Nuclear Chemistry
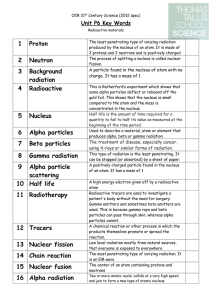
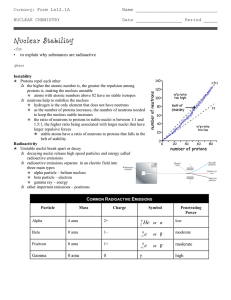
... A Beta particle is an electron created and emitted when a neutron is transformed* into a proton and an electron during radioactive decay. This action adds a proton and thus changes the identity of the atom. The mass number stays the same. ...
... A Beta particle is an electron created and emitted when a neutron is transformed* into a proton and an electron during radioactive decay. This action adds a proton and thus changes the identity of the atom. The mass number stays the same. ...
File - Dr. Wall`s Science
... • Put experiment in desk drawer, where there was no light • Image still showed up on paper, due to energy from minerals • This energy is nuclear radiation ...
... • Put experiment in desk drawer, where there was no light • Image still showed up on paper, due to energy from minerals • This energy is nuclear radiation ...
Chemistry: Matter and Change
... • Gamma rays (short wavelength) are photons, which are high-energy • Gamma rays have no mass or charge so the emission of gamma rays does not change the atomic number or mass number of a nucleus. • Gamma rays almost always accompany alpha and beta radiation. • X rays are a form of high-energy electr ...
... • Gamma rays (short wavelength) are photons, which are high-energy • Gamma rays have no mass or charge so the emission of gamma rays does not change the atomic number or mass number of a nucleus. • Gamma rays almost always accompany alpha and beta radiation. • X rays are a form of high-energy electr ...
3 Background radiation
... A high energy electron given off by a radioactive atom Radioactive tracers are used to investigate a patient's body without the need for surgery. Gamma emitters and sometimes beta emitters are used. This is because gamma rays and beta particles can pass through skin, whereas alpha particles cannot. ...
... A high energy electron given off by a radioactive atom Radioactive tracers are used to investigate a patient's body without the need for surgery. Gamma emitters and sometimes beta emitters are used. This is because gamma rays and beta particles can pass through skin, whereas alpha particles cannot. ...
nuclear test 2006
... Carbon Dating is a technique used to determine the age of organic material. The activity (rate of decay) of 14C atoms in the sample is measured. 14C has a half life of 5730 years. a) Explain how the activity of 14C in an organic sample can be used to determine its age. ...
... Carbon Dating is a technique used to determine the age of organic material. The activity (rate of decay) of 14C atoms in the sample is measured. 14C has a half life of 5730 years. a) Explain how the activity of 14C in an organic sample can be used to determine its age. ...
Chp 7.1 Atomic Theory and Radioactive Decay
... • Natural background radiation exists all around us. • This radiation consists of high energy particles or waves being emitted from a variety of materials. ...
... • Natural background radiation exists all around us. • This radiation consists of high energy particles or waves being emitted from a variety of materials. ...
Name
... 1. The minimum mass of fissionable material in a reactor or nuclear bomb that will sustain a chain reaction. 5. A self-sustaining reaction in which the products of one reaction event stimulate further reaction events 8. The tendency of some elements, such as uranium, to emit radiation as a result of ...
... 1. The minimum mass of fissionable material in a reactor or nuclear bomb that will sustain a chain reaction. 5. A self-sustaining reaction in which the products of one reaction event stimulate further reaction events 8. The tendency of some elements, such as uranium, to emit radiation as a result of ...
File - Ms M - EARL MARRIOTT SECONDARY
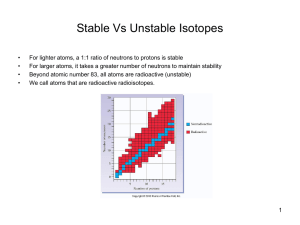
... Atomic mass = (0.932 x 39) + (0.001 x 40) + (0.067 x 41) = 39.1 Unlike all previously discovered chemical reactions, radioactivity sometimes results in the formation of completely new atoms. ...
