonia in a continuous process, with the Solvay tower the centre of reactions. Sodium carbonate is made as economically as possible because both the carbon dioxide and the ammonia components are recycled continuously. ...
MINDS-ON 3: Fuel Combustion
... Explain that in an internal combustion engine, when the fuel is ignited, a large amount of energy is released as gases (CO2 and H2O) are formed. These gases are able to exert force against a piston, thus transforming the chemical energy of fuel into mechanical energy. ...
... Explain that in an internal combustion engine, when the fuel is ignited, a large amount of energy is released as gases (CO2 and H2O) are formed. These gases are able to exert force against a piston, thus transforming the chemical energy of fuel into mechanical energy. ...
Thermodynamics
... From knowing the value of ∆G, you may predict whether a process/reaction will be spontaneous at a certain temperature. A process is spontaneous if ∆G is negative (∆G < 0), nonspontaneous if ∆G is positive (∆G > 0), and at equilibrium if ∆G = 0. The enthalpy change, ∆Hrxn, is the heat gained or lost ...
... From knowing the value of ∆G, you may predict whether a process/reaction will be spontaneous at a certain temperature. A process is spontaneous if ∆G is negative (∆G < 0), nonspontaneous if ∆G is positive (∆G > 0), and at equilibrium if ∆G = 0. The enthalpy change, ∆Hrxn, is the heat gained or lost ...
PPT: Chemical Reactions and Equations
... Predict if a reaction will occur when you combine aqueous solutions of iron (II) chloride and sodium carbonate… If the reaction does occur, write a Balanced ...
... Predict if a reaction will occur when you combine aqueous solutions of iron (II) chloride and sodium carbonate… If the reaction does occur, write a Balanced ...
Higher Chemistry summary 3a
... suitable for fast single step processes more easily automated using computer control smaller work force operates round the clock, 365 days per year tend to operate with relatively low volumes of reactants allowing easy removal of excess heat energy ...
... suitable for fast single step processes more easily automated using computer control smaller work force operates round the clock, 365 days per year tend to operate with relatively low volumes of reactants allowing easy removal of excess heat energy ...
- E3S Web of Conferences
... drop in carbon dioxide content from 16% (for the gasification of RDF fuel only) to a very low level of 1÷2.5% when Bio-CONOx was added. In addition, the amount of CO2 in the process remains unchanged (Figure 2 - points are arranged parallely to the axis X). This reflects the dominant reducing zone i ...
... drop in carbon dioxide content from 16% (for the gasification of RDF fuel only) to a very low level of 1÷2.5% when Bio-CONOx was added. In addition, the amount of CO2 in the process remains unchanged (Figure 2 - points are arranged parallely to the axis X). This reflects the dominant reducing zone i ...
Unit 1 Cycle 2: Interactions and Energy
... Since the chemical formulas, molar masses, and characteristic physical properties are clearly different for a starting material (calcium carbonate) and a product (carbon dioxide), this was a chemical change. ...
... Since the chemical formulas, molar masses, and characteristic physical properties are clearly different for a starting material (calcium carbonate) and a product (carbon dioxide), this was a chemical change. ...
Unit 8 Note Packet
... 2. Balance a chemical equation based upon the law of conservation of matter. 3. Analyze or draw a graph for the energy change of a chemical reaction. 4. Calculate the heat of solution for a given compound. We are looking for: 1a. Given the word equation/sentence, translate it into a formula chemical ...
... 2. Balance a chemical equation based upon the law of conservation of matter. 3. Analyze or draw a graph for the energy change of a chemical reaction. 4. Calculate the heat of solution for a given compound. We are looking for: 1a. Given the word equation/sentence, translate it into a formula chemical ...
chemistry 102 fall 2001 part 1
... 17&18. For a reaction where ∆H is +255 kJ/mol rxn and ∆S = +52 J/K, _______. (a) the reaction is spontaneous at all temperatures. (b) the reaction is nonspontaneous at all temperatures (c) the reaction is spontaneous only at temperatures above a certain value. (d) the reaction is spontaneous only at ...
... 17&18. For a reaction where ∆H is +255 kJ/mol rxn and ∆S = +52 J/K, _______. (a) the reaction is spontaneous at all temperatures. (b) the reaction is nonspontaneous at all temperatures (c) the reaction is spontaneous only at temperatures above a certain value. (d) the reaction is spontaneous only at ...
SODIUM HYDROGEN CARBONATE
... The CO2 can be recycled to produce more NaHCO3 by the Solvay process. In recent years, only about 15% of Na2CO3 was produced by the Solvay process. The remainder was mined from large deposits of trona, Na2CO3∙NaHCO3∙H2O, which were discovered in 1938 near the Green River in Wyoming. It is cheaper to ...
... The CO2 can be recycled to produce more NaHCO3 by the Solvay process. In recent years, only about 15% of Na2CO3 was produced by the Solvay process. The remainder was mined from large deposits of trona, Na2CO3∙NaHCO3∙H2O, which were discovered in 1938 near the Green River in Wyoming. It is cheaper to ...
reviewTWO
... What amount, in moles, of aluminium is needed to form 1.2mol of Al2O3(s)? b) What amount of oxygen gas is needed to react completely with 1.25 mol of aluminium? c) Calculate the amount of Al2O3(s)¬ formed when 6.5mol of oxygen reacts completely with aluminium. 3. Acetylene gas (C2H2) is produced in ...
... What amount, in moles, of aluminium is needed to form 1.2mol of Al2O3(s)? b) What amount of oxygen gas is needed to react completely with 1.25 mol of aluminium? c) Calculate the amount of Al2O3(s)¬ formed when 6.5mol of oxygen reacts completely with aluminium. 3. Acetylene gas (C2H2) is produced in ...
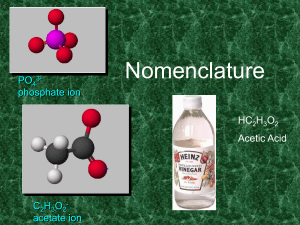
8B31A38F-1279-3B00-CDA90244BEA11A7B
... 2. Add prefixes to indicate # of atoms. Omit mono- prefix on the FIRST element. Mono- is OPTIONAL on the SECOND element (in this class, it’s NOT optional!). 3. Change the ending of the second element to -ide. ...
... 2. Add prefixes to indicate # of atoms. Omit mono- prefix on the FIRST element. Mono- is OPTIONAL on the SECOND element (in this class, it’s NOT optional!). 3. Change the ending of the second element to -ide. ...