Lesson 23. Clinical enzymology
... 23.3.3.2 Lactate Dehydrogenase (EC 1.1.1.27; L-lactate: NAD+ oxidoreductase; LD) catalyses the reversible interconversion of lactate and pyruvate. The enzyme is widely distributed in the body, with high concentrations in cells of cardiac and skeletal muscle, liver, kidney, brain and erythrocytes: me ...
... 23.3.3.2 Lactate Dehydrogenase (EC 1.1.1.27; L-lactate: NAD+ oxidoreductase; LD) catalyses the reversible interconversion of lactate and pyruvate. The enzyme is widely distributed in the body, with high concentrations in cells of cardiac and skeletal muscle, liver, kidney, brain and erythrocytes: me ...
Catalysts in biochemical reactions
... Enzyme is a class of proteins that function as catalysts in biochemical reactions. On their Characteristics is that they increases the rate of reactions by several orders of magnitude A very dramatic example of enzyme kinetics is given by decomposition of hydrogen Peroxide. Enzymes are usually prote ...
... Enzyme is a class of proteins that function as catalysts in biochemical reactions. On their Characteristics is that they increases the rate of reactions by several orders of magnitude A very dramatic example of enzyme kinetics is given by decomposition of hydrogen Peroxide. Enzymes are usually prote ...
What are the different types of enzymes
... What are Enzymes? Enzymes are substances that function as organic catalysts, in other words, they either start chemical reactions or make them run faster. They accomplish this while remaining unchanged themselves. Enzymes are composed of two parts, a protein portion called the apoenzyme and a nonpro ...
... What are Enzymes? Enzymes are substances that function as organic catalysts, in other words, they either start chemical reactions or make them run faster. They accomplish this while remaining unchanged themselves. Enzymes are composed of two parts, a protein portion called the apoenzyme and a nonpro ...
Pre-‐lab AP Lab: Enzyme Catalysis Name
... You will calculate the rate using Milliliters of Oxygen collected during 30 second intervals. The above calculation will use milliliters instead of µmoles in your calculation of reaction rate. The above calculations may easily be seen on the AP EXAM. The rate of a chemical reaction may be studied in ...
... You will calculate the rate using Milliliters of Oxygen collected during 30 second intervals. The above calculation will use milliliters instead of µmoles in your calculation of reaction rate. The above calculations may easily be seen on the AP EXAM. The rate of a chemical reaction may be studied in ...
Enzymes revision
... Small changes in pH outside the optimum can cause small reversible changes in enzyme structure and results in inactivation. Extremes of pH can denature the enzyme. The charges on the amino acid side-chains of the enzyme’s active site are affected by free hydrogen ions or hydroxyl ions. In the format ...
... Small changes in pH outside the optimum can cause small reversible changes in enzyme structure and results in inactivation. Extremes of pH can denature the enzyme. The charges on the amino acid side-chains of the enzyme’s active site are affected by free hydrogen ions or hydroxyl ions. In the format ...
1.4 Enzymes
... Small changes in pH outside the optimum can cause small reversible changes in enzyme structure and results in inactivation. Extremes of pH can denature the enzyme. The charges on the amino acid side-chains of the enzyme’s active site are affected by free hydrogen ions or hydroxyl ions. In the format ...
... Small changes in pH outside the optimum can cause small reversible changes in enzyme structure and results in inactivation. Extremes of pH can denature the enzyme. The charges on the amino acid side-chains of the enzyme’s active site are affected by free hydrogen ions or hydroxyl ions. In the format ...
Analysis of Single Ionizing Group
... Analysis of Single Ionizing Group Similarily, suppose one group on the enzyme has to be protonated for activity: ...
... Analysis of Single Ionizing Group Similarily, suppose one group on the enzyme has to be protonated for activity: ...
Lecture * 4 The Kinetics of Enzyme
... Modulation and Regulation of Enzyme Activity • enzyme-substrate inhibitors systems classify by their influence on the Michaelis-Menten equation parameters vmax and Km • Reversible inhibitors are termed competitive if their presence increases the value of Km but does not alter vmax The effect of suc ...
... Modulation and Regulation of Enzyme Activity • enzyme-substrate inhibitors systems classify by their influence on the Michaelis-Menten equation parameters vmax and Km • Reversible inhibitors are termed competitive if their presence increases the value of Km but does not alter vmax The effect of suc ...
Enzyme Inhibition
... Enzymes are NOT used up in a reaction Enzymes may be used again over and over again (so ...
... Enzymes are NOT used up in a reaction Enzymes may be used again over and over again (so ...
