Low-Temperature Alkaline pH Hydrolysis of Oxygen-Free
... is used to maintain the low pressure in the system. By using a high-voltage generator, an electric discharge (2 kV–23.2 mA) is applied on the gas mixture, which establishes a cold plasma that induces the dissociation of N2 and CH4 and other chemical reactions leading to the formation of tholins that ...
... is used to maintain the low pressure in the system. By using a high-voltage generator, an electric discharge (2 kV–23.2 mA) is applied on the gas mixture, which establishes a cold plasma that induces the dissociation of N2 and CH4 and other chemical reactions leading to the formation of tholins that ...
EIT Review S2012 Part 2 Dr. J. Mack CSUS Department of Chemistry
... The rate of effusion of a gas is proportional to its uRMS. over time, the mixture becomes homogeneous. ...
... The rate of effusion of a gas is proportional to its uRMS. over time, the mixture becomes homogeneous. ...
Document
... What would happen to the system if the pressure were decreased? A) Nothing would happen. B) More oxygen would be produced. C) The water vapor would become liquid water. D) The ammonia concentration would increase. E) The NO concentration would increase. ...
... What would happen to the system if the pressure were decreased? A) Nothing would happen. B) More oxygen would be produced. C) The water vapor would become liquid water. D) The ammonia concentration would increase. E) The NO concentration would increase. ...
Topical KCSE Mock-Chemistry Answers(15 Schools)
... Adds excess dilute hydrochloric acid/ sulphuric (vi) acid Filter to obtain copper metal Wash with distilled water To separate samples of CUO and charcoal in test tubes, dilute mineral acid is added with shaking CUO black dissolves to form blue solution ½ Charcoal does not dissolve in dilute mineral ...
... Adds excess dilute hydrochloric acid/ sulphuric (vi) acid Filter to obtain copper metal Wash with distilled water To separate samples of CUO and charcoal in test tubes, dilute mineral acid is added with shaking CUO black dissolves to form blue solution ½ Charcoal does not dissolve in dilute mineral ...
Chemical Engineering Thermodynamics II
... to observe that the electrical resistance of the warmer object decreases with time, and that of the colder block increases with time; eventually there would be no change in the electrical resistances of these objects. The two objects are then in thermal equilibiurm. They are at the same Temperature. ...
... to observe that the electrical resistance of the warmer object decreases with time, and that of the colder block increases with time; eventually there would be no change in the electrical resistances of these objects. The two objects are then in thermal equilibiurm. They are at the same Temperature. ...
Covalently Bonded Platinum(II) Complexes of [alpha]
... are direct consequences of the steric and electronic environment around the observed nuclei, and different values are therefore usually obtained, depending on the R group attached to the organometallic site. Thus, peptide functionalization with these complexes provides a biomarker not only for bioch ...
... are direct consequences of the steric and electronic environment around the observed nuclei, and different values are therefore usually obtained, depending on the R group attached to the organometallic site. Thus, peptide functionalization with these complexes provides a biomarker not only for bioch ...
Kinetic isotope effects of 12CH3D+OH and 13CH3D+OH from 278 to
... This was repeated 5–6 times. Ideally, concentration calculations from the spectral fits (data analysis described below) of the resulting spectra should give a linear fit with the slope of 1. The slope of these dilution tests is presented in Table 2. In an extra experiment with 12 CH3 D, N2 O (Air Li ...
... This was repeated 5–6 times. Ideally, concentration calculations from the spectral fits (data analysis described below) of the resulting spectra should give a linear fit with the slope of 1. The slope of these dilution tests is presented in Table 2. In an extra experiment with 12 CH3 D, N2 O (Air Li ...
Vinnitsa National Pirogov Memorial Medical University Biological
... What parts of chemistry did you study: inorganic chemistry yes/no, organic chemistry yes/no. After I had finished to study chemistry ______________ years/months passed. The students who studied chemistry at their countries we ask to read attentively the question given below. While answering the ques ...
... What parts of chemistry did you study: inorganic chemistry yes/no, organic chemistry yes/no. After I had finished to study chemistry ______________ years/months passed. The students who studied chemistry at their countries we ask to read attentively the question given below. While answering the ques ...
Chem Course Desc2. New
... 2) they know where to get the help, and 3) they act on that need to get the necessary help. This help may take the form of voluntary tutoring before or after school, assigned tutoring during school, or group study sessions. The vocabulary in chemistry is specific. For this reason, particular attent ...
... 2) they know where to get the help, and 3) they act on that need to get the necessary help. This help may take the form of voluntary tutoring before or after school, assigned tutoring during school, or group study sessions. The vocabulary in chemistry is specific. For this reason, particular attent ...
hydrogen storage
... cycle (Linde cycle). The gas is first compressed and then cooled in a heat exchanger, before it passes through a throttle valve where it undergoes an isenthalpic JouleThomson expansion, producing some liquid. The cooled gas is separated from the liquid and returned to the compressor via the heat exc ...