... Atomic mass = (0.932 x 39) + (0.001 x 40) + (0.067 x 41) = 39.1 Unlike all previously discovered chemical reactions, radioactivity sometimes results in the formation of completely new atoms. ...
radioisotopes and radiotherapy - video
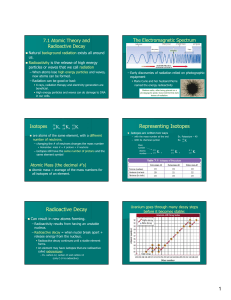
... 1. Isotopes are nuclides with the same number of protons but different number of neutrons. Isotopes have same atomic number but different mass number. 2. Radioisotopes are unstable isotopes, where their nuclei decay to a more stable state or type of nuclei, giving off matter and energy as radiation. ...
... 1. Isotopes are nuclides with the same number of protons but different number of neutrons. Isotopes have same atomic number but different mass number. 2. Radioisotopes are unstable isotopes, where their nuclei decay to a more stable state or type of nuclei, giving off matter and energy as radiation. ...
Ionizing radiation
Ionizing (or ionising in British English) radiation is radiation that carries enough energy to free electrons from atoms or molecules, thereby ionizing them. Ionizing radiation is made up of energetic subatomic particles, ions or atoms moving at relativistic speeds, and electromagnetic waves on the high-energy end of the electromagnetic spectrum.Gamma rays, X-rays, and the higher ultraviolet part of the electromagnetic spectrum are ionizing, whereas the lower ultraviolet part of the electromagnetic spectrum, visible light (including nearly all types of laser light), infrared, microwaves, and radio waves are considered non-ionizing radiation. The boundary between ionizing and non-ionizing electromagnetic radiation that occurs in the ultraviolet is not sharply defined, since different molecules and atoms ionize at different energies. Conventional definition places the boundary at a photon energy between 10 eV and 33 eV in the ultraviolet (see definition boundary section below).Typical ionizing subatomic particles from radioactivity include alpha particles, beta particles and neutrons. Almost all products of radioactive decay are ionizing because the energy of radioactive decay is typically far higher than that required to ionize. Other subatomic ionizing particles which occur naturally are muons, mesons, positrons, neutrons and other particles that constitute the secondary cosmic rays that are produced after primary cosmic rays interact with Earth's atmosphere. Cosmic rays may also produce radioisotopes on Earth (for example, carbon-14), which in turn decay and produce ionizing radiation.Cosmic rays and the decay of radioactive isotopes are the primary sources of natural ionizing radiation on Earth referred to as background radiation.In space, natural thermal radiation emissions from matter at extremely high temperatures (e.g. plasma discharge or the corona of the Sun) may be ionizing. Ionizing radiation may be produced naturally by the acceleration of charged particles by natural electromagnetic fields (e.g. lightning), although this is rare on Earth. Natural supernova explosions in space produce a great deal of ionizing radiation near the explosion, which can be seen by its effects in the glowing nebulae associated with them.Ionizing radiation can also be generated artificially using X-ray tubes, particle accelerators, and any of the various methods that produce radioisotopes artificially.Ionizing radiation is invisible and not directly detectable by human senses, so radiation detection instruments such as Geiger counters are required. However, ionizing radiation may lead to secondary emission of visible light upon interaction with matter, such as in Cherenkov radiation and radioluminescence.Ionizing radiation is applied constructively in a wide variety of fields such as medicine, research, manufacturing, construction, and many other areas, but presents a health hazard if proper measures against undesired exposure aren't followed. Exposure to ionizing radiation causes damage to living tissue, and can result in mutation, radiation sickness, cancer, and death.