Chemical Vapor Deposition (CVD)
... coatings, powders, fibers and monolithic components. • This technique is often used in many thin film applications. • By varying the experimental conditions—substrate material, substrate temperature, composition of the reaction gas mixture, total pressure gas flows, etc.— materials with different pr ...
... coatings, powders, fibers and monolithic components. • This technique is often used in many thin film applications. • By varying the experimental conditions—substrate material, substrate temperature, composition of the reaction gas mixture, total pressure gas flows, etc.— materials with different pr ...
Part A Completion
... Identify the origin of the electrical potential of a cell Explain the value of the standard reduction potential of the hydrogen half-cell Describe how the standard reduction potential of a half-cell is determined Interpret the meaning of a positive standard cell potential ...
... Identify the origin of the electrical potential of a cell Explain the value of the standard reduction potential of the hydrogen half-cell Describe how the standard reduction potential of a half-cell is determined Interpret the meaning of a positive standard cell potential ...
The shock tube as wave reactor for kinetic studies and material
... mention a few, techniques like interferometric, emission or absorption of molecules in the infrared, visible or ultraviolet spectral regions have been tried out before the more sensitive atomic resonance absorption spectroscopy (ARAS) and laser based diagnostic techniques emerged on the scene. The s ...
... mention a few, techniques like interferometric, emission or absorption of molecules in the infrared, visible or ultraviolet spectral regions have been tried out before the more sensitive atomic resonance absorption spectroscopy (ARAS) and laser based diagnostic techniques emerged on the scene. The s ...
Version A
... 52. When dilute nitric acid was added to a solution of one of the following chemicals, a gas was evolved, This gas turned a drop of limewater, Ca(OH)2, cloudy, due to the formation of a white precipitate. The chemical was ...
... 52. When dilute nitric acid was added to a solution of one of the following chemicals, a gas was evolved, This gas turned a drop of limewater, Ca(OH)2, cloudy, due to the formation of a white precipitate. The chemical was ...
ExamView - 1984 AP Chemistry Exam.tst
... (1) Test Questions are Copyright © 1984-2002 by College Entrance Examination Board, Princeton, NJ. All rights reserved. For face-to-face teaching purposes, classroom teachers are permitted to reproduce the questions. Web or Mass distribution prohibited. (2) AP® is a registered trademark of the Colle ...
... (1) Test Questions are Copyright © 1984-2002 by College Entrance Examination Board, Princeton, NJ. All rights reserved. For face-to-face teaching purposes, classroom teachers are permitted to reproduce the questions. Web or Mass distribution prohibited. (2) AP® is a registered trademark of the Colle ...
Determination of the Vapor Pressure of Triacetone Triperoxide (TATP)
... or 1.3 x 109 TNT molecules. There are about 13000 times as many molecules of TATP than TNT in the headspace at 25oC. The vapor pressure of TNT was first reported around 1950; at that time a Knudson effusion technique was used.10 In the late 1970’s TNT vapor pressure was re-determined using gas chrom ...
... or 1.3 x 109 TNT molecules. There are about 13000 times as many molecules of TATP than TNT in the headspace at 25oC. The vapor pressure of TNT was first reported around 1950; at that time a Knudson effusion technique was used.10 In the late 1970’s TNT vapor pressure was re-determined using gas chrom ...
Chemistry 133 Problem Set Introduction
... 1.77 Highly toxic hydrogen cyanide (HCN) has many industrial uses. As little as 1.0 × 10 –5 g/L is cause for concern. Calculate the number of grams of HCN at 1.0 × 10–5 g/L is in a room measuring 25 ft × 22 ft × 8.0 ft. 1.78 Nickel tetracarbonyl is one of the most toxic substances known. The recomme ...
... 1.77 Highly toxic hydrogen cyanide (HCN) has many industrial uses. As little as 1.0 × 10 –5 g/L is cause for concern. Calculate the number of grams of HCN at 1.0 × 10–5 g/L is in a room measuring 25 ft × 22 ft × 8.0 ft. 1.78 Nickel tetracarbonyl is one of the most toxic substances known. The recomme ...