Enzyme Activity
... 20. List 2 internal environmental factors that affect how well enzymes function. 21. What happens to water when you heat it to 100°C? 22. What happens to proteins dissolved in that water when you heat it to 100°C? 23. What specific change happens to an enzyme that stops it from working when you heat ...
... 20. List 2 internal environmental factors that affect how well enzymes function. 21. What happens to water when you heat it to 100°C? 22. What happens to proteins dissolved in that water when you heat it to 100°C? 23. What specific change happens to an enzyme that stops it from working when you heat ...
University of Groningen Characterization of 4,6
... enzyme may thus find industrial application in synthesis of IMMP modified starch. However, the expression level of soluble enzymes, which is preferable for industrial production, has remained unsatisfactorily low. The LAB GS are homologous to 4,6-α-GTase enzymes, and deletion of (parts of) their N-t ...
... enzyme may thus find industrial application in synthesis of IMMP modified starch. However, the expression level of soluble enzymes, which is preferable for industrial production, has remained unsatisfactorily low. The LAB GS are homologous to 4,6-α-GTase enzymes, and deletion of (parts of) their N-t ...
lab1
... certain RNA molecules can be effective biocatalysts too. These RNA molecules have come to be known as ribozymes. synthesized by the living cells ...
... certain RNA molecules can be effective biocatalysts too. These RNA molecules have come to be known as ribozymes. synthesized by the living cells ...
Rate of enzymatic reactions
... the enzymatic reaction, this is so because when more enzyme molecules are present, more substrate molecules can be acted upon at the same time producing more products (providing that the substrate concentration is limitless). 4. Effect of PH: The point where the enzyme is most active - is known as ...
... the enzymatic reaction, this is so because when more enzyme molecules are present, more substrate molecules can be acted upon at the same time producing more products (providing that the substrate concentration is limitless). 4. Effect of PH: The point where the enzyme is most active - is known as ...
Enzymes 1. All cells in multicellular organisms contain thousands of
... 7. As the graph shows, pepsin only remains active up to a pH of about 4.8. If the pH is any higher than this, the pepsin's folds will become so distorted that its active site will no longer function. Thus, pepsin is only able to catalyze the digestion of proteins in the highly acidic environment of ...
... 7. As the graph shows, pepsin only remains active up to a pH of about 4.8. If the pH is any higher than this, the pepsin's folds will become so distorted that its active site will no longer function. Thus, pepsin is only able to catalyze the digestion of proteins in the highly acidic environment of ...
Catalase Enzyme Lab
... 1. Place 2 ml of the 3% hydrogen peroxide solution into a clean test tube. 2. Add a small piece of liver to the test tube. Observe the bubbles, what gas is being released? _______ Throughout this investigation you will estimate the rate of the reaction (how rapidly the solution bubbles) on a scale o ...
... 1. Place 2 ml of the 3% hydrogen peroxide solution into a clean test tube. 2. Add a small piece of liver to the test tube. Observe the bubbles, what gas is being released? _______ Throughout this investigation you will estimate the rate of the reaction (how rapidly the solution bubbles) on a scale o ...
Exploration Activity: Enzymes
... Below you’ll find graphs that show how pH affects four different enzymes. For each, explain the trend you see, and where the enzyme is most efficient, shown by the optimum, or best pH. I. Chymotrypsin (enzyme that digests proteins):: ...
... Below you’ll find graphs that show how pH affects four different enzymes. For each, explain the trend you see, and where the enzyme is most efficient, shown by the optimum, or best pH. I. Chymotrypsin (enzyme that digests proteins):: ...
Enzyme. Kinetics Mechanisms of enzyme action Specificity and
... (amino group), transmethylases (a methyl group), transacetylases (an acetyl group). Splitting chemical bonds with water: Hydrolases - esterases, nucleases, deaminases, amidases, and proteases. Splitting different chemical bonds without water: Lyases (decarboxylases, dehydratases). Changing geometry ...
... (amino group), transmethylases (a methyl group), transacetylases (an acetyl group). Splitting chemical bonds with water: Hydrolases - esterases, nucleases, deaminases, amidases, and proteases. Splitting different chemical bonds without water: Lyases (decarboxylases, dehydratases). Changing geometry ...
Energy/Chemical Energy in the Cell Chapter 5
... • chemical binds to the enzyme at area other than the active site • this alters the enzyme’s shape enough so the substrate doesn’t fit well, or at all, and therefore the rate of reaction slows down or stops • adding more substrate does not help (increase the rate) since the active site is non-functi ...
