Diapositiva 1
... evolutionary separation justify their recognition as a unique protein category These proteins are characterized by an almost complete lack of folded structure and an extended conformation with high intramolecular flexibility and little secondary structure. ...
... evolutionary separation justify their recognition as a unique protein category These proteins are characterized by an almost complete lack of folded structure and an extended conformation with high intramolecular flexibility and little secondary structure. ...
Enzyme Mechanisms
... system is the ribosome. The central catalytic activity of the ribosome (peptide bond formation) is catalyzed by an RNA component. ...
... system is the ribosome. The central catalytic activity of the ribosome (peptide bond formation) is catalyzed by an RNA component. ...
Some factors affecting polyphenol oxidase activity
... the enzyme to "mold" itself over the substrate. ...
... the enzyme to "mold" itself over the substrate. ...
HERE - Oregon State University
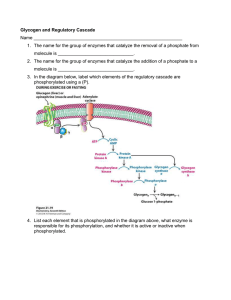
... 1. The name for the group of enzymes that catalyze the removal of a phosphate from molecule is ______________________________. 2. The name for the group of enzymes that catalyze the addition of a phosphate to a molecule is ______________________________. 3. In the diagram below, label which elements ...
... 1. The name for the group of enzymes that catalyze the removal of a phosphate from molecule is ______________________________. 2. The name for the group of enzymes that catalyze the addition of a phosphate to a molecule is ______________________________. 3. In the diagram below, label which elements ...
27-36
... of the antibiotic. The molecular mass of penicillase is 30,000g/mol. The turnover number of the enzyme at 28°C is 2,000 s-1. If 6.4μg of penicillase catalyzes the destruction of 3.11mg of amoxicillin, an antibiotic with a molecular mass of 364 g/mol, in 20s at 28°C, how many active sites does the en ...
... of the antibiotic. The molecular mass of penicillase is 30,000g/mol. The turnover number of the enzyme at 28°C is 2,000 s-1. If 6.4μg of penicillase catalyzes the destruction of 3.11mg of amoxicillin, an antibiotic with a molecular mass of 364 g/mol, in 20s at 28°C, how many active sites does the en ...
Phytic acid continued - Whole Grain Connection
... the milling process will result in corresponding alterations in this distribution pattern. A particular property of phytic acid is that it binds mineral ions of potassium, calcium, magnesium, iron and zinc, which are naturally present in the wheat grain, sometimes in association with protein. The lo ...
... the milling process will result in corresponding alterations in this distribution pattern. A particular property of phytic acid is that it binds mineral ions of potassium, calcium, magnesium, iron and zinc, which are naturally present in the wheat grain, sometimes in association with protein. The lo ...
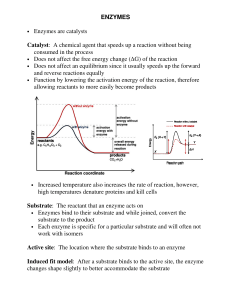
ENZYMES • Enzymes are catalysts Catalyst: A chemical agent that
... • Receptor site remote from the active site that binds with a regulator that can either inhibit or stimulate an enzyme’s activities • Usually present in proteins with quaternary structure • Activator – A substance that stabilizes the protein and keeps the active sites available • Allosteric inhibito ...
... • Receptor site remote from the active site that binds with a regulator that can either inhibit or stimulate an enzyme’s activities • Usually present in proteins with quaternary structure • Activator – A substance that stabilizes the protein and keeps the active sites available • Allosteric inhibito ...
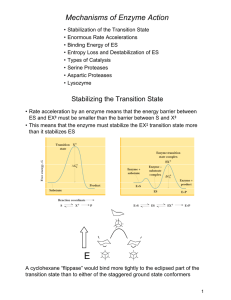
Mechanisms of Enzyme Action - Chemistry at Winthrop University
... However, a successful drug must be able to kill the virus in a human subject without blocking other essential proteases in the body ...
... However, a successful drug must be able to kill the virus in a human subject without blocking other essential proteases in the body ...
Event Poster PDF
... studies often reveal a change in enzyme structure from an open to a closed state after substrate binding, which could be explained by the need for an open site to bind substrate, but a closed state to align residues for catalysis. Enzyme specificity is a kinetic phenomena that cannot be addressed by ...
... studies often reveal a change in enzyme structure from an open to a closed state after substrate binding, which could be explained by the need for an open site to bind substrate, but a closed state to align residues for catalysis. Enzyme specificity is a kinetic phenomena that cannot be addressed by ...
Enzymes
... Enzymes are biological catalysts. Recall that a catalyst speeds up the rate of a reaction by decreasing the activation energy needed for the reaction to occur. A catalyst remains unchanged in a reaction and does not affect the amount of product formed, but only the rate at which the product forms. A ...
... Enzymes are biological catalysts. Recall that a catalyst speeds up the rate of a reaction by decreasing the activation energy needed for the reaction to occur. A catalyst remains unchanged in a reaction and does not affect the amount of product formed, but only the rate at which the product forms. A ...