CHE 1402 Lab Manual
... 4. Write the ideal-gas equation and give the units for each term when R = 0.0821 L-atm /mol-K. 5. Show by mathematical equations how one can determine the molar mass of a volatile liquid by measurement of the pressure, volume, temperature, and weight of the liquid. 6. If 0.75 g of a gas occupies 300 ...
... 4. Write the ideal-gas equation and give the units for each term when R = 0.0821 L-atm /mol-K. 5. Show by mathematical equations how one can determine the molar mass of a volatile liquid by measurement of the pressure, volume, temperature, and weight of the liquid. 6. If 0.75 g of a gas occupies 300 ...
Numerical solution methods for shock and detonation jump
... determine the properties of shock and detonation waves in a multi-component, reacting, ideal gas mixture. Only the idealized situations of perfect (constant heat-capacity) gases with fixed chemical energy release can be treated analytically (see the results given in Appendix A and B.1). Although wid ...
... determine the properties of shock and detonation waves in a multi-component, reacting, ideal gas mixture. Only the idealized situations of perfect (constant heat-capacity) gases with fixed chemical energy release can be treated analytically (see the results given in Appendix A and B.1). Although wid ...
sch103manual - university of nairobi staff profiles
... Under certain conditions of pressure and temperature, most substances can exist in any of the three states of matter: Solids, liquid or gas. Water for example, exists in the solid state as ice, liquid state as water and in the gaseous state as steam. The physical properties of a substance often depe ...
... Under certain conditions of pressure and temperature, most substances can exist in any of the three states of matter: Solids, liquid or gas. Water for example, exists in the solid state as ice, liquid state as water and in the gaseous state as steam. The physical properties of a substance often depe ...
Appendix
... glassware, and the repeatability of our measurements. In this appendix we take a more detailed look at the propagation of uncertainty, using the standardization of NaOH as an example. Standardizing a Solution of NaOH1 Because solid NaOH is an impure material, we cannot directly prepare a stock solut ...
... glassware, and the repeatability of our measurements. In this appendix we take a more detailed look at the propagation of uncertainty, using the standardization of NaOH as an example. Standardizing a Solution of NaOH1 Because solid NaOH is an impure material, we cannot directly prepare a stock solut ...
Chem 171-2-3: Final Exam Review Multiple Choice Problems 1
... In which one of the following substances is the kinetic energy greatest relative to the intermolecular forces of attraction? a. H2O (l) ...
... In which one of the following substances is the kinetic energy greatest relative to the intermolecular forces of attraction? a. H2O (l) ...
Lactose/D-Galactose
... adding D-galactose (qualitative or quantitative): if the absorbance is altered subsequent to the addition of the standard material, this is also an indication that no interference has occurred. The reaction cannot be restarted with lactose as, subsequent to altering the reaction conditions from pH 6 ...
... adding D-galactose (qualitative or quantitative): if the absorbance is altered subsequent to the addition of the standard material, this is also an indication that no interference has occurred. The reaction cannot be restarted with lactose as, subsequent to altering the reaction conditions from pH 6 ...
Lactose/D-Galactose
... adding D-galactose (qualitative or quantitative): if the absorbance is altered subsequent to the addition of the standard material, this is also an indication that no interference has occurred. The reaction cannot be restarted with lactose as, subsequent to altering the reaction conditions from pH 6 ...
... adding D-galactose (qualitative or quantitative): if the absorbance is altered subsequent to the addition of the standard material, this is also an indication that no interference has occurred. The reaction cannot be restarted with lactose as, subsequent to altering the reaction conditions from pH 6 ...
the chemical and physical properties of condensed
... more useful than the polyphosphate glasses and should be explored in greater detail during the next decade. Several areas deserve more attention than they have received. The three component M2O—MO—P2O5 phase systems should contain stable polyphosphates in the penta- to decaphosphate range. The curre ...