... • chemical binds to the enzyme at area other than the active site • this alters the enzyme’s shape enough so the substrate doesn’t fit well, or at all, and therefore the rate of reaction slows down or stops • adding more substrate does not help (increase the rate) since the active site is non-functi ...
Enzyme Lab - marric.us
... INTRODUCTION: What would happen to your cells if they made a poisonous chemical as a byproduct? You might think that they would die. In fact, your cells are always making poisonous chemicals. They do not die because your cells use enzymes to break down these poisonous chemicals into harmless substan ...
... INTRODUCTION: What would happen to your cells if they made a poisonous chemical as a byproduct? You might think that they would die. In fact, your cells are always making poisonous chemicals. They do not die because your cells use enzymes to break down these poisonous chemicals into harmless substan ...
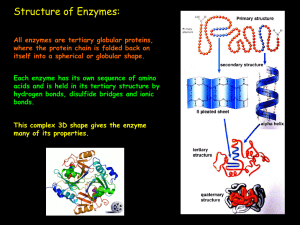
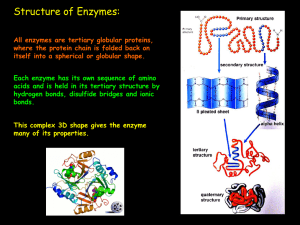
3+7 – HL Enzymes Page 1 1. Structure of Enzymes Like all proteins
... sucrose and the water must collide with enough energy to break the glycosidic bond. To catalyse this reaction, the enzyme Sucrase could be added which would lower the activation energy. Sucrose and water collide with one another and form an unstable high-energy intermediate that quickly changes into ...
... sucrose and the water must collide with enough energy to break the glycosidic bond. To catalyse this reaction, the enzyme Sucrase could be added which would lower the activation energy. Sucrose and water collide with one another and form an unstable high-energy intermediate that quickly changes into ...
File
... Enzymes are NOT used up in a reaction Enzymes may be used again over and over again (so ...
... Enzymes are NOT used up in a reaction Enzymes may be used again over and over again (so ...
What are Enzymes?
... can easily read the directions and answer questions concerning the chemistry of life. Students will be divided into 3 teams (Team A,B, C). Team A will go first. If they answer correctly they get full points (3). If they answer incorrectly, Team B can answer for 1 point, and attempt to answer the nex ...
... can easily read the directions and answer questions concerning the chemistry of life. Students will be divided into 3 teams (Team A,B, C). Team A will go first. If they answer correctly they get full points (3). If they answer incorrectly, Team B can answer for 1 point, and attempt to answer the nex ...
Enzyme Kinetics Lab
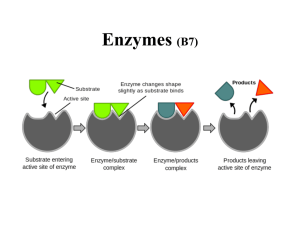
... Enzymes are a class of proteins that greatly speed up (catalyze) reactions between specific substances, usually at their functional groups. The substances that each type of enzyme acts upon are called its substrates. Enzymes have four common features: 1. They don’t make anything happen that couldn’t ...
... Enzymes are a class of proteins that greatly speed up (catalyze) reactions between specific substances, usually at their functional groups. The substances that each type of enzyme acts upon are called its substrates. Enzymes have four common features: 1. They don’t make anything happen that couldn’t ...
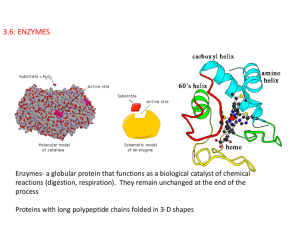
3.6: ENZYMES
... contain many +/- regions, some around the active site. An excess of H+ ions in an acidic solution can lead to bonding between the H+ ions and the negative charges in the active site. (same with OH- in basic solutions where the OH- ions bond to the positive sites). This will inhibit the matching proc ...
... contain many +/- regions, some around the active site. An excess of H+ ions in an acidic solution can lead to bonding between the H+ ions and the negative charges in the active site. (same with OH- in basic solutions where the OH- ions bond to the positive sites). This will inhibit the matching proc ...
Enzymes - Bioclass
... The number of occupied active site is increasing and there is competition for the active site. (c) The rate is constant. The enzyme active site is fully saturated with substrate such that adding more substrate does not increase the rate of reaction. The enzymes molecules are fully occupied convertin ...
... The number of occupied active site is increasing and there is competition for the active site. (c) The rate is constant. The enzyme active site is fully saturated with substrate such that adding more substrate does not increase the rate of reaction. The enzymes molecules are fully occupied convertin ...