... cycle (Linde cycle). The gas is first compressed and then cooled in a heat exchanger, before it passes through a throttle valve where it undergoes an isenthalpic JouleThomson expansion, producing some liquid. The cooled gas is separated from the liquid and returned to the compressor via the heat exc ...
國 立 交 通 大 學
... Chemical vapor deposition (CVD) is a technique for synthesizing materials in which chemical components in vapor phase react to form a solid film at surfaces. The occurrence of a chemical reaction is essential to these means of film growth, as it is the requirement that reactants must start out in va ...
... Chemical vapor deposition (CVD) is a technique for synthesizing materials in which chemical components in vapor phase react to form a solid film at surfaces. The occurrence of a chemical reaction is essential to these means of film growth, as it is the requirement that reactants must start out in va ...
Fate of Fuel-Bound Nitrogen and Sulfur in Biomass-Fired
... concentrations of NH3 were measured above the fuel inlet. No significant reduction of the nitrogen species to N2 took place in the lower furnace, however, a drastic reduction was observed over the secondary air jet level. The final reduction of fuel bound nitrogen to N2 was over 90%, although only a ...
... concentrations of NH3 were measured above the fuel inlet. No significant reduction of the nitrogen species to N2 took place in the lower furnace, however, a drastic reduction was observed over the secondary air jet level. The final reduction of fuel bound nitrogen to N2 was over 90%, although only a ...
Stoichiometry - Mr Field's Chemistry Class
... If you get to a point where you just need a fractional amount of a compound, write it in and then multiply everything by the denominator of the fraction. ...
... If you get to a point where you just need a fractional amount of a compound, write it in and then multiply everything by the denominator of the fraction. ...
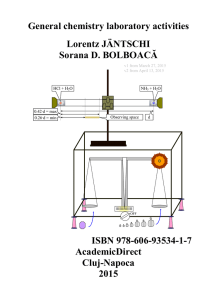
General chemistry laboratory activities, Lorentz
... accompanied by their necks, at the ends of which are ground glass joints to quickly and tightly connect to the rest of the apparatus (such as a reflux condenser or dropping funnel). The reaction flask is usually made of thick glass and they can tolerate large pressure differences, with the result th ...
... accompanied by their necks, at the ends of which are ground glass joints to quickly and tightly connect to the rest of the apparatus (such as a reflux condenser or dropping funnel). The reaction flask is usually made of thick glass and they can tolerate large pressure differences, with the result th ...
Chapter 2 - hrsbstaff.ednet.ns.ca
... group particles so that they know how many are present in a given mass of substance? On its own, the mass of a chemical is not very useful to a chemist. The chemical reactions that take place depend on the number of atoms present, not on their masses. Since atoms are far too small and numerous to co ...
... group particles so that they know how many are present in a given mass of substance? On its own, the mass of a chemical is not very useful to a chemist. The chemical reactions that take place depend on the number of atoms present, not on their masses. Since atoms are far too small and numerous to co ...
Study of C4F8 `N2 and C4F8 `Ar`N2 plasmas for highly
... in the etching of various substrate materials. This layer inhibits the ions or neutrals from directly reacting with the substrate. Normally, etch rates are thought to decrease with the fluorocarbon thickness.5 On the other hand, ion-induced defluorination of the fluorocarbon layer enhances the etch ...
... in the etching of various substrate materials. This layer inhibits the ions or neutrals from directly reacting with the substrate. Normally, etch rates are thought to decrease with the fluorocarbon thickness.5 On the other hand, ion-induced defluorination of the fluorocarbon layer enhances the etch ...
Carbon Dating Method
... number of radioactive events. They cannot identify the source of the activity. They only can operate successfully if it is assured that the detected event actually has originated in the 14C decay. That requires background suppression techniques. The main background is originated from the cosmic rays ...
... number of radioactive events. They cannot identify the source of the activity. They only can operate successfully if it is assured that the detected event actually has originated in the 14C decay. That requires background suppression techniques. The main background is originated from the cosmic rays ...
CHAPTER VI Fischer-Tropsch Refineries
... been reproduced directly from Refs.(2) and (4), and are copyright protected). in the tubes to regulate the temperature. The catalyst bed was typically operated 5-8°C higher than the temperature in the tubes and in practise the reactor temperature could be controlled to within 1°C in the range 170-20 ...
... been reproduced directly from Refs.(2) and (4), and are copyright protected). in the tubes to regulate the temperature. The catalyst bed was typically operated 5-8°C higher than the temperature in the tubes and in practise the reactor temperature could be controlled to within 1°C in the range 170-20 ...
Lecture notes
... Can behave as solvents dissolving a wide range of substances; also new industrial reaction medium. ...
... Can behave as solvents dissolving a wide range of substances; also new industrial reaction medium. ...
Oxygen Carriers Materials for Chemical
... an expensive and energy-intensive process resulting in a large decrease in efficiency. Thus there is a need to find cheaper and more efficient methods to perform the separation. Chemical-looping combustion (CLC) and chemical-looping reforming (CLR) are innovative technologies for power and hydrogen ...