... more useful than the polyphosphate glasses and should be explored in greater detail during the next decade. Several areas deserve more attention than they have received. The three component M2O—MO—P2O5 phase systems should contain stable polyphosphates in the penta- to decaphosphate range. The curre ...
Electrospun Polyaniline Fibers as Highly Sensitive Room
... model is used to extract physical parameters from fitting experimental sensor data. The model is then used to illustrate the selection of optimal material design parameters for gas sensing by nanofibers. ...
... model is used to extract physical parameters from fitting experimental sensor data. The model is then used to illustrate the selection of optimal material design parameters for gas sensing by nanofibers. ...
File - IB CHEM NINJA
... rate of reaction (b) with time in establishing a chemical equilibrium In an equilibrium all of the species involved, both reactants and products, are present at a constant concentration. As a consequence, macroscopic properties of the system (that is those that can be observed or measured, such as i ...
... rate of reaction (b) with time in establishing a chemical equilibrium In an equilibrium all of the species involved, both reactants and products, are present at a constant concentration. As a consequence, macroscopic properties of the system (that is those that can be observed or measured, such as i ...
Unit 2: Matter as Solutions and Gases
... 2. Polar Molecule: - unequal charged distribution due to the electron pairs around the oxygen atom. 3. Strong O−H Hydrogen Bond: - a type of hydrogen bond that is fairly strong compared to other types of intermolecular bonds (bonds between molecules). ...
... 2. Polar Molecule: - unequal charged distribution due to the electron pairs around the oxygen atom. 3. Strong O−H Hydrogen Bond: - a type of hydrogen bond that is fairly strong compared to other types of intermolecular bonds (bonds between molecules). ...
5548-4.pdf
... Initially when the coating was produced in a high-activity aluminum pack, 97% of the coating consisted of the y phase (Ni2Al3). By heat treatment at the same temperature, the y phase was transformed to the h phase (NiAl), whose thickness and concentration profile were calculated. The assumption that ...
... Initially when the coating was produced in a high-activity aluminum pack, 97% of the coating consisted of the y phase (Ni2Al3). By heat treatment at the same temperature, the y phase was transformed to the h phase (NiAl), whose thickness and concentration profile were calculated. The assumption that ...
Chemistry
... one response which you want to mark on the Response Sheet. In case you feel that there is more than one correct response, mark the response which you consider the best. In any case, choose ONLY ONE response for each item. 5. In case you find any discrepancy in this test booklet in any question(s) or ...
... one response which you want to mark on the Response Sheet. In case you feel that there is more than one correct response, mark the response which you consider the best. In any case, choose ONLY ONE response for each item. 5. In case you find any discrepancy in this test booklet in any question(s) or ...
Unit- 5.pmd
... adsorption increases with the increase of surface area per unit mass of the adsorbent at a given temperature and pressure. Another important factor featuring adsorption is the heat of adsorption. During adsorption, there is always a decrease in residual forces of the surface, i.e., there is decrease ...
... adsorption increases with the increase of surface area per unit mass of the adsorbent at a given temperature and pressure. Another important factor featuring adsorption is the heat of adsorption. During adsorption, there is always a decrease in residual forces of the surface, i.e., there is decrease ...
Date: 16 / 01 / 2014 - Qatar University QSpace
... series of experiments were carried out to study the variation of preparation parameters such as support type, temperature, ‘ion-exchange’ time and the concentration of the (precursor) salt. ...
... series of experiments were carried out to study the variation of preparation parameters such as support type, temperature, ‘ion-exchange’ time and the concentration of the (precursor) salt. ...
Chemistry_Stoichiome..
... weight 114.9041 and abundance of 95.72 %. Which of the following isotopic weights is the most likely for the other isotope? a) 112.94 b) 115.90 c) 113.90 d) 114. 90 37. Suppose two elements X & Y combine to form two compounds XY2 & X2Y3 when 0.05 mole of XY2 weighs 5 g while 3.011 ×1023 molecules of ...