Enzyme Control of Metabolic Pathways
... The end products of one reaction very often become the starting substrate for a second reaction or pathway. The consumption of the end products may stimulate the first reaction. In this way an equilibrium situation is avoided and metabolic pathways are continuously restimulated. Each step of a metab ...
... The end products of one reaction very often become the starting substrate for a second reaction or pathway. The consumption of the end products may stimulate the first reaction. In this way an equilibrium situation is avoided and metabolic pathways are continuously restimulated. Each step of a metab ...
Catalytic Strategies
... • Different bacteria share common gene sequences and a similar active site conformation ...
... • Different bacteria share common gene sequences and a similar active site conformation ...
Lecture 5: Metabolic Pathways and beyond
... aminoacids on their surface: this process is called phosphorylation. ...
... aminoacids on their surface: this process is called phosphorylation. ...
A change in temperature affects an enzymatic reaction because
... The square molecule is activing as a competitive inhibitor The square molecule is acting as an allosteric inhibitor The substrate will bond to the allosteric site The end product of the reaction will stop the enzyme from working ...
... The square molecule is activing as a competitive inhibitor The square molecule is acting as an allosteric inhibitor The substrate will bond to the allosteric site The end product of the reaction will stop the enzyme from working ...
Alg13,14 GAO - Stony Brook University
... Name and History In LLO synthetic pathway, N-acetylglucosaminyldiphosphodolichol N-acetylglucosaminyltransferase catalyzes the transfer of a β1, 4-linked GlcNAc from UDP-GlcNAc to GlcNAc-pp-dol to produce GlcNAc2-PP-dol. Early purification and biochemical studies of this enzyme have been performed ...
... Name and History In LLO synthetic pathway, N-acetylglucosaminyldiphosphodolichol N-acetylglucosaminyltransferase catalyzes the transfer of a β1, 4-linked GlcNAc from UDP-GlcNAc to GlcNAc-pp-dol to produce GlcNAc2-PP-dol. Early purification and biochemical studies of this enzyme have been performed ...
Enzyme Regulation Strategies
... c. Despite the fact that they are very specific – that each kinase recognizes a certain protein, the mechanism by which they do this is very common. d. Kinases are pretty large –about 260 residues make up the kinase core domain e. One way that they regulate themselves is by intrasteric control i. De ...
... c. Despite the fact that they are very specific – that each kinase recognizes a certain protein, the mechanism by which they do this is very common. d. Kinases are pretty large –about 260 residues make up the kinase core domain e. One way that they regulate themselves is by intrasteric control i. De ...
Enzyme Catalysis - Studentportalen
... Frequently, in the transition state, the substrate and the enzyme have slightly different structure (strain or distortion) and increase the reactivity of the substrate. ...
... Frequently, in the transition state, the substrate and the enzyme have slightly different structure (strain or distortion) and increase the reactivity of the substrate. ...
ENZYMES - Bio12.com
... This reaction lies near the beginning of the respiration pathway in cells The end product of respiration is ATP If there is a lot of ATP in the cell this enzyme is inhibited Respiration slows down and less ATP is produced As ATP is used up the inhibition stops and the reaction speeds up again ...
... This reaction lies near the beginning of the respiration pathway in cells The end product of respiration is ATP If there is a lot of ATP in the cell this enzyme is inhibited Respiration slows down and less ATP is produced As ATP is used up the inhibition stops and the reaction speeds up again ...
Chapter 2-ROLE OF ENZYMES
... together as shown in the diagram? 12. What happens to the active site of an enzyme at high temperatures? 13. What pH does pepsin enzyme work best at in the stomach? 14. What word is used to describe the best set of conditions that an enzyme can work at? 15. What happens to the rate of reaction if th ...
... together as shown in the diagram? 12. What happens to the active site of an enzyme at high temperatures? 13. What pH does pepsin enzyme work best at in the stomach? 14. What word is used to describe the best set of conditions that an enzyme can work at? 15. What happens to the rate of reaction if th ...
Amoeba Sisters Video Recap: Enzymes
... _________________________________________________ 3. Describe the effects that enzymes can have on substrates. _________________________________________________ _________________________________________________ _________________________________________________ _______________________________________ ...
... _________________________________________________ 3. Describe the effects that enzymes can have on substrates. _________________________________________________ _________________________________________________ _________________________________________________ _______________________________________ ...
The effect of calcium on conformational change of thimet
... and homeostasis of the heart. Also, TOP cleaves an important peptide involved in Alzheimer’s disease, so understanding this enzyme and how it works could be important in new findings about this so far incurable disease. Investigating the role of calcium on enzyme conformation and substrate specifici ...
... and homeostasis of the heart. Also, TOP cleaves an important peptide involved in Alzheimer’s disease, so understanding this enzyme and how it works could be important in new findings about this so far incurable disease. Investigating the role of calcium on enzyme conformation and substrate specifici ...
Enzymes
... Enzymes Enzymes are globular proteins, with a specific tertiary structure, which catalyse metabolic reactions in living organisms. They may be intracellular or extracellular Describe, with the aid of diagrams, the mechanism of action of enzyme molecules, with reference to specificity, active site, l ...
... Enzymes Enzymes are globular proteins, with a specific tertiary structure, which catalyse metabolic reactions in living organisms. They may be intracellular or extracellular Describe, with the aid of diagrams, the mechanism of action of enzyme molecules, with reference to specificity, active site, l ...