... an expensive and energy-intensive process resulting in a large decrease in efficiency. Thus there is a need to find cheaper and more efficient methods to perform the separation. Chemical-looping combustion (CLC) and chemical-looping reforming (CLR) are innovative technologies for power and hydrogen ...
Document
... Isotopes Isotopes are atoms of an element with differing numbers number of neutrons Isotopes are (almost) undistinguishable in their chemical properties, because these are mostly determined by the electron shell However, isotopes differ in some of their physical properties (mass!) ...
... Isotopes Isotopes are atoms of an element with differing numbers number of neutrons Isotopes are (almost) undistinguishable in their chemical properties, because these are mostly determined by the electron shell However, isotopes differ in some of their physical properties (mass!) ...
å¾è湿çå¦
... 5. Carefully detach the last page. It is the datasheet. 6. Now answer the exam questions. Questions are not in order of difficulty. Indicate your choice on the STUDENT RESPONSE sheet by marking one letter beside the question number. • Mark only one answer for each question. • Questions are all of th ...
... 5. Carefully detach the last page. It is the datasheet. 6. Now answer the exam questions. Questions are not in order of difficulty. Indicate your choice on the STUDENT RESPONSE sheet by marking one letter beside the question number. • Mark only one answer for each question. • Questions are all of th ...
Mole Concept
... MASTERING PHYSICAL CHEMISTRY Rules for Working Limiting Reagent Problems Determine the number of moles of each reactant. ...
... MASTERING PHYSICAL CHEMISTRY Rules for Working Limiting Reagent Problems Determine the number of moles of each reactant. ...
kcse chemistry questions
... State what would be observed when dilute hydrochloric acid is added to the products formed when a mixture of iron fillings and sulphur? (1mk) Describe how the following reagents can be used to prepare lead sulphate solid potassium sulphate, solid lead carbonate, dilute nitric acid and distilled wate ...
... State what would be observed when dilute hydrochloric acid is added to the products formed when a mixture of iron fillings and sulphur? (1mk) Describe how the following reagents can be used to prepare lead sulphate solid potassium sulphate, solid lead carbonate, dilute nitric acid and distilled wate ...
OC 583- ISOTOPE BIGEOCHEMISTRY
... 3. Since isotopes have the same number of protons and electrons this means isotopes have same chemical behavior, i.e., all isotopic species enter into same chemical reactions and form the same bonds -in this sense, the rare or less abundant isotopic species is an excellent tracer of the abundant iso ...
... 3. Since isotopes have the same number of protons and electrons this means isotopes have same chemical behavior, i.e., all isotopic species enter into same chemical reactions and form the same bonds -in this sense, the rare or less abundant isotopic species is an excellent tracer of the abundant iso ...
Gas chromatography

Gas chromatography (GC) is a common type of chromatography used in analytical chemistry for separating and analyzing compounds that can be vaporized without decomposition. Typical uses of GC include testing the purity of a particular substance, or separating the different components of a mixture (the relative amounts of such components can also be determined). In some situations, GC may help in identifying a compound. In preparative chromatography, GC can be used to prepare pure compounds from a mixture.In gas chromatography, the mobile phase (or ""moving phase"") is a carrier gas, usually an inert gas such as helium or an unreactive gas such as nitrogen. The stationary phase is a microscopic layer of liquid or polymer on an inert solid support, inside a piece of glass or metal tubing called a column (a homage to the fractionating column used in distillation). The instrument used to perform gas chromatography is called a gas chromatograph (or ""aerograph"", ""gas separator"").The gaseous compounds being analyzed interact with the walls of the column, which is coated with a stationary phase. This causes each compound to elute at a different time, known as the retention time of the compound. The comparison of retention times is what gives GC its analytical usefulness.Gas chromatography is in principle similar to column chromatography (as well as other forms of chromatography, such as HPLC, TLC), but has several notable differences. First, the process of separating the compounds in a mixture is carried out between a liquid stationary phase and a gas mobile phase, whereas in column chromatography the stationary phase is a solid and the mobile phase is a liquid. (Hence the full name of the procedure is ""Gas–liquid chromatography"", referring to the mobile and stationary phases, respectively.) Second, the column through which the gas phase passes is located in an oven where the temperature of the gas can be controlled, whereas column chromatography (typically) has no such temperature control. Finally, the concentration of a compound in the gas phase is solely a function of the vapor pressure of the gas.Gas chromatography is also similar to fractional distillation, since both processes separate the components of a mixture primarily based on boiling point (or vapor pressure) differences. However, fractional distillation is typically used to separate components of a mixture on a large scale, whereas GC can be used on a much smaller scale (i.e. microscale).Gas chromatography is also sometimes known as vapor-phase chromatography (VPC), or gas–liquid partition chromatography (GLPC). These alternative names, as well as their respective abbreviations, are frequently used in scientific literature. Strictly speaking, GLPC is the most correct terminology, and is thus preferred by many authors.



![Covalently Bonded Platinum(II) Complexes of [alpha]](http://s1.studyres.com/store/data/022412983_1-66c66ee18551a43164a79702fd995f95-300x300.png)