... weight 114.9041 and abundance of 95.72 %. Which of the following isotopic weights is the most likely for the other isotope? a) 112.94 b) 115.90 c) 113.90 d) 114. 90 37. Suppose two elements X & Y combine to form two compounds XY2 & X2Y3 when 0.05 mole of XY2 weighs 5 g while 3.011 ×1023 molecules of ...
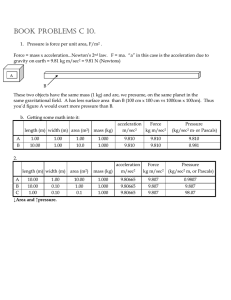
book problems c 10.
... and molecules. The "atoms" of nitrogen and oxygen are in reality "molecules" containing two atoms each. Thus two molecules of hydrogen can combine with one molecule of oxygen to produce two molecules of water. Avogadro suggested that equal volumes of all gases at the same temperature and pressure co ...
... and molecules. The "atoms" of nitrogen and oxygen are in reality "molecules" containing two atoms each. Thus two molecules of hydrogen can combine with one molecule of oxygen to produce two molecules of water. Avogadro suggested that equal volumes of all gases at the same temperature and pressure co ...
Exam 2 Key
... 6. How does the kinetic-molecular theory of gases help explain why a helium-filled balloon shrinks if it is taken outside on a cold winter day? (10 points) You should discuss how the kinetic energy of a gas depends on temperature. As T decreases, KE decreases. As KE drops, the average velocity of a ...
... 6. How does the kinetic-molecular theory of gases help explain why a helium-filled balloon shrinks if it is taken outside on a cold winter day? (10 points) You should discuss how the kinetic energy of a gas depends on temperature. As T decreases, KE decreases. As KE drops, the average velocity of a ...
Gas chromatography

Gas chromatography (GC) is a common type of chromatography used in analytical chemistry for separating and analyzing compounds that can be vaporized without decomposition. Typical uses of GC include testing the purity of a particular substance, or separating the different components of a mixture (the relative amounts of such components can also be determined). In some situations, GC may help in identifying a compound. In preparative chromatography, GC can be used to prepare pure compounds from a mixture.In gas chromatography, the mobile phase (or ""moving phase"") is a carrier gas, usually an inert gas such as helium or an unreactive gas such as nitrogen. The stationary phase is a microscopic layer of liquid or polymer on an inert solid support, inside a piece of glass or metal tubing called a column (a homage to the fractionating column used in distillation). The instrument used to perform gas chromatography is called a gas chromatograph (or ""aerograph"", ""gas separator"").The gaseous compounds being analyzed interact with the walls of the column, which is coated with a stationary phase. This causes each compound to elute at a different time, known as the retention time of the compound. The comparison of retention times is what gives GC its analytical usefulness.Gas chromatography is in principle similar to column chromatography (as well as other forms of chromatography, such as HPLC, TLC), but has several notable differences. First, the process of separating the compounds in a mixture is carried out between a liquid stationary phase and a gas mobile phase, whereas in column chromatography the stationary phase is a solid and the mobile phase is a liquid. (Hence the full name of the procedure is ""Gas–liquid chromatography"", referring to the mobile and stationary phases, respectively.) Second, the column through which the gas phase passes is located in an oven where the temperature of the gas can be controlled, whereas column chromatography (typically) has no such temperature control. Finally, the concentration of a compound in the gas phase is solely a function of the vapor pressure of the gas.Gas chromatography is also similar to fractional distillation, since both processes separate the components of a mixture primarily based on boiling point (or vapor pressure) differences. However, fractional distillation is typically used to separate components of a mixture on a large scale, whereas GC can be used on a much smaller scale (i.e. microscale).Gas chromatography is also sometimes known as vapor-phase chromatography (VPC), or gas–liquid partition chromatography (GLPC). These alternative names, as well as their respective abbreviations, are frequently used in scientific literature. Strictly speaking, GLPC is the most correct terminology, and is thus preferred by many